Volume 28, Number 5—May 2022
Research
Effectiveness of BNT162b2 Vaccine Booster against SARS-CoV-2 Infection and Breakthrough Complications, Israel
Figure 3
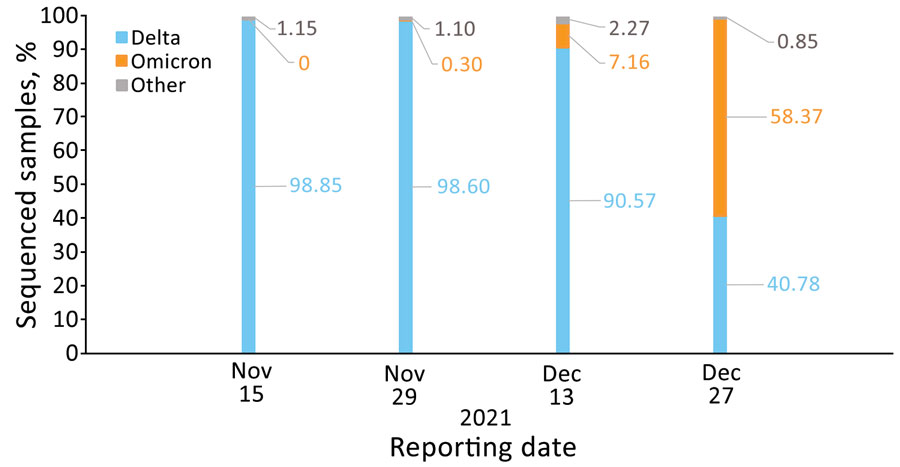
Figure 3. Percentage of sequenced severe acute respiratory syndrome coronavirus 2 samples by variant and reporting date, Israel, November 15, November 29, December 13, and December 27, 2021. Based on (9). Numbers within the figure represent percentages of sequenced samples.
References
- Glatman-Freedman A, Hershkovitz Y, Kaufman Z, Dichtiar R, Keinan-Boker L, Bromberg M. Effectiveness of BNT162b2 vaccine in adolescents during outbreak of SARS-CoV-2 Delta variant infection, Israel, 2021. Emerg Infect Dis. 2021;27:2919–22. DOIPubMedGoogle Scholar
- Goldberg Y, Mandel M, Bar-On YM, Bodenheimer O, Freedman L, Haas EJ, et al. Waning immunity after the BNT162b2 vaccine in Israel. N Engl J Med. 2021;385:
e85 . DOIPubMedGoogle Scholar - Levin EG, Lustig Y, Cohen C, Fluss R, Indenbaum V, Amit S, et al. Waning immune humoral response to BNT162b2 COVID-19 vaccine over 6 months. N Engl J Med. 2021;385:
e84 . DOIPubMedGoogle Scholar - Israel Ministry of Health. The vaccination advisory committee presented data and recommended the administration of a third dose to older adults. 2021 Jul 30 [cited 2021 Nov 21]. https://www.gov.il/en/departments/news/29072021-04
- Israel Ministry of Health. Discussions on administering the third vaccine to additional populations. 2021 Aug 20 [cited 2021 Nov 21]. https://www.gov.il/en/departments/news/19082021-04
- Glatman-Freedman A, Bromberg M, Dichtiar R, Hershkovitz Y, Keinan-Boker L. The BNT162b2 vaccine effectiveness against new COVID-19 cases and complications of breakthrough cases: A nation-wide retrospective longitudinal multiple cohort analysis using individualised data. EBioMedicine. 2021;72:
103574 . DOIPubMedGoogle Scholar - National Institutes of Health. Coronavirus disease 2019 (COVID-19) treatment guidelines: clinical spectrum of SARS-CoV-2 infection [cited 2021 Feb 25]. https://www.covid19treatmentguidelines.nih.gov
- Central Bureau of Statistics. Statistical abstract of Israel 2021—no. 72 [cited 2021 Nov 22]. https://www.cbs.gov.il/en/publications/Pages/2021/Population-Statistical-Abstract-of-Israel-2021-No.72.aspx
- Our World in Data. SARS-CoV-2 variants in in analyzed sequences, Israel. 2022 [cited 2022 March 6]. https://ourworldindata.org/grapher/covid-variants-area?country=~ISR
- Bayart JL, Douxfils J, Gillot C, David C, Mullier F, Elsen M, et al. Waning of IgG, total and neutralizing antibodies 6 months post-vaccination with BNT162b2 in healthcare workers. Vaccines (Basel). 2021;9:1092. DOIPubMedGoogle Scholar
- UK Health Security Agency. SARS-CoV-2 variants of concern and variants under investigation in England. Technical briefing 33. 2021 [cited 2022 Jan 9]. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1043807/technical-briefing-33.pdf
- Collie S, Champion J, Moultrie H, Bekker LG, Gray G. Effectiveness of BNT162b2 vaccine against Omicron variant in South Africa. N Engl J Med. 2022;386:494–6. DOIPubMedGoogle Scholar
- Bar-On YM, Goldberg Y, Mandel M, Bodenheimer O, Freedman L, Kalkstein N, et al. Protection of BNT162b2 vaccine booster against COVID-19 in Israel. N Engl J Med. 2021;385:1393–400. DOIPubMedGoogle Scholar
- Bar-On YM, Goldberg Y, Mandel M, Bodenheimer O, Freedman L, Alroy-Preis S, et al. Protection against COVID-19 by BNT162b2 booster across age groups. N Engl J Med. 2021;385:2421–30. DOIPubMedGoogle Scholar
- Barda N, Dagan N, Cohen C, Hernán MA, Lipsitch M, Kohane IS, et al. Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study. Lancet. 2021;398:2093–100. DOIPubMedGoogle Scholar
- Tartof SY, Slezak JM, Puzniak L, Hong V, Frankland TB, Ackerson BK, et al. Effectiveness of a third dose of BNT162b2 mRNA COVID-19 vaccine in a large US health system: a retrospective cohort study. Lancet Reg Health Am. 2022 Feb 14 [Epub ahead of print].
- Andrews N, Stowe J, Kirsebom F, Toffa S, Sachdeva R, Gower C, et al. Effectiveness of COVID-19 booster vaccines against covid-19 related symptoms, hospitalisation and death in England. Nat Med. 2022; [Epub ahead of print]. DOIGoogle Scholar
Page created: March 18, 2022
Page updated: April 19, 2022
Page reviewed: April 19, 2022
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.