Volume 32, Number 4—April 2026
Research Letter
Seroepidemiologic Study of Oropouche Virus, Amazonas State, Brazil, 2015–2016
Cite This Article
Citation for Media
Abstract
We conducted a cross-sectional serosurvey for Oropouche virus (OROV) among residents of Amazonas State, Brazil, during 2015–2016. We detected OROV neutralizing antibodies in 85/814 (10.4%) participants; seroprevalence was higher in Manaus (49/440 [11.1%]) than in Coari (36/374 [9.6%]). Those findings suggest OROV circulation in Amazonas State before 2015.
Oropouche virus (OROV) is a neglected and reemerging vectorborne orthobunyavirus endemic in Central and South America since the 1950s (1,2). OROV is mainly transmitted to humans by the bite of infected midges (Culicoides paraensis), which also spread OROV among sloths and other animals in the sylvatic cycle (3). OROV infection in humans causes Oropouche fever, characterized by self-limiting febrile illnesses but that can, in rare cases, lead to hemorrhagic, neurologic, and maternal–fetal complications, even death (1,4).
OROV has been endemic in the Amazon region and expanding in 2024 toward areas of high population density in all Brazil states, as well as to the Caribbean, including Cuba, Barbados, and Panama (5). In addition, traveler-imported cases were identified in the United States, Canada, and Europe. Reports of OROV infection have been identified predominantly by molecular assays; OROV serosurvey studies remain scarce, even in OROV-endemic areas. In this study, we conducted a retrospective cross-sectional serosurvey among residents from Coari and Manaus municipalities in Amazonas State, Brazil, during 2015–2016. The ethics committee of the Federal University of Amazonas approved all procedures for this study (approval nos. 5.876.612 and 6.629.451).
We collected blood samples by venipuncture from residents in Manaus and Coari municipalities (Figure 1). No participants reported any symptoms in the 30 days before blood sample collection. We tested serum samples by 90% plaque reduction neutralization test (PRNT90) to assess the presence and titer of neutralizing antibodies against OROV (5). We determined PRNT90 values from the mean of 2 technical replicates by fitting a 3-parameter log-logistic regression curve to plaque counts normalized to the positive control (Appendix).
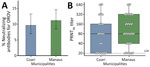
We analyzed serum samples from 814 persons, 374 (45.9%) participants residing in Coari and 440 (54.1%) in Manaus. Median age was 36 (interquartile range [IQR] 26–48) years; 567 (69.7%) participants were female and 247 (30.3%) male. We detected OROV neutralizing antibodies in 85 (10.4%, 95% CI 8.5%–12.8%) participants (Figure 2, panel A). Seroprevalence, as indicated by presence of neutralizing antibodies, was higher in Manaus (49/440 [11.1%, 95% CI 8.4%–14.5%] positive) than in Coari (9.6%, 36/374, 95% CI 6.9%–13.2%) (Figure 2, panel A). The median PRNT90 titer in Manaus was 80 (IQR 40–320), whereas in Coari the median PRNT90 titer was 80 (IQR 20–160) (Figure 2, panel B). Participants testing seropositive for OROV had a median age of 47 (IQR 38–54) years; median age in Manaus was 48 (IQR 42–54) years and in Coari, 45 (IQR 33–53) years. OROV seropositivity rates were higher in female than male participants (65/85 [76.5%]) overall, 39/49 (79.6%) in Manaus and 26/36 (72.2%) in Coari. We observed no statistically significant difference in sex or age distribution in neutralizing antibody–positive participants between Coari and Manaus.
Our findings identified OROV neutralizing antibodies in residents of Coari and Manaus, suggesting prior OROV transmission in those locations during 2015–2016. The seropositivity rate found in our study was lower than that reported in previous studies from the Peruvian Amazon (18%–35%) (6,7). However, our results are similar to those of a 2014–2015 study in Amapá State, Brazil, which found a 10.2% seropositivity rate (8). We cannot ascertain the timing of OROV infection on the basis of neutralizing antibody data; however, some seropositive cases might be linked with OROV outbreak in Manaus during 1980–1981, where 83/496 (16.7%) of participants from 6 neighborhoods in Manaus tested positive for OROV-specific antibodies after a major OROV outbreak (9). Conversely, Oropouche fever was first reported to the Ministry of Health in Coari on December 18, 2023, suggesting a potential undetected OROV transmission not captured by the local surveillance system. We hypothesize that underreporting of OROV infections in Coari could result from several factors, including variable healthcare-seeking behavior, potential higher levels of oligosymptomatic or asymptomatic cases during OROV infection, and misdiagnosis from similar clinical manifestations of other endemic arboviruses, such as dengue, chikungunya, Zika, and Mayaro viruses (10).
A limitation of our study is that we focused on previous OROV infections, using a serologic approach for previous infection; molecular studies and detection of IgM are needed to determine the percentage of participants actively infected, particularly during outbreaks. Second, we were unable to determine whether OROV infections occurred in urban or forest settings. Third, some positive samples could have resulted from the cross-reaction with other orthobunyaviruses that remain to be detected in this region. Finally, larger-scale longitudinal studies are needed to determine the effect of OROV, both before and after future OROV outbreaks.
In conclusion, our study demonstrated evidence of OROV circulation in Coari and Manaus before 2015–2016. Our findings highlight the critical need for continuous molecular and serologic laboratory surveillance for OROV to accurately assess its burden in the Amazon Region and beyond, particularly since its reemergence in 2023.
Ms. Forato is a PhD student at the Department of Genetics, Evolution, Microbiology, and Immunology at the University of Campinas, São Paulo, Brazil. Her research interests include epidemiologic surveillance of emerging viruses, with a focus on arboviruses.
Acknowledgment
P.L. is supported by Fundação de Amparo à Pesquisa do Estado do Amazonas (Posgrad, CT&I Áreas Prioritárias, Iniciativa Amazônia +10, Chamada ERC 2022, and PEX-CT&I/FAPEAM); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Funding 001). J.L.P.M. is supported by São Paulo Research Foundation (no. 2022/10442-0), and National Council for Scientific and Technological Development (CNPq, no. 309971/2023-3). W.M.dS. is supported by Burroughs Wellcome Fund – Climate Change and Human Health Seed Grants (no. 1022448), and Wellcome Trust–Digital Technology Development Award in Climate Sensitive Infectious Disease Modelling (no. 226075/Z/22/Z). J.F. is supported by the São Paulo Research Foundation (grant no. 25/18991-0). G.C.S. is supported by the São Paulo Research Foundation (grant no. 24/12668-0). B.B.S. is supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (grant no. 88887.967871/2024-00).
References
- Travassos da Rosa JF, de Souza WM, Pinheiro FP, Figueiredo ML, Cardoso JF, Acrani GO, et al. Oropouche virus: clinical, epidemiological, and molecular aspects of a neglected orthobunyavirus. Am J Trop Med Hyg. 2017;96:1019–30. DOIPubMedGoogle Scholar
- de Souza WM, Calisher CH, Carrera JP, Hughes HR, Nunes MRT, Russell B, et al. ICTV virus taxonomy profile: Peribunyaviridae 2024. J Gen Virol. 2024;105:
002034 . DOIPubMedGoogle Scholar - Pinheiro FP, Travassos da Rosa APA, Gomes MLC, LeDuc JW, Hoch AL. Transmission of Oropouche virus from man to hamster by the midge Culicoides paraensis. Science. 1982;215:1251–3.PubMedGoogle Scholar
- das Neves Martins FE, Chiang JO, Nunes BTD, Ribeiro BFR, Martins LC, Casseb LMN, et al. Newborns with microcephaly in Brazil and potential vertical transmission of Oropouche virus: a case series. Lancet Infect Dis. 2024;25:155–65.PubMedGoogle Scholar
- Scachetti GC, Forato J, Claro IM, Hua X, Salgado BB, Vieira A, et al. Re-emergence of Oropouche virus between 2023 and 2024 in Brazil: an observational epidemiological study. Lancet Infect Dis. 2025;25:166–75. DOIPubMedGoogle Scholar
- Watts DM, Phillips I, Callahan JD, Griebenow W, Hyams KC, Hayes CG. Oropouche virus transmission in the Amazon River basin of Peru. Am J Trop Med Hyg. 1997;56:148–52. DOIPubMedGoogle Scholar
- Baisley KJ, Watts DM, Munstermann LE, Wilson ML. Epidemiology of endemic Oropouche virus transmission in upper Amazonian Peru. Am J Trop Med Hyg. 1998;59:710–6. DOIPubMedGoogle Scholar
- de Lima RC, Dias HG, de Souza TMA, Familiar-Macedo D, Ribeiro ED, Corrêa VCE, et al. Oropouche virus exposure in febrile patients during chikungunya virus introduction in the state of Amapá, Amazon Region, Brazil. Pathogens. 2024;13:469. DOIPubMedGoogle Scholar
- Borborema CA, Pinheiro FP, Albuquerque BC, da Rosa AP, da Rosa JF, Dourado HV. 1st occurrence of outbreaks caused by Oropouche virus in the State of Amazonas [in Portuguese]. Rev Inst Med Trop São Paulo. 1982;24:132–9.PubMedGoogle Scholar
- de Souza WM, Ribeiro GS, de Lima STS, de Jesus R, Moreira FRR, Whittaker C, et al. Chikungunya: a decade of burden in the Americas. Lancet Reg Health Am. 2024;30:
100673 . DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: April 10, 2026
1These authors contributed equally to this article.
2These senior authors contributed equally to this article.
Table of Contents – Volume 32, Number 4—April 2026
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Pritesh Lalwani, Instituto Leônidas e Maria Deane, Fiocruz Amazônia, Manaus, Brazil, Rua Terezina, 476–Adrianópolis, Manaus, Amazonas State, 69057-070, Brazil
Top