Volume 28, Number 8—August 2022
Research Letter
Early SARS-CoV-2 Reinfections within 60 Days and Implications for Retesting Policies
Figure
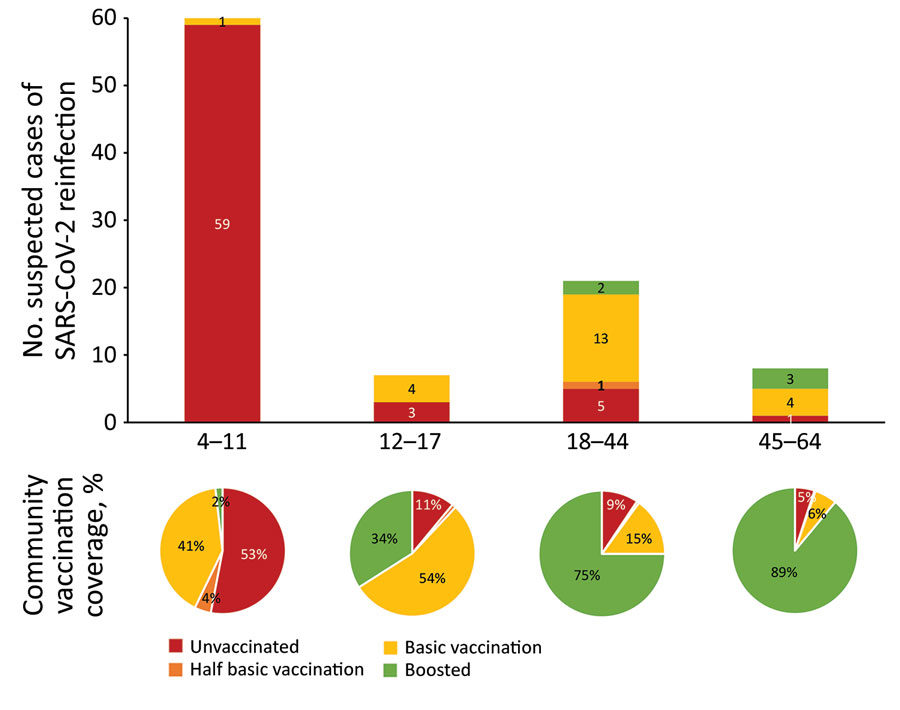
Figure. Number of patients with presumed SARS-CoV-2 reinfection including vaccination status compared with age-corresponding vaccination coverage in the community, Flanders, Belgium. Consecutive infections were detected during December 1, 2021–February 7, 2022 (reinfection with Omicron BA.1 shortly after Delta infection, n = 91 patients) and during January 1–March 10, 2022 (reinfection with Omicron BA.2 shortly after Omicron BA.1 infection, n = 5 patients). Half basic vaccination indicates 1 vaccine of ChAdOx1 nCoV-19 (AstraZeneca, https://www.astrazeneca.com), BNT162b2 (Pfizer-BioNTech, https://www.pfizer.com), or mRNA-1273 (Moderna, https://www.modernatx.com); basic vaccination indicates 2 vaccines of ChAdOx1, BNT162b2, or mRNA-1273 or 1 vaccine of Ad26.COV2.S (Johnson & Johnson/Janssen, https://www.janssen.com); boosted indicates basic vaccination followed by 1 vaccine of BNT162b2 or mRNA-1273.