Volume 31, Number 7—July 2025
Dispatch
Human Infections by Novel Zoonotic Species Corynebacterium silvaticum, Germany
Cite This Article
Citation for Media
Abstract
We report 2 human Corynebacterium silvaticum infections in Germany with axillary lymphadenitis and abscess formation; in 1 case the infection likely originated from a slaughtered wild boar. This recently described member of the diphtheria toxin gene–bearing C. diphtheriae species complex might be a new zoonotic pathogen.
Diphtheria is caused by toxigenic Corynebacterium spp. producing the major pathogenicity factor diphtheria toxin (DT). DT is encoded on the tox gene and causes local respiratory or cutaneous symptoms, as well as systemic neurologic and cardiologic symptoms. The 3 potentially DT-producing species are the mainly human pathogen C. diphtheriae and the 2 zoonotic pathogens C. ulcerans and C. pseudotuberculosis. In recent years, the C. diphtheriae species complex (CdSC) was expanded, mostly on the basis of genomic data, in some instances supported by biochemical properties (1). Two closely related species were separated from C. diphtheriae (i.e., C. belfantii [2] and C. rouxii [3]), and 2 were separated from C. ulcerans (i.e., C. silvaticum [4] and C. ramonii [5], previously known as lineage 2 of C. ulcerans).
C. silvaticum (formerly C. ulcerans wildlife cluster), was recently described as a novel zoonotic species causing caseous lymphadenitis in 33 wild boars and a roe deer in Germany (4,6). Subsequently, C. silvaticum was identified in a wild boar from Germany (7) and in a domestic pig from Portugal (8). So far, all animal isolates from Germany were nontoxigenic tox-bearing (NTTB) (4,8,9), whereas isolates from Portugal and Austria were found to be either NTTB or tox-positive, resulting in 2 recently postulated clades depending on molecular characteristics of the tox gene (10). Although C. silvaticum is considered a zoonotic pathogen with the potential to infect humans, no human infections had been described. In this study, we report 2 cases of human C. silvaticum infection.
In case 1, a 37-year-old male butcher sought care for an indolent tumor in the lateral thorax and the right axilla that had been present for 3 weeks. A bacterial swab specimen taken during surgical extirpation grew Corynebacterium sp. after 48 hours in pure culture; the pathogen was C. ulcerans by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (Bruker, https://www.bruker.com). The strain was sent to the German Consiliary Laboratory for Diphtheria (Oberschleißheim, Germany) for further analysis. The strain (KL1848) showed an atypical colony morphology (small waxy white, discrete β-hemolytic colonies after 48 hours’ incubation) and biochemical reactions: fermentation of glucose, ribose, and maltose (like C. ulcerans), but no use of D-xylose, mannitol, lactose, sucrose, or glycogen (like C. pseudotuberculosis). It was sensitive to clindamycin, according to the European Committee on Antimicrobial Susceptibility Testing guidelines for C. diphtheriae and C. ulcerans (https://www.eucast.org/clinical_breakpoints). The isolate was identified as a NTTB C. silvaticum strain using previously described methods (11) (i.e., tox-PCR, an optimized Elek test, a lateral flow immunoassay and whole-genome sequencing [WGS] analyses) (Appendix 1). The patient was successfully treated with surgery and cefuroxime for 14 days. The patient reported to have recently field-dressed a hunted wild boar that showed suspicious mesenterial lymph nodes. The patient’s immunization status was unknown. No secondary human cases occurred.
In case 2, a 21-year-old man sought care for an indolent axillary tumor that had been present for 4 weeks. Lymphoma was suspected on the basis of computed tomography results. The affected lymph node was removed, revealing a fibrosed soft tissue with florid purulent abscess formation and chronic granulating histiocyte-rich granulomatous inflammation. After a few days of incubation, the tissue sample grew a pure culture of Corynebacterium sp., identified as C. ulcerans by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (Bruker). The strain (KL1281) was sent to the German Consiliary Laboratory for Diphtheria. The atypical colony morphology, biochemical reactions, and antimicrobial susceptibility were identical to those of strain KL1848. We identified the isolate as NTTB C. silvaticum using the same methods as in case 1. The case 2 patient had a dog and lived in a rural area of northern Germany. He reported no contact with wild animals. Vaccination against diphtheria was completed. No secondary human case was observed.
Average nucleotide identity analysis of WGS assemblies compared with public type-strain genomes of the CdSC classified both isolates with highest concordance as C. silvaticum with identity values >99.9%. Identity values for the other species were nearly identical for both strains and clearly below the species threshold of 94%–96% (12); values were 90.6% for C. ramonii FRC0011T, 90.5% for C. ulcerans NCTC7910T, 85.0% for C. pseudotuberculosis ATCC19410T, 74.3% for C. diphtheriae NCTC11397T, 74.1% for C. rouxii FRC190T, and 74.0% for C. belfantii FRC0043T.
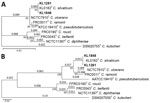
We constructed gene phylogenies of 16S and rpoB from the WGS data and compared them with the respective genes of the CdSC-type strains and C. kutscheri as an outgroup. The phylogenetic trees (Figure 1) show a clear assignment to C. silvaticum, because both isolates share the branch of the C. silvaticum–type strain with clear separation from the other species. Multilocus sequence typing (MLST) analysis with the C. diphtheriae scheme showed sequence type (ST) 578, exclusive to C. silvaticum and typically also found in isolates from wild boar and roe deer in Germany (Appendix 2). Evaluation of the tox gene and an alignment with tox sequences from Germany, published sequences from Portugal (10) and a C. diphtheriae tox sequence as reference revealed the presence of the insertion of 2 guanine residues after position 44 (Appendix 1 Figure), as found in the other Germany isolates from wild boar and roe deer, enabling assignment to C. silvaticum clade 2 (10).
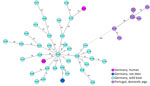
To investigate the clade classification with a more detailed core genome MLST analysis, we constructed an ad hoc core genome MLST scheme on the basis of publicly available C. silvaticum genomes from both clades (Appendix 1). We generated a minimum-spanning tree from 45 genomes, consisting of KL1281, KL1848, an additional 35 isolates from animals from Germany, and the 8 clade 1 genomes from Portugal (10). The tree layout (Figure 2) and the allelic distances (Appendix 2) clearly affiliate the patient cases with the cases from clade 2 from Germany, whereas the genomes from clade 1 from Portugal sit on another branch and show >500 allelic differences to clade 2. Thus, the classification by the tox gene is also supported by a whole-genome context.
C. silvaticum (4) has been suspected to be a possible zoonotic member of the newly expanded CdSC. Originally isolated from forest-associated animals such as wild boars and roe deer, it has also been identified in a domestic pig. In this study, we report 2 cases of human infection linked to close animal contact (wild boar and dog). In case 1, the most probable route of infection was direct contact with infectious tissue and possible microtrauma lesions during a slaughtering or animal field-dressing procedure. In case 2, no close contact to wild animals was reported, although the patient lived in a rural area and owned a dog. However, C. silvaticum has so far been only identified in wild forest-dwelling animals and a pig. All cases in Germany, including the 2 human cases described in this study, were found to be NTTB and summarized in a corresponding NTTB clade with a 2-bp insertion in the tox gene sequence, named clade 2 (10). In contrast, in Portugal and Austria, another monophyletic clade of DT-producing C. silvaticum was reported and named clade 1 (10).
In conclusion, C. silvaticum should be considered a zoonotic pathogen with possible animal-to-human transmission. Therefore, animal sources, especially wild boar and domestic pigs, should be included when tracing sources of potentially toxigenic Corynebacterium spp. of the broader C. diphtheriae and the narrower C. ulcerans complex. C. ulcerans has also been transmitted from pigs to humans (13,14). With respect to the known clindamycin resistance in C. ulcerans (15), antimicrobial susceptibility testing should support antibiotic therapy. Suitable precaution measures should be taken when handling wild boars and pigs to avoid zoonotic risk for C. ulcerans or C. silvaticum infection.
Dr. Berger is a board-certified clinical microbiologist with special interest in public health microbiology, antimicrobial resistance, and bacteriology. She is deputy head of the German National Consiliary Laboratory on Diphtheria, the German part of the EU Public Health Reference Laboratory for Diphtheria and Pertussis, and the World Health Organization Collaborating Centre for Diphtheria.
Acknowledgments
We thank Wolfgang Schmidt, Cengiz-Turgut Dedeoglu, Juliane Breitenberger, Juliana Webersberger, Andrea Seifarth, Helga Kocak, Sabine Lohrer, Jasmin Scholz, Anne Könitzer, and Marion Lindermayer for excellent technical assistance. We thank all diagnostic laboratories submitting samples and strains to the GNCLD.
Ethical approval was not necessary for this study because it was performed as part of routine surveillance activities, outbreak investigation, and diphtheria diagnostics in Germany. Whole-genome sequencing data are available in the National Center for Biotechnology Information Short Read Archive (https://www.ncbi.nlm.nih.gov/sra; Bioproject PRJNA490531). Detailed accession numbers are given in Appendix 2.
The study was partly supported by the Bavarian State Ministry of Health, Care and Prevention, as well as by the German Federal Ministry of Health via the Robert Koch-Institute and its National Reference Laboratories Network (09-47, FKZ 1369-359, and FKZ 415).
A.B., K.B., A.S., C.S., V.G.M. were involved in laboratory work-up; A.D. performed whole-genome sequencing. T.R. and H.J.M. were involved in clinical care. All authors interpreted the results. A.B. was involved in epidemiological work-up. A.B., A.D., and A.S. wrote the paper. All authors reviewed and approved the final version of the manuscript.
References
- Prygiel M, Polak M, Mosiej E, Wdowiak K, Formińska K, Zasada AA. New Corynebacterium species with the potential to produce diphtheria toxin. Pathogens. 2022;11:1264. DOIPubMedGoogle Scholar
- Dazas M, Badell E, Carmi-Leroy A, Criscuolo A, Brisse S. Taxonomic status of Corynebacterium diphtheriae biovar Belfanti and proposal of Corynebacterium belfantii sp. nov. Int J Syst Evol Microbiol. 2018;68:3826–31. DOIPubMedGoogle Scholar
- Badell E, Hennart M, Rodrigues C, Passet V, Dazas M, Panunzi L, et al. Corynebacterium rouxii sp. nov., a novel member of the diphtheriae species complex. Res Microbiol. 2020;171:122–7. DOIPubMedGoogle Scholar
- Dangel A, Berger A, Rau J, Eisenberg T, Kämpfer P, Margos G, et al. Corynebacterium silvaticum sp. nov., a unique group of NTTB corynebacteria in wild boar and roe deer. Int J Syst Evol Microbiol. 2020;70:3614–24. DOIPubMedGoogle Scholar
- Crestani C, Arcari G, Landier A, Passet V, Garnier D, Brémont S, et al. Corynebacterium ramonii sp. nov., a novel toxigenic member of the Corynebacterium diphtheriae species complex. Res Microbiol. 2023;174:
104113 . DOIPubMedGoogle Scholar - Eisenberg T, Kutzer P, Peters M, Sing A, Contzen M, Rau J. Nontoxigenic tox-bearing Corynebacterium ulcerans infection among game animals, Germany. Emerg Infect Dis. 2014;20:448–52. DOIPubMedGoogle Scholar
- Busch A, Möller J, Burkovski A, Hotzel H. Genome sequence of a pathogenic Corynebacterium ulcerans strain isolated from a wild boar with necrotizing lymphadenitis. BMC Res Notes. 2019;12:692. DOIPubMedGoogle Scholar
- Viana MVC, Profeta R, da Silva AL, Hurtado R, Cerqueira JC, Ribeiro BFS, et al. Taxonomic classification of strain PO100/5 shows a broader geographic distribution and genetic markers of the recently described Corynebacterium silvaticum. PLoS One. 2020;15:
e0244210 . DOIPubMedGoogle Scholar - Möller J, Busch A, Berens C, Hotzel H, Burkovski A. Newly isolated animal pathogen Corynebacterium silvaticum is cytotoxic to human epithelial cells. Int J Mol Sci. 2021;22:3549. DOIPubMedGoogle Scholar
- Viana MVC, Galdino JH, Profeta R, Oliveira M, Tavares L, de Castro Soares S, et al. Analysis of Corynebacterium silvaticum genomes from Portugal reveals a single cluster and a clade suggested to produce diphtheria toxin. PeerJ. 2023;11:
e14895 . DOIPubMedGoogle Scholar - Berger A, Zasada AA, Dangel A, Piekarska K, Paradowska-Stankiewicz I, Bengs K, et al. A case of fatal respiratory diphtheria imported from Poland to Germany: possible link to an undetected imported diphtheria cluster in Poland? Infection. 2025; Epub ahead of print. DOIPubMedGoogle Scholar
- Richter M, Rosselló-Móra R. Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci U S A. 2009;106:19126–31. DOIPubMedGoogle Scholar
- Schuhegger R, Schoerner C, Dlugaiczyk J, Lichtenfeld I, Trouillier A, Zeller-Peronnet V, et al. Pigs as source for toxigenic Corynebacterium ulcerans. Emerg Infect Dis. 2009;15:1314–5. DOIPubMedGoogle Scholar
- Berger A, Boschert V, Konrad R, Schmidt-Wieland T, Hörmansdorfer S, Eddicks M, et al. Two cases of cutaneous diphtheria associated with occupational pig contact in Germany. Zoonoses Public Health. 2013;60:539–42. DOIPubMedGoogle Scholar
- Berger A, Badell E, Åhman J, Matuschek E, Zidane N, Kahlmeter G, et al. Corynebacterium diphtheriae and Corynebacterium ulcerans: development of EUCAST methods and generation of data on which to determine breakpoints. J Antimicrob Chemother. 2024;79:968–76. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: June 18, 2025
1These authors contributed equally to this article.
2Current affiliation: University of Cambridge, Cambridge, UK.
Table of Contents – Volume 31, Number 7—July 2025
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Anja Berger, National Consiliary Laboratory for Diphtheria, WHO Collaborating for Diphtheria, Bavarian Health and Food Safety Authority, Veterinärstraße 2, 85764 Oberschleißheim, Germany
Top