Extensively Drug-Resistant Carbapenemase-Producing Pseudomonas aeruginosa and Medical Tourism from the United States to Mexico, 2018–2019
Ian Kracalik
1
, D. Cal Ham
1, Gillian McAllister, Amanda R. Smith, Maureen Vowles, Kelly Kauber, Melba Zambrano, Gretchen Rodriguez, Kelley Garner, Kaitlyn Chorbi, P. Maureen Cassidy, Shannon McBee, Rhett J. Stoney, Kathleen Moser, Margarita E. Villarino, Oscar E. Zazueta, Amelia Bhatnagar, Erisa Sula, Richard A. Stanton, Allison C. Brown, Alison L. Halpin, Lauren Epstein, Maroya Spalding Walters, and
for the Verona Integron-Encoded Metallo-β-Lactamase–Producing Carbapenem-Resistant Pseudomonas aeruginosa Medical Tourism Investigation Team2
Author affiliations: Centers for Disease Control and Prevention, Atlanta, Georgia, USA (I. Kracalik, D. Cal Ham, G. McAllister, R.J. Stoney, K. Moser, M.E. Villarino, A. Bhatnagar, E. Sula, R.A. Stanton, A.C. Brown, A.L. Halpin, L. Epstein, M. Spalding Walters); Utah Department of Health, Salt Lake City, Utah, USA (A.R. Smith, M. Vowles); Washington State Department of Health, Olympia, Washington, USA (K. Kauber); Texas Department of State Health Services, Austin, Texas, USA (M. Zambrano, G. Rodriguez); Arkansas Department of Health, Little Rock, Arkansas, USA (K. Garner); Arizona Department of Health Services, Phoenix, Arizona, USA (K. Chorbi); Oregon Health Authority, Portland, Oregon, USA (P.M. Cassidy); West Virginia Department of Health and Human Resources, Charleston, West Virginia, USA (S. McBee); Secretaría de Salud de Baja California, Mexicali, Mexico (O.E. Zazueta)
Main Article
Figure 2
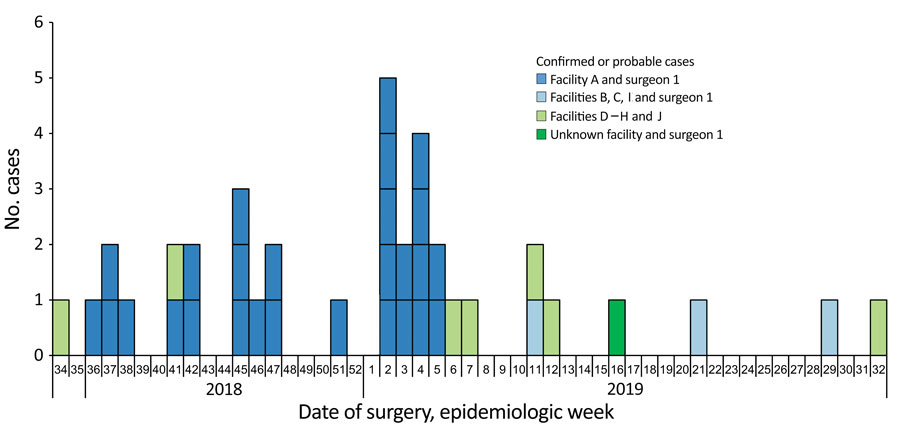
Figure 2. Confirmed and probable cases of infection with Verona integron‒encoded, metallo-β-lactamase‒producing, carbapenem-resistant Pseudomonas aeruginosa, by week of surgery, among US medical tourists undergoing elective invasive procedures in Tijuana, Mexico, January 2018‒December 2019. Dark blue bars show cases associated with surgery performed at facility A by surgeon 1; light green bars show cases associated with surgery at facilities D–H and J by surgeons other than surgeon 1; and light blue bars show cases associated with facilities B, C, and I by surgeon 1; and dark green bar shows a case associated with surgeon 1 and an unknown facility. A confirmed case was isolation of Verona integron‒encoded, metallo-β-lactamase‒producing, carbapenem-resistant P. aeruginosa from a patient who had an elective invasive medical procedure in Mexico during January 2018–December 2019 and within 45 days before specimen collection. A probable case was isolation of carbapenem-resistant P. aeruginosa, with an isolate unavailable for carbapenemase testing, from a patient who had an elective invasive medical procedure in Mexico during January 2018–December 2019 and within 45-days before specimen collection. No cases were identified from patients who underwent surgery before August 2018 (week 34). The peak of the outbreak encompassed epidemiologic weeks 2–5 (January 2019).
Main Article
Page created: October 28, 2021
Page updated: December 20, 2021
Page reviewed: December 20, 2021
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.
