Volume 28, Number 11—November 2022
Research
Prevalence of Histoplasmosis among Persons with Advanced HIV Disease, Nigeria
Cite This Article
Citation for Media
Abstract
We sought to determine the prevalence of probable disseminated histoplasmosis among advanced HIV disease (AHD) patients in Nigeria. We conducted a cross-sectional study in 10 sites across 5 of 6 geopolitical zones in Nigeria. We identified patients with CD4 cell counts <200 cells/mm3 or World Health Organization stage 3 or 4 disease who also had >2 clinical features of disseminated histoplasmosis, and we tested them for Histoplasma antigen using a Histoplasma enzyme immune assay. Of 988 participants we recruited, 76 (7.7%) were antigen-positive. The 76 Histoplasma antigen–positive participants had significantly lower (p = 0.03) CD4 counts; 9 (11.8%) were also co-infected with tuberculosis. Most antigen-positive participants (50/76; 65.8%; p = 0.015) had previously received antiretroviral treatment; 26/76 (34.2%) had not. Because histoplasmosis is often a hidden disease among AHD patients in Nigeria, Histoplasma antigen testing should be required in the AHD package of care.
Histoplasmosis, an invasive fungal infection endemic in the Americas, Africa, and Asia, with a few cases reported among immigrants to Europe, was classified as an AIDS-defining disease in 1987 (1,2). Incidence of disseminated histoplasmosis is 5%–25% in persons with advanced HIV disease (AHD; World Health Organization [WHO]–preferred term for AIDS), and according to recent data from South America, mortality rates are similar to those for tuberculosis among this patient group (3,4). In Latin America, high prevalence rates have been reported for disseminated histoplasmosis in AHD populations in Brazil (22%; 123/570) and Mexico (30%; 85/288) (5,6). Histoplasmosis is the most common AIDS-defining infection in Guatemala, more common than tuberculosis (7). In a recent study from Cameroon, 26% (36/138) of HIV patients had Histoplasma antigen in their urine regardless of CD4 count; a 2015 report indicated a 13% (7/56) prevalence in the AHD population (8,9).
WHO in 2020 published its first guidelines for disseminated histoplasmosis among persons with AHD, including recommendations for diagnosis (10). WHO and the US President’s Emergency Plan for AIDS Relief (PEPFAR) recommend providing differentiated care tailored to the unique needs of different HIV patient populations. Screening, treatment, and prophylaxis for major opportunistic infections is recommended for AHD (10). These key evidence-based interventions reduce illness and death among this clinically unstable population. Nigeria recently adopted a package of care for AHD that includes histoplasmosis screening, which has yet to be implemented. Nigeria has the 7th highest global tuberculosis rate and, because histoplasmosis is commonly misdiagnosed as tuberculosis (11), the histoplasmosis rate in Nigeria is likely higher than currently estimated. A recent review also revealed that Nigeria had 124 documented historical cases of histoplasmosis, the highest number in Africa, but almost all were described before the HIV pandemic began (12). Therefore, the effect of histoplasmosis on AHD in Nigeria is largely unknown. Our primary objective was to determine the prevalence of histoplasmosis among AHD patients in Nigeria and to generate data that will help with designing and implementing guidelines for differentiated care.
We conducted a cross-sectional survey in 10 sites across large areas of Nigeria during November 2019–June 2021. The geopolitical zones we captured were South East (site: Enugu), South West (Lagos and Ibadan), South South (Benin, Port Harcourt, and Calabar), North Central (Bida, Jos, and Makurdi), and North West (Sokoto). Because of insurgent activities and security challenges, North East was excluded. We included antiretroviral treatment (ART) clinics and infectious disease units, in partnership with the AIDS Prevention Initiative in Nigeria program and other implementing partners in the zones; all sites included tertiary facilities (teaching hospitals). On the basis of data from the national database, selected sites all had >30% AHD prevalence among their overall populations. Five of the sites—Ibadan, Port Harcourt, Enugu, Jos, and Calabar—had histoplasmosis cases reported before the HIV epidemic (12); in 1 city, Benin, Histoplasma exposure had recently been determined by positive histoplasmin skin tests (13). The other 4 sites had no documented cases of histoplasmosis.
We obtained ethics clearance from national and institutional ethics committees before recruiting participants and received permission to contact patients from principal investigators or coordinators of the ART programs at each site. Managing clinicians assisted in recruiting participants. We obtained informed written consent from each study participant after adequately explaining the study and its objectives.
We recruited both ART-naive and ART-exposed outpatient or hospitalized HIV-infected patients who had a CD4 count <200 cells/mm3 and met other inclusion criteria. Inclusion criteria were presence of AHD and >2 of 6 features commonly seen in patients with disseminated histoplasmosis: fever, chronic cough, weight loss, cutaneous lesions, oral ulcers, and diarrhea. Among participants in 1 study, 93.8% had fever, 87% weight loss, 76% cough; and 53.4% diarrhea (14). Whenever possible, we collected 2 urine samples from each participant with an interval of 1 week between collections. We collected other relevant biologic samples (sputum, bronchoalveolar lavage, skin lesion biopsy, and whole blood specimen) and stored them at −80°C for future research, including histologic and genomic studies.
Case Definitions
For our study we used the WHO AHD definition of CD4 cell count <200 cells/mm3 or WHO stage 3 or 4 disease in adults and adolescents (15). We followed the European Organisation for Research and Treatment of Cancer Mycoses Study Group consensus definition for probable disseminated histoplasmosis as a Histoplasma antigenuria–positive test in the presence of compatible clinical findings (16).
Data Gathering and Response
We interviewed participants and reviewed their medical records and charts using a standardized checklist. This checklist encompassed sociodemographic characteristics, signs and symptoms, occupational history or exposure (e.g., gardening, civil construction, agriculture), recreational and travel history (e.g., visits to caves or farms, travel to South America), physical examination findings, working diagnoses (including the presence of other opportunistic infections), laboratory and imaging investigations, and current medications (including ARTs).
We collected participant urine samples in sterile universal screw cap containers and transported them with ice packs in refrigerator bags. Specimens were batched and stored for <2 months at −20°C before being shipped to a central laboratory for sample processing. We tested for urine Histoplasma antigen using the Clarus IMMY Histoplasma GM Enzyme immune assay from Immuno-Mycologics (https://www.immy.com) according to manufacturer instructions. We used the 9 standard positive control range and set an optical density cutoff value of 2.0 on the basis of a 4-parameter graph. We collected sputum samples from participants suspected of having tuberculosis because of signs or symptoms, such as cough, weight loss, fever, or other suggestive syndromes, and tested the samples for tuberculosis using the Cepheid Xpert MTB/RIF assay (https://www.cepheid.com).
We communicated positive Histoplasma antigenuria results to managing clinicians and advised them to manage those patients with a probable diagnosis of disseminated histoplasmosis according to current standard-of-care guidelines. We contacted positive participants who had been hospitalized when recruited but released by the time testing results were received to schedule an outpatient clinic visit to propose a treatment plan. Duration of follow-up varied among sites; the longest recorded follow-up duration was 30 days for a study participant receiving antifungal therapy for treatment of histoplasmosis. Treatment, intravenous amphotericin B deoxycholate for 2 weeks followed by oral itraconazole until adequate immune reconstitution occurred, was rarely given because of logistic and financial constraints. Patients were provided ART according to national treatment guidelines.
Data Analysis
We entered all clinical and laboratory results into a spreadsheet and subsequently analyzed data by using SPSS Statistics 21 (https://www.ibm.com). We used descriptive statistics to summarize the data and determine mean, SD, median, interquartile range [IQR], and minimum and maximum for continuous variables. We determined absolute and relative frequencies to summarize categorical variables and used χ2 testing to check for associations and either a 2-sample or paired-sample t-test to compare continuous variables. We stratified results by ART status (naive, experienced, failed treatment), demographics, and clinical features. We used p<0.05 as the cutoff for significant associations.
Sociodemographic and Clinical Data
We recruited 988 participants, 377 (38.2%) male and 611 (61.8%) female, across 10 sites (Table 1); 685 (69.3%) were outpatients, 303 (30.7%) hospitalized. All participants had clinical symptoms suggestive of tuberculosis or histoplasmosis as stipulated in the inclusion criteria. Median age was 39 years (IQR 32–47 years). The most common age range for study participants was 25–40 years (n = 484; 48.9%); 80 (8.1%) were <25 years of age, 16 (1.6%) of those 13–19 years of age, and 43 (4.4%) were >60 years of age. Among participants, 259 (26.3%) had completed tertiary education and 216 (22%) had no formal education. We classified occupations into 6 groups (Table 1); the largest proportion (n = 437; 44.2%) were professionals, followed by unskilled laborers (n = 320; 32.4%), with pensioners (n = 13; 1.3%) the least common.
Histoplasmosis and Study Outcomes
We found 76 participants had Histoplasma antigenuria, a 7.7% prevalence of probable disseminated histoplasmosis; 44 (57.9%) were female and 32 (42.1%) were male. Among the 76 positive cases, both the first and second samples were positive in 45 (59.2%); 6 (7.9%) participants whose first samples were positive never returned to have a second test. Most (51.3%) participants with probable disseminated histoplasmosis were in the 41–60-year age range; 47.3% were <40 years of age (Table 1). The South West zone of Nigeria had the highest rate of probable histoplasmosis (12.9%), while the North Central had the lowest prevalence (5.6%). Across the various study sites, Ibadan in the South West zone had the highest rate, 20%; Benin in the South South had the lowest prevalence, 1.4% (Appendix Figure). Among possible risk factors, only occupation (p<0.001; Table 1) and smoking (p = 0.037; Table 2) were significantly associated with histoplasmosis.
Probable disseminated histoplasmosis participants had significantly lower CD4 counts (p = 0.03), and almost half, 36/76 (47.4%), had been hospitalized and had a median CD4 count of 96 cells/mm3 (IQR 40.75–176.00 cells/mm3) compared with nonhistoplasmosis participants, 128 cells/mm3 (IQR 70–180 cells/mm3) (Table 3). Prevalence of probable histoplasmosis was not significantly higher among hospitalized participants, 30/303 (9.9%), than outpatients, 46/685 (6.7%; p = 0.505) Conversely, the association between tuberculosis and participant group was significant (p<0.001); hospitalized patients (59/303, 21.6%) tested positive for tuberculosis more frequently than did outpatients (58/685, 8.5%). Fifty (65.8%) participants with probable histoplasmosis were ART experienced (p = 0.015), whereas the other 26 (34.2%) were ART naive.
Most (788, 79.8%) study participants experienced weight loss, among whom 60 (7.6%) were positive for Histoplasma urinary antigen; 551 (55.8%) had a cough, 40 (7.3%) of whom were antigen positive. Among 102 (10.3%) participants with cutaneous lesions, only 5 (4.9%) tested positive for Histoplasma urinary antigen. No clinical signs or symptoms distinguished tuberculosis from disseminated histoplasmosis (Table 4). Using the Xpert MTB/RIF assay, we identified 117 (11.8%) participants who tested positive for tuberculosis.
Among participants, 420 (42.5%) had CD4 cell counts of 101–200 cells/mm3 (IQR 126.00–181.25 cells/mm3), but only 23/420 (5.5%) were Histoplasma urinary antigen positive; 303 (30.7%) were classified as having WHO clinical stage 3 or 4 disease, of whom 30/303 (9.9%) were antigen positive (Table 3). Despite comprising only 129/988 (13.1%), the lowest number of participants, those in the 0–50 cells/mm3 CD4 cell count group, had the highest (15/129, 11.6%) frequency of Histoplasma urinary antigen positivity (Table 3).
Eleven (1.1%) participants died during the 30-day study period, 2 from Port Harcourt and 9 from Ibadan; 2/76 (2.6%) were positive for Histoplasma antigenuria, 1 co-infected with Mycobacterium tuberculosis. The other 9 who died had negative tests for histoplasmosis and tuberculosis; cause of death was not determined in these cases.
Histoplasmosis and Tuberculosis Coinfection
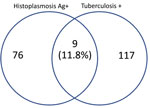
Nine (11.8%) participants, 8 female, had both histoplasmosis and tuberculosis. Co-infection occurred in all age groups. Seven of the participants were stage 3 or 4 HIV patients and 2 had 101–200/mm3 CD4 counts (Table 3). One co-infected participant, a 32-year-old hospitalized patient with a working diagnosis of stage 4 HIV with pulmonary tuberculosis, died during the course of the study (Figure).
Although histoplasmosis is endemic in Nigeria, published data have been restricted to case reports that largely predate the HIV era. Disease incidence has not been well characterized, especially among persons living with AHD (1). We attempted to determine the frequency of disseminated histoplasmosis in the HIV population of Nigeria and found that >7% of persons with AHD have probable histoplasmosis on the basis of European Organisation for Research and Treatment of Cancer Mycoses Study Group consensus definitions.
The countrywide prevalence of Histoplasma antigenuria among AHD patients in this study was <26% in Cameroon and <14% in South Africa (8,17). Whereas we used a previously validated monoclonal Histoplasma galactomannan enzyme immunosorbent assays to detect Histoplasma antigen (18), the study from Cameroon used a different commercial assay. It has been acknowledged that using a higher cutoff would have been more realistic and would have changed prevalence to 8%, which is closer to our findings. The lower rate in our study may have been because of technical factors such as length and conditions of storage of urine samples because specimens had to be transferred to a central location for testing to optimize the use of the antigen detection kits. Furthermore, the studies (8,17) were both conducted in single locations in South Africa and Cameroon that might both have been hyperendemic for histoplasmosis. Our multicenter study showed regional variability with prevalence ranging from 5.6% in the North Central zone to 12.9% in the South West. Even within regions, prevalence varied widely from site to site. Such variability was similarly described among regions in a multicenter study conducted in Brazil (5) that demonstrated a pooled prevalence of 21.6% from 14 centers, far exceeding the pooled prevalence from Nigeria. However, in the study from Brazil, use of the antigen detection method was combined with classical mycology tests including culturing, whereas we used only antigen detection (5). Laboratory tests for histoplasmosis are seldom performed in Nigeria because of a combination of lack of awareness, facilities, biosafety cabinets, and staff with the expertise needed to perform isolator methods of blood culturing and other laboratory testing.
Histoplasmin skin sensitivity rates predict the level of exposure to Histoplasma spp. in a given geographic location (13,19,20). Surprisingly, antigenuria prevalence did not correlate well with histoplasmin reactivity rates observed in a previous multicenter survey (13) that included 4 of the sites in our study: Benin City, Calabar, Ibadan, and Lagos. Benin, which recorded the highest skin sensitivity in the previous study (13), ended up with the lowest antigenuria prevalence in our study. It is noteworthy that in the histoplasmin sensitivity survey (13), skin sensitivity was significantly associated with study site. A corresponding association between site and outcome of interest, Histoplasma antigenuria, was not demonstrated in this study, which suggests that other factors, such as the extent of immunosuppression, may have played a greater role in determining antigenuria prevalence. The histoplasmin employed in the skin sensitivity study is known to be cross-reactive for H. capsulatum var. capsulatum and H. capsulatum var. duboisii both of which cause disseminated histoplasmosis in persons with AHD and are present in Nigeria (19,20). On the other hand, there is no evidence that the EIA deployed in this study, or any other antigen detection method for that matter, reliably detects H. capsulatum var. duboisii. In a review of histoplasmosis caused by H. capsulatum var. duboisii, diagnosis relied mostly on direct examination of body fluids and skin scrapings or histopathologic examination of clinical specimens; few were confirmed by culture or PCR and none relied on Histoplasma antigen detection (21). Because of this methodologic variability among studies, the effect on observed antigenuria prevalence of Histoplasma spp. distribution in the various study sites deserves further investigation.
As observed in other studies, exposure to classic environmental risk factors such as caves, heavy construction, fruit trees, and poultry were not notable risk factors for antigenuria in this study (8,22). However, contrary to findings from Cameroon, occupation was linked to positivity, with some skilled laborers, including painters, electricians, and plumbers, being more at risk than others (8). Another notable risk factor was smoking. Although not historically associated with progressive disseminated histoplasmosis, smoking has been recognized as a risk factor for the chronic pulmonary form of the disease (22).
Co-infection occurs commonly in AHD patients who have progressive disseminated histoplasmosis. Multiple studies from the Americas report tuberculosis as the most common coinfection (23–27). In the index cohort, 11.8% of participants with antigenuria had tuberculosis co-infection, which is close to the tuberculosis co-infection rate of 15.4% of participants with histoplasmosis in Brazil and 13.1% in Guatemala, both high-burden tuberculosis countries (5,28). The fact that histoplasmosis is often mistaken for and can coexist with tuberculosis is a substantial confounder in areas where the diseases are coendemic. Because tuberculosis awareness has grown and diagnostics have become more readily available, a diagnosis of tuberculosis alone might explain the signs and symptoms similar between the diseases, hiding diagnosis of the more obscure and neglected histoplasmosis in AHD patients. This shortfall suggests the need for active histoplasmosis screening in persons suspected to have tuberculosis, irrespective of confirmation with GeneXpert or other diagnostics. It is also critical to ensure that patients who screen positive for histoplasmosis can receive treatment. Several participants found to have probable histoplasmosis in this study were not treated because of financial constraints. Therefore, histoplasmosis treatment should also be included in the AHD package of care.
We found that 6.6% of participants with antigenuria had skin lesions, similar to what was found in Cameroon (6%) (8). However, among participants with lesions, histoplasma urinary antigen was no more common (p = 0.329). Skin lesions, which occur in 10%–25% of AIDS patients with disseminated histoplasmosis, have been linked with genetic variation among specific strains of the fungus that are dermatotropic or might be markers of histoplasmosis diagnosis when made at a very late stage (29). When present, biopsied lesions provide useful specimens for diagnostic confirmation of histoplasmosis. However, lesions were not very common among participants in our study, requiring us to use more available specimens. In addition, the skilled personnel needed to perform these biopsies might not be available in some settings.
One major strength of this study was that we included sites in virtually all the geopolitical zones in Nigeria that have had the most reported cases of histoplasmosis in the past. However, a study limitation was our lack of the mycology data from cultures or PCR needed to provide definitive proof of histoplasmosis and clarify the relative contributions of H. capsulatum var. capsulatum and H. capsulatum var. duboisii to its prevalence in Nigeria. Second, because it is unclear whether detecting Histoplasma antigen in urine provides reliable data for diagnosing H. capsulatum var. duboisii–caused histoplasmosis, we might have underestimated histoplasmosis prevalence. Third, the possibility of false positive antigenuria results cannot be entirely ruled out; however, although not tested on samples from the settings in our study, the assay we used has been validated in several studies to have good sensitivity and specificity. Fourth, our selection criteria increased the pretest probability for histoplasmosis among this cohort of participants. Fifth, we might have recorded some false-negative results as a consequence of the prolonged storage of samples. Another limitation was the lack of detailed ancillary tests, such as lactate dehydrogenase, aminotransferase, alkaline phosphatase, ferritin, and complete blood counts, which would have helped characterize patients.
Much remains to be elucidated about histoplasmosis in Nigeria, but this study confirms that it is certainly underreported among persons with HIV and AIDS, partly obscured by a diagnosis of tuberculosis, a disease with several manifestations in common with histoplasmosis. Further research using highly sensitive diagnostic approaches such as PCR and bone marrow examination is needed to gain insight into the precise epidemiology of the disease in Nigeria. To encourage proactive searching for histoplasmosis, use of specific diagnostic tools, including culturing, needs to be scaled up and management guidelines for AHD patients revised. After diagnosis, patients should be treated with appropriate antifungal agents, following the 2020 WHO guidelines. Patients suspected or confirmed to have tuberculosis should be investigated for histoplasmosis as well. Development of a molecular test in an easy-to-use format, such as the GeneXpert platform, that could be deployed in HIV treatment centers would be welcome.
In conclusion, histoplasmosis is not uncommon among AHD patients in Nigeria. Therefore, Histoplasma antigen screening should be included in the AHD package of care as a matter of urgent need to improve efficiency of diagnosis and reduce illness and death from histoplasmosis in an at-risk population.
Dr. Oladele is an associate professor of medical microbiology at the Department of Medical Microbiology and Parasitology, University of Lagos, and a consultant clinical microbiologist at the Lagos University Teaching Hospital, both in Lagos. Her research interests include epidemiology of fungal infections and health systems strengthening for diagnosis of serious fungal diseases.
Acknowledgments
We thank Tina Nwosu for processing the samples and collating laboratory data.
This study was funded by a grant from Gilead Sciences. IMMY supported this study with donations of test kits.
R.O.O. provided conceptualization, data curation, formal analysis, methodology, resources, validation, and writing of the original draft, review, and editing. I.I.O., A.S.A., O.A.A., B.E.E., Y.M., M.A.A.-W, M.O.O., U.S.U., and A.D.-J. provided data curation, resources, writing, review, and editing. S.T.A., I.E.A., and D.I. provided data curation and resources. D.W.D., A.C.P., and T.C. provided resources, writing, review, and editing.
References
- Ekeng BE, Edem K, Amamilo I, Panos Z, Denning D, Oladele RO. Histoplasmosis in children; HIV/AIDS not a major driver. J Fungi (Basel). 2021;7:530. DOIPubMedGoogle Scholar
- Sepúlveda VE, Márquez R, Turissini DA, Goldman WE, Matute DR. Genome sequences reveal cryptic speciation in the human pathogen Histoplasma capsulatum. MBio. 2017;8:e01339–17. DOIPubMedGoogle Scholar
- Silva TC, Treméa CM, Zara ALSA, Mendonça AF, Godoy CSM, Costa CR, et al. Prevalence and lethality among patients with histoplasmosis and AIDS in the Midwest Region of Brazil. Mycoses. 2017;60:59–65. DOIPubMedGoogle Scholar
- Pasqualotto AC, Quieroz-Telles F. Histoplasmosis dethrones tuberculosis in Latin America. Lancet Infect Dis. 2018;18:1058–60. DOIPubMedGoogle Scholar
- Falci DR, Monteiro AA, Braz Caurio CF, Magalhães TCO, Xavier MO, Basso RP, et al. Histoplasmosis, an underdiagnosed disease affecting people living with HIV/AIDS in Brazil: results of a multicenter prospective cohort study using both classical mycology tests and Histoplasma urine antigen detection. Open Forum Infect Dis. 2019;6:
ofz073 . DOIPubMedGoogle Scholar - Torres-González P, Niembro-Ortega MD, Martínez-Gamboa A, Ahumada-Topete VH, Andrade-Villanueva J, Araujo-Meléndez J, et al. Diagnostic accuracy cohort study and clinical value of the Histoplasma urine antigen (ALPHA Histoplasma EIA) for disseminated histoplasmosis among HIV infected patients: A multicenter study. PLoS Negl Trop Dis. 2018;12:
e0006872 . DOIPubMedGoogle Scholar - Medina N, Rodriguez-Tudela JL, Aguirre L, Salazar LR, Gamboa O, Bonilla O, et al. Incidence of histoplasmosis in a cohort of people with HIV: from estimations to reality. Microorganisms. 2021;9:2596. DOIPubMedGoogle Scholar
- Kuate MPN, Nyasa R, Mandengue C, Tendongfor N, Bongomin F, Denning DW. Screening for acute disseminated histoplasmosis in HIV disease using urinary antigen detection enzyme immunoassay: A pilot study in Cameroon. J Microbiol Methods. 2021;185:
106226 . DOIPubMedGoogle Scholar - Mandengue CE, Ngandjio A, Atangana PJA. Histoplasmosis in HIV-infected persons, Yaoundé, Cameroon. Emerg Infect Dis. 2015;21:2094–6. DOIPubMedGoogle Scholar
- Guidelines for diagnosing and managing disseminated histoplasmosis among people living with HIV. Washington, DC: Pan American Health Organization, and Geneva: World Health Organization; 2020.
- Global tuberculosis report 2018. Geneva: World Health Organization; 2018.
- Oladele RO, Ayanlowo OO, Richardson MD, Denning DW. Histoplasmosis in Africa: An emerging or a neglected disease? PLoS Negl Trop Dis. 2018;12:
e0006046 . DOIPubMedGoogle Scholar - Oladele RO, Toriello C, Ogunsola FT, Ayanlowo OO, Foden P, Fayemiwo AS, et al. Prior subclinical histoplasmosis revealed in Nigeria using histoplasmin skin testing. PLoS One. 2018;13:
e0196224 .DOIPubMedGoogle Scholar - Brilhante RS, Fechine MA, Mesquita JR, Cordeiro RA, Rocha MF, Monteiro AJ, et al. Histoplasmosis in HIV-positive patients in Ceará, Brazil: clinical-laboratory aspects and in vitro antifungal susceptibility of Histoplasma capsulatum isolates. Trans R Soc Trop Med Hyg. 2012;106:484–8. DOIPubMedGoogle Scholar
- World Health Organisation. Interim WHO clinical staging of HIV/AIDS and HIV/AIDS case definitions for surveillance: African Region [cited 2022 Aug 3]. https://apps.who.int/iris/bitstream/handle/10665/69058/WHO_HIV_2005.02.pdf
- Donnelly JP, Chen SC, Kauffman CA, Steinbach WJ, Baddley JW, Verweij PE, et al. Revision and Update of the Consensus Definitions of Invasive Fungal Disease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020;71:1367–76. DOIPubMedGoogle Scholar
- van Schalkwyk E, Mhlanga M, Maphanga TG, Mpembe RS, Shillubane A, Iyaloo S, et al. Screening for invasive fungal disease using non-culture-based assays among inpatients with advanced HIV disease at a large academic hospital in South Africa. Mycoses. 2020;63:478–87. DOIPubMedGoogle Scholar
- Cáceres DH, Samayoa BE, Medina NG, Tobón AM, Guzmán BJ, Mercado D, et al. Multicenter validation of commercial antigenuria reagents to diagnose progressive disseminated histoplasmosis in people living with HIV/AIDS in two Latin American countries. J Clin Microbiol. 2018;56:e01959–17. DOIPubMedGoogle Scholar
- Gugnani HC, Egere JU, Larsh H. Skin sensitivity to capsulatum and duboisii histoplasmins in Nigeria. J Trop Med Hyg. 1991;94:24–6.PubMedGoogle Scholar
- Gugnani HC. Histoplasmosis in Africa: a review. Indian J Chest Dis Allied Sci. 2000;42:271–7.PubMedGoogle Scholar
- Develoux M, Amona FM, Hennequin C. Histoplasmosis caused by Histoplasma capsulatum var. duboisii: a comprehensive review of cases from 1993 to 2019. Clin Infect Dis. 2021;73:e543–9. DOIPubMedGoogle Scholar
- Ekeng BE, Oladele RO, Emanghe UE, Ochang EA, Mirabeau TY. Prevalence of histoplasmosis and molecular characterization of histoplasma species in patients with presumptive pulmonary tuberculosis in Calabar, Nigeria. Open Forum Infect Dis. 2022;9:ofac368.
- Caceres DH, Tobón ÁM, Restrepo Á, Chiller T, Gómez BL. The important role of co-infections in patients with AIDS and progressive disseminated histoplasmosis (PDH): A cohort from Colombia. Med Mycol Case Rep. 2018;19:41–4. DOIPubMedGoogle Scholar
- Gutierrez ME, Canton A, Sosa N, Puga E, Talavera L. Disseminated histoplasmosis in patients with AIDS in Panama: a review of 104 cases. Clin Infect Dis. 2005;40:1199–202. DOIPubMedGoogle Scholar
- Mora DJ, dos Santos CT, Silva-Vergara ML. Disseminated histoplasmosis in acquired immunodeficiency syndrome patients in Uberaba, MG, Brazil. Mycoses. 2008;51:136–40. DOIPubMedGoogle Scholar
- Huber F, Nacher M, Aznar C, Pierre-Demar M, El Guedj M, Vaz T, et al. AIDS-related Histoplasma capsulatum var. capsulatum infection: 25 years experience of French Guiana. AIDS. 2008;22:1047–53. DOIPubMedGoogle Scholar
- Adenis A, Nacher M, Hanf M, Basurko C, Dufour J, Huber F, et al. Tuberculosis and histoplasmosis among human immunodeficiency virus-infected patients: a comparative study. Am J Trop Med Hyg. 2014;90:216–23. DOIPubMedGoogle Scholar
- Samayoa B, Aguirre L, Bonilla O, Medina N, Lau-Bonilla D, Mercado D, et al.; “Fungired”. The Diagnostic Laboratory Hub: a new health care system reveals the incidence and mortality of tuberculosis, histoplasmosis, and cryptococcosis of PWH in Guatemala. Open Forum Infect Dis. 2019;7:
ofz534 . DOIPubMedGoogle Scholar - Morote S, Nacher M, Blaizot R, Ntab B, Blanchet D, Drak Alsibai K, et al. Temporal trends of cutaneo-mucous histoplasmosis in persons living with HIV in French Guiana: Early diagnosis defuses South American strain dermotropism. PLoS Negl Trop Dis. 2020;14:
e0008663 . DOIPubMedGoogle Scholar
Figure
Tables
Cite This ArticleOriginal Publication Date: October 12, 2022
Table of Contents – Volume 28, Number 11—November 2022
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Rita O. Oladele, Lagos University Teaching Hospital, Ishaga Rd, Idi-Araba 102215, Lagos, Nigeria
Top