Volume 29, Number 5—May 2023
Research Letter
Mpox among Public Festival Attendees, Chicago, Illinois, USA, July–August 2022
Cite This Article
Citation for Media
Abstract
We investigated an mpox outbreak after a 2022 LGBTQ event in Chicago, Illinois, USA. Among case-patients, 38% had received 1 dose of mpox vaccine, none 2 doses; most reported sexual activity during the probable exposure period. Among other preventive measures, persons at risk should complete mpox vaccination 14 days before an event.
Monkeypox virus transmission may be associated with large events where at-risk or potentially infected persons engage in sexual activity at or outside the event (1). The Chicago Department of Public Health (CDPH) investigated an mpox outbreak associated with Market Days (MD), an annual LGBTQ outdoor festival held in Chicago, Illinois, USA, August 6–7, 2022, attended by ≈100,000 persons from across the country. We describe persons with mpox who attended the festival.
CDPH interviewed mpox case-patients (2) to ascertain MD attendance and conducted a supplemental survey about exposures, sexual behavior, and preventive measures. Event-attending case-patients were defined as persons with positive mpox or orthopoxvirus test results who attended MD on August 6–7 and had symptoms within 21 days before or after attendance (July 16–August 28). Illinois surveillance systems verified laboratory and JYNNEOS (https://www.jynneos.com) vaccination data. CDPH also solicited cases from other jurisdictions in a national call for cases related to the event. Event-associated case-patients were categorized by vaccination status at the time of MD. This investigation was reviewed by the Centers for Disease Control and Prevention and conducted in accordance with its policies and applicable federal law (45 C.F.R. part 46.102(l) (2), 21 C.F.R. part 56; 42 U.S.C. §241(d); 5 U.S.C. §552a; 44 U.S.C. §3501 et seq).
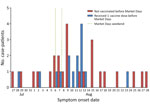
We identified 40 event-associated case-patients from Chicago and other jurisdictions who attended MD; symptom onset occurred before MD for 7 and during or after for 33 (Figure). Case-patients with symptom onset before MD had a median of 1 (interquartile range [IQR] 1–2) sex partner during MD, and case-patients with symptom onset after MD had a median of 5 (IQR 2–5) sex partners during MD (Appendix). Many case-patients were non-Hispanic/Latino White (45%), most were male at birth (98%), and most reported having sex with men (70%) (Table). Two case-patients, 1 living with HIV, were hospitalized for mpox.
Fifteen (38%) case-patients had received 1 dose of JYNNEOS vaccine before MD; none received 2 doses or were due for their second dose by MD (within 28 days of first dose). Among 1-dose recipients, 8 (53%) were vaccinated <14 days before symptom onset. Among all 1-dose recipients vaccinated before symptom onset, median time from vaccination to symptom onset was 13 (IQR 9–29) days. Fewer non-Hispanic/Latino Black case-patients were vaccinated before MD (2/15, 13%) than non-Hispanic/Latino White (8/15, 53%) or Hispanic/Latino (5/15, 33%) case-patients.
Most (32/40, 80%) case-patients reported having had sexual activity during the probable exposure period (21 days before symptom onset). Case-patient reports included sexual activity during MD (August 6–7) at any location, meeting a partner at MD, and sexual activity at the event. Only 14 (35%) case-patients reported sexual activity during MD at any location, including 3 with symptom onset before those dates. Nine (23%) persons engaged in sex with >1 partner during MD, 9 (23%) met a sex partner at MD, and 4 (10%) case-patients reported sexual activity at MD. Case-patients reporting sexual activity during MD had a median of 3.5 (IQR 1.3–5.0) partners; more had anonymous sex partners (7/14, 50%) than casual or main partners (4/14, 29%). More case-patients who received 1 vaccine dose before MD had sex with an anonymous partner during MD (4/15, 27%) than did unvaccinated case-patients (3/25, 12%).
Among all event-associated case-patients, 28 (70%) adopted nonvaccine prevention measures during MD; 9 (23%) abstained from sex, and 8 (20%) avoided skin-to-skin contact. More case-patients unvaccinated before MD abstained from sex during MD (7/25, 28%) than did 1-dose recipients (2/15, 13%). Seven (78%) of 9 case-patients who abstained from sex during MD reported having sex during the probable exposure period. Among case-patients reporting sexual activity during MD, 2 (2/14, 14%) reduced their number of sex partners and 4 (4/14, 29%) adopted other measures (e.g., isolating after MD).
Although MD might have presented more opportunities for sex partnerships, <50% of case-patients reported sex related to the event. Among case-patients who had sex during MD, most had anonymous sex partners, potentially increasing transmission (3). Most case-patients also reported adopting >1 prevention measure during MD but may not have maintained measures throughout the probable exposure period. For example, among 9 persons who abstained from sex at MD, 7 (78%) reported sexual activity with >1 partner outside the event.
More than one third of case-patients reported 1 vaccine dose before MD. Symptoms developed within 14 days after the first dose for most (8, 53%) who were partially vaccinated before MD. Although equal proportions of vaccinated and unvaccinated persons reported sex during MD, 1-dose recipients reported taking fewer precautions, such as abstaining from sex or avoiding anonymous sex.
Among study limitations, we may have undercounted event-associated case-patients. Out-of-state case-patients may not have reported or been asked about event attendance. Also, without a comparison group, we could not compare preventive measures by case-patient status. Information about prevention measures and sexual behavior were self-reported and subject to social desirability bias.
Vaccination uptake before large gatherings may affect behavior and perceived infection risk. Risk messaging should emphasize completing vaccination 14 days before an event and taking other measures to prevent mpox.
Dr. Faherty is an infectious disease epidemiologist and CDC Epidemic Intelligence Service Officer assigned to the Chicago Department of Public Health Bureau of Disease Control in the Communicable Diseases Program. Her research interests include vaccine-preventable disease and sexual and reproductive health.
Acknowledgment
We thank Suzanne Beavers, Palak Panchal, Hannah Matzke, Leah Quinn, Saul Ayala, Sarah Love, Elizabeth Shane, Alexandria Davis, Andrew Weidemiller, Angelica Serra, Anthony Garcia, Ben Cammack, Cecilia Pigozzi, Dailha Acevedo, Damian Plaza, Daniel Liguori, Hallie Hutchison, Janetta Prokopowicz, Jazmine Wright, Jose Medina, Lily Fahrenwald, Maria Ferreira-Alharb, Marsha Flowers, Maureen Love, Mawiyah Coates, Salma Ali, Seliciano Douthard, Susan Woods, Tiffany Evans, and Vilma Alicea for their work on this investigation.
References
- Iñigo Martínez J, Gil Montalbán E, Jiménez Bueno S, Martín Martínez F, Nieto Juliá A, Sánchez Díaz J, et al. Monkeypox outbreak predominantly affecting men who have sex with men, Madrid, Spain, 26 April to 16 June 2022. Euro Surveill. 2022;27:
2200471 . DOIPubMedGoogle Scholar - Centers for Disease Control and Prevention. Case definitions for use in the 2022 monkeypox response [cited 2022 Oct 19]. https://www.cdc.gov/poxvirus/monkeypox/clinicians/case-definition.html
- van Kesteren NM, Hospers HJ, Kok G. Sexual risk behavior among HIV-positive men who have sex with men: a literature review. Patient Educ Couns. 2007;65:5–20. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleOriginal Publication Date: March 27, 2023
Table of Contents – Volume 29, Number 5—May 2023
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Emily A.G. Faherty, Chicago Department of Public Health, 1340 S Damen Ave, Chicago, IL 60608, USA;tqy5@cdc.gov
Top