Volume 28, Number 12—December 2022
Synopsis
Iceland as Stepping Stone for Spread of Highly Pathogenic Avian Influenza Virus between Europe and North America
Cite This Article
Citation for Media
Abstract
Highly pathogenic avian influenza viruses (HPAIVs) of hemagglutinin type H5 and clade 2.3.4.4b have widely spread within the northern hemisphere since 2020 and threaten wild bird populations, as well as poultry production. We present phylogeographic evidence that Iceland has been used as a stepping stone for HPAIV translocation from northern Europe to North America by infected but mobile wild birds. At least 2 independent incursions of HPAIV H5N1 clade 2.3.4.4b assigned to 2 hemagglutinin clusters, B1 and B2, are documented for summer‒autumn 2021 and spring 2022. Spread of HPAIV H5N1 to and among colony-breeding pelagic avian species in Iceland is ongoing. Potentially devastating effects (i.e., local losses >25%) on these species caused by extended HPAIV circulation in space and time are being observed at several affected breeding sites throughout the North Atlantic.
Potentially zoonotic highly pathogenic avian influenza (HPAI) viruses (HPAIVs) of subtype hemagglutinin (HA) 5 (H5) emerged from a domestic geese flock in southern China in the mid-1990s. Since then, descendants of this so-called goose/Guangdong (gs/GD) lineage have continued to circulate, evolved into various clades, and formed a plethora of subgenotypes and genotypes that threaten poultry production worldwide (1,2). Because of repeated incursions from poultry into migratory aquatic wild bird populations in Asia, these viruses have spread, since 2005, in several waves westward and southward across Eurasia, into Africa and eastward, through the Bering strait, into North America. Infected but mobile migratory birds aided in linking geographically widely separated areas along overlapping flyways; palearctic breeding areas were serving as an additional link between Eurasia and America during 2014 (3,4).
Because Europe was facing the most severe HPAIV epizootics in the influenza winter seasons of 2020–21 and 2021–22 in terms of case numbers and genetic diversity of characterized viruses (5,6), concerns about spread to North America, this time by westward virus spread, were renewed. By December 2021, HPAI H5N1 detection in wild birds in Canada was reported, followed by numerous additional wild bird cases and incursions into poultry holdings along the eastern coastline of the United States (7,8). Phylogenetic analyses of the viruses in North America confirmed a close relationship to HPAIV H5N1 genotypes from Europe (7–9). Although the outcomes of the transatlantic HPAIV transfer are evident, the steps taken by the virus to cross the Atlantic are not. We present data supporting HPAIV transfer from Europe to North America by bird migration through Iceland.
Although low pathogenicity avian influenza virus (AIV) strains have been detected in sea birds around Iceland (10,11), outbreaks of HPAIV were not reported from Iceland until spring 2022. However, retrospective screening of wild bird samples from Iceland showed that an HPAI case was in a juvenile white-tailed sea eagle (Haliaeetus albicilla) found dead in the southern Westfjords, Iceland, during October 2021 (12). This bird had been equipped with a satellite transmitter (global positioning system/global system for mobile communications) as a nestling on July 24, 2021. After fledging on August 11, 2021, the eagle stayed in the nesting area of its parents and moved within a range of 1.6 km2 (95% minimum complex polygon) for ≈2 months. The juvenile eagle died at the shore of the region in Iceland on October 8, 2021, and was kept frozen until necropsy in the spring of 2022.
Postmortem examination showed a female weighing 5,540 g that had extensive subcutaneous and body cavity fat tissue indicating a good nutritional condition. Gross pathologic alterations (fibrinous pericarditis, swollen hyperemic liver, spleen, and kidneys) were indicative of a severe infectious disease, which led to an acute death of the young eagle. We analyzed organ samples for AIV by using quantitative reverse transcription PCR as described (13). HPAIV of subtype H5N1 was found at high viral loads in all tissue samples examined, including the brain (cycle threshold 16.2).
Despite an appeal from the veterinary authorities in Iceland to the general public to report finding of sick or dead wild birds, only 17 birds came to be sampled and AIV was tested in the first 9 months of 2021, and all samples were AIV negative. In the beginning of 2022, the veterinary authorities in Iceland enhanced passive surveillance through reports from the public of sick or dead wild birds. In mid-April, a common raven (Corvus corax) and a pink-footed goose (Anser brachyrhynchus) tested HPAIV H5N1 positive. In addition, in the same period, a northern gannet (Morus bassanus) tested positive for H5N1, but HPAI could not be confirmed. The raven was found on a farm in southern Iceland where 6 days later a backyard chicken flock on the same farm showed abruptly increased mortality rate, and chicken carcasses tested HPAIV H5N1 positive. Consequently, public awareness and reporting of dead wild birds increased markedly after a press release on these first findings.
From April 2022 onward, including the already identified wild birds, HPAIV H5N1 was detected in 21 wild birds from 10 species: northern gannets (n = 7), European herring gull (Larus argentatus) (n = 2), great black-backed gull (Larus marinus) (n = 2), great skua (Stercorarius skua) (n = 2), greylag goose (Anser anser) (n = 2), pink-footed goose (n = 2), barnacle goose (Branta leucopsis) (n = 1), black-headed gull (Chroicocephalus ridibundus) (n = 1), common raven (n = 1), and lesser black-backed gull (Larus fuscus) (n = 1). Because in 1 sample from a northern gannet, neuraminidase 1 could not be confirmed, the bird was reported as positive for HPAIV H5Nx (last updated on June 21, 2022).
Phylogeographic Identification of >2 Virus Introduction Events
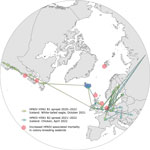
We performed direct MinION (Oxford Nanopore Technologies, https://nanoporetech.com) full-genome sequencing as described (5) for 3 samples from Iceland (2022AI02104: white-tailed eagle, brain tissues; 2022AI02564 and 2022AI02565: backyard chickens, oropharyngeal swab specimens) that were immediately available for analysis and showed high viral loads. Presence of HPAIV H5N1 of clade 2.3.4.4b was confirmed. Phylogenetic and phylogeographic analyses of the genomes (Appendix) and associated data (14) showed close relationships to HPAIV H5N1 viruses from Europe and North America, grouping in 2 different HA clusters (B1 and B2) recently defined in clade 2.3.4.4b viruses from Europe (Figure 1) (6,15–18).
Those findings point to >2 independent incursions into Iceland. The sequence from Iceland isolated during 2021 clusters in the B1 HA cluster between sequences from countries in northern Europe (the Netherlands, Ireland) and sequences from Canada and eastern coastal states of the United States (Figure 2). Analyses of concatenated genome sequences showed no evidence of reassortment with other AIV strains currently or recently circulating in Europe. Time-scaled phylogenetic analyses and inferred phylogeography (Figures 1, 2) demonstrate the circulation of similar viruses of the B1 HA cluster in northern Europe from the winter of 2020 to spring and summer of 2021 (6), and point toward viral spread from locations on the British Isles to Iceland and from there onwards to Canada and eastern coast of the United States.
White-tailed sea eagles are known to be a resident bird species in Iceland, and introduction of virus with this species is highly unlikely. Instead, the white-tailed sea eagle infection is likely caused by feeding of the eagle on infected, therefore weakened, prey or scavenging on carcasses, as described for raptor species (19). Some of the contemplable prey species of the taxonomic orders of Anseriformes or Charadriiformes, including geese, gulls and waders, are known to migrate from the British Isles and the North Sea region and are confirmed to have been infected in spring and early summer of 2021 in their overwintering areas (20–22). Iceland is situated along overlapping flyways that connect the Eastern and Western Hemispheres, and it has been suggested that Iceland connects virus movements between mainland Europe and North America (7–11,23).
In addition, HPAIV H5N1 genomes from 2 chickens dying in a backyard farm on Iceland during April 2022 were sequenced and could be traced back to a second, independent incursion featuring viruses of HA cluster B2. Inferred phylogeographic analysis showed that viruses collected in northern Asia were a possible source of this second introduction into central Europe and further spread throughout the continent (6). The Iceland chicken sequences cluster between viruses of HA cluster B2 collected from the British Islands and Ireland during the winter of 2021/2022 (Figures 1, 2). Viruses of this HA cluster (B2) have not been detected in North America to date.
Our data provide evidence for 2 translocation events of HPAIV H5N1 clade 2.3.4.4b viruses from central Europe through the British Isles into Iceland observed during October 2021 with a most recent ancestor in summer 2021 (most recent common ancestor 2021.5). Onward transmission to Newfoundland and possibly additional regions in the North Atlantic raises several concerns.
Large breeding colonies of pelagic bird species, such as puffins, northern gannets, and kittiwakes are located along the coasts of the North Atlantic. Confirmed HPAIV H5N1 infection in 9/12 gannet carcasses and daily public reporting of sick and dead gannets in the Reykjanes Peninsula, Iceland, since beginning of April 2022 underline that these colonies are now in danger of HPAIV H5 outbreaks of larger scale, which might affect the continuity of these local populations. Concerns extend to local populations of species with narrowly circumscribed breeding/resting ranges in the North Atlantic region such as great skua, long-tailed skua, red knots, pink-footed geese, and barnacle geese, as well as birds of prey exposed during opportunistic scavenging (e.g., white-tailed sea eagles and great skuas) and active hunting of weakened, infected prey (e.g., gyrfalcons [Falco rusticolus]). Therefore, enhanced passive surveillance should focus on such spots and scavenging and colony-breeding species.
The massively extended circulation in space and time of recent HPAIV H5N1 clade 2.3.4.4b viruses in migratory wild birds in the North Atlantic will further threaten endangered species. Grossly increased mortality rates for colonies of northern gannets and several tern species are being observed at several breeding sites throughout the North Atlantic (Figure 2). The most recent incursion of these viruses into wider palearctic areas of the Atlantic will inevitably lead to viral contamination of northern breeding habitats where ambient conditions prevail that are considered favorable for a prolonged retainment of viral infectivity outside avian hosts (23,24).
Increased alertness should now also extend to the Southern Hemisphere. In the 2 reported incursion events of gs/GD HPAI viruses into North America by migrating wild birds, during 2014 and 2021/2022, virus spread along the Pacific (2014) and the Atlantic coastline (2021) from north to south and further inland affecting wild birds and poultry in Canada, as well as in most of the United States (7–9). However, for unknown reasons, spread seems to be interrupted between North America and South America because no incursions had been reported during 2014/2015 or since 2021 from the Caribbean region and South America.
Similar observations have been made along the east side of the Pacific Ocean. Despite endemic presence of gs/GD HPAIV in several regions of Southeast Asia, and frequent incursions into migratory wild bird populations, cases have so far not been reported from Australia/Oceania (4). It is only at the most southern tip of Africa that gs/GD-like HPAIVs have reached and stayed within the Southern Hemisphere. However, this bridgehead of the virus might put geographically sequestrated subantarctic species, such as penguins and albatrosses, or the highly endangered avifauna of New Zealand at increased risk for exposure.
In conclusion, as shown by the rapid and devastating spread of HPAIV H5N1 through poultry holdings in North America after primary incursions from infected wild birds (10), the avian‒human interface has expanded again. Infections in 1 human (25) and in several terrestrial scavenging carnivores, such as foxes, skunks, and raccoons (12), illustrate the increased risk for spillover transmissions.
Dr. Guenther is a veterinarian and a doctoral candidate at the Friedrich-Loeffler-Institute, Greifswald–Insel Riems, Germany. Her primary research interests are avian viruses and other pathogens with potential influence on avian species conservation and public health.
Acknowledgments
We thank Aline Maksimov, Diana Parlow, Mareen Lange, and Cornelia Illing for providing excellent technical assistance, and all submitting laboratories for providing sequence data in the GISAID database (https://gisaid.org).
All sequence data including raw data are available in the INSDC (https://www.insdc.org) and GISAID database via ENA (https://www.ebi.ac.uk) project accession no. PRJEB53596 and EPI (https://www.ncbi.nlm.nih.gov) isolate accession nos. 13245602, 13246267, and 13246657. Details of methods are described separately. All associated information is available on Zenodo (https://zenodo.org).
This study was supported by the European Union Horizon 2020 (program grant VEO no. 874735) and by the German Federal Ministry of Education and Research (project PREPMEDVET, grant no. 13N15449).
A.P. and T.H. conceptualized the study; A.G., O.K., V.S., A.P., J.K., B.B., and T.H. provided and validated methods; A.G., O.K., V.S., A.P., J.K., B.B., and T.H. performed investigations; A.G., O.K., V.S., A.P., G.T.H., B.B., and T.H. formerly analyzed data; M.B., B.B., and O.K. provided resources; A.G., O.K., V.S., A.P., J.K., B.B., and T.H. provided data curation; A.G., T.H., O.K., and B.B. wrote and prepared the original draft; A.G., O.K., V.S., A.P., J.K., G.T.H., K.H.S., H.S., S.R.J., M.B., B.B., and T.H. wrote, reviewed, and edited the manuscript; A.P. and A.G. visualized the study; T.H., M.B., and B.B. supervised the study; M.B., T.H., and A.P. administered the study; and A.P. and M.B. provided funding. All authors have read and agreed to the published version of the manuscript.
References
- Lee DH, Criado MF, Swayne DE. Pathobiological origins and evolutionary history of highly pathogenic avian influenza viruses. Cold Spring Harb Perspect Med. 2021;11:
a038679 . DOIPubMedGoogle Scholar - Dhingra MS, Artois J, Dellicour S, Lemey P, Dauphin G, Von Dobschuetz S, et al. Geographical and historical patterns in the emergences of novel highly pathogenic avian influenza (HPAI) H5 and H7 viruses in poultry. Front Vet Sci. 2018;5:84. DOIPubMedGoogle Scholar
- Lee DH, Torchetti MK, Winker K, Ip HS, Song CS, Swayne DE. Intercontinental spread of Asian-origin H5N8 to North America through Beringia by migratory birds. J Virol. 2015;89:6521–4. DOIPubMedGoogle Scholar
- Global Consortium for H5N8 and Related Influenza Viruses. Role for migratory wild birds in the global spread of avian influenza H5N8. Science. 2016;354:213–7. DOIGoogle Scholar
- King J, Harder T, Globig A, Stacker L, Günther A, Grund C, et al. Highly pathogenic avian influenza virus incursions of subtype H5N8, H5N5, H5N1, H5N4, and H5N3 in Germany during 2020‒21. Virus Evol. 2022;8:veac025. DOIGoogle Scholar
- Pohlmann A, King J, Fusaro A, Zecchin B, Banyard AC, Brown IH, et al. Has epizootic become enzootic? Evidence for a fundamental change in the infection dynamics of highly pathogenic avian influenza in Europe, 2021. MBio. 2022;13:
e0060922 . DOIPubMedGoogle Scholar - Caliendo V, Lewis NS, Pohlmann A, Baillie SR, Banyard AC, Beer M, et al. Transatlantic spread of highly pathogenic avian influenza H5N1 by wild birds from Europe to North America in 2021. Sci Rep. 2022;12:11729. DOIPubMedGoogle Scholar
- US Department of Agriculture, Animal and Plant Health Inspection Service. Detections of highly pathogenic avian influenza in wild birds. 2022 [cited 2022 Aug 4]. https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/avian/avian-influenza/hpai-2022/2022-hpai-wild-birds
- Bevins SN, Shriner SA, Cumbee JC Jr, Dilione KE, Douglass KE, Ellis JW, et al. Intercontinental movement of highly pathogenic avian influenza A(H5N1) clade 2.3.4.4 virus to the United States, 2021. Emerg Infect Dis. 2022;28:1006–11. DOIPubMedGoogle Scholar
- Gass JD Jr, Kellogg HK, Hill NJ, Puryear WB, Nutter FB, Runstadler JA. Epidemiology and ecology of influenza A viruses among wildlife in the Arctic. Viruses. 2022;14:1531. DOIPubMedGoogle Scholar
- Dusek RJ, Hallgrimsson GT, Ip HS, Jónsson JE, Sreevatsan S, Nashold SW, et al. North Atlantic migratory bird flyways provide routes for intercontinental movement of avian influenza viruses. PLoS One. 2014;9:
e92075 . DOIPubMedGoogle Scholar - Adlhoch C, Fusaro A, Gonzales JL, Kuiken T, Marangon S, Niqueux É, et al.; European Food Safety Authority; European Centre for Disease Prevention and Control; European Union Reference Laboratory for Avian Influenza. Avian influenza overview March - June 2022. EFSA J. 2022;20:
e07415 .PubMedGoogle Scholar - Hassan KE, Ahrens AK, Ali A, El-Kady MF, Hafez HM, Mettenleiter TC, et al. Improved subtyping of avian influenza viruses using an RT-qPCR-based low density array: ‘Riems Influenza a Typing Array’, Version 2 (RITA-2). Viruses. 2022;14:415. DOIPubMedGoogle Scholar
- Pohlmann A. Iceland as stepping stone for intercontinental spread of highly pathogenic avian influenza H5N1 virus between Europe and North America: data set on phylogeographic analysis, 2022 [cited 2022 Aug 17].
- The Guardian. The scale is hard to grasp: avian flu wreaks devastation on sea birds [cited 2022 Oct 18]. https//www.the guardian.com/environment/2022/jul/20/avian-flu-h5n1-wreaks-devastation-seabirds-aoe
- Norwegian Veterinary Institute. Avian influenza detected on Svalbard [cited 2022 Oct 18]. https://www.vetinst.no/en/news/avian-influenza-detected-on-svalbard
- Columbia Broadcasting System. Avian flu responsible for thousands of dead birds in Newfoundland, suggest preliminary tests [cited 2022 Oct 18]. https://www.cbc.ca/news/canada/newfoundland-labrador/avian-flu-newfoundland-1.6529433
- Audubon. Avian flu threatens seabird nesting colonies on both sides of the Atlantic [cited 2022 Oct 18]. https://www.audubon.org/news/avian-flu-threatens-seabid-nesting-colonies-both-sides-atlantic
- Krone O, Globig A, Ulrich R, Harder T, Schinköthe J, Herrmann C, et al. White-tailed sea eagle (Haliaeetus albicilla) die-off due to infection with highly pathogenic avian influenza virus, subtype H5N8, in Germany. Viruses. 2018;10:478. DOIPubMedGoogle Scholar
- Lean FZX, Vitores AG, Reid SM, Banyard AC, Brown IH, Núñez A, et al. Gross pathology of high pathogenicity avian influenza virus H5N1 2021-2022 epizootic in naturally infected birds in the United Kingdom. One Health. 2022;14:
100392 . DOIPubMedGoogle Scholar - Banyard AC, Lean FZX, Robinson C, Howie F, Tyler G, Nisbet C, et al. Detection of highly pathogenic avian influenza virus H5N1 clade 2.3.4.4b in Great Skuas: a species of conservation concern in Great Britain. Viruses. 2022;14:212. DOIPubMedGoogle Scholar
- Hill NJ, Bishop MA, Trovão NS, Ineson KM, Schaefer AL, Puryear WB, et al. Ecological divergence of wild birds drives avian influenza spillover and global spread. PLoS Pathog. 2022;18:
e1010062 . DOIPubMedGoogle Scholar - Stallknecht DE, Goekjian VH, Wilcox BR, Poulson RL, Brown JD. Avian influenza virus in aquatic habitats: what do we need to learn? Avian Dis. 2010;54(Suppl):461–5. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. U.S. case of human avian influenza A(H5) virus reported. 2022 [cited 2022 Jul 7]. https://www.cdc.gov/media/releases/2022/s0428-avian-flu.html
Figures
Cite This ArticleOriginal Publication Date: October 19, 2022
1These authors contributed equally to this article.
Table of Contents – Volume 28, Number 12—December 2022
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Timm Harder, Institute of Diagnostic Virology, Friedrich-Loeffler-Institut, Südufer 10, 17493 Greifswald–Insel Riems, Germany; e-mail:timm.harder@fli.de
Top