Volume 5, Number 2—April 1999
Dispatch
Acute Hemorrhagic Conjunctivitis Due to Enterovirus 70 in India
Figure
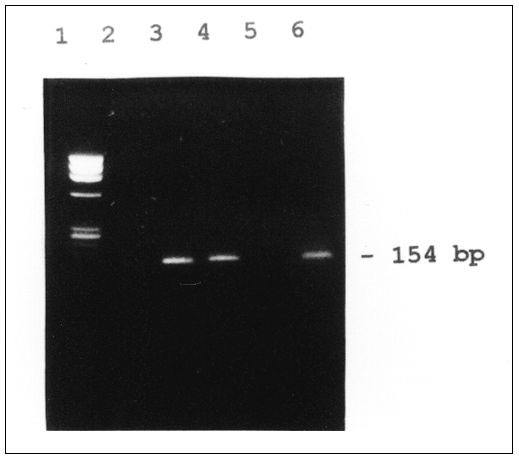
Figure. Reverse transcriptase-polymerase chain reaction (RT-PCR) for enteroviral RNA from acute hemorrhagic conjunctivitis cases. Lane 1: pX174 DNA/Hae III marker; lane 2: negative control; lane 3: positive control (enteroviral RNA); lanes 4-6: clinical samples. For PCR, viral RNA was extracted by the guanidinium thiocyanate method (2) from 200 µl of clinical specimen. The RNA pellet was resuspended in 10 µl of diethyl pyrocarbonate treated water. A 5-µl volume was used for cDNA synthesis. The primers used for PCR amplification were selected from the highly conserved 5' noncoding region of enterovirus described by Rotbart et al. (3) and, using the same protocol, cDNA for PCR was synthesized by reverse transcription. Briefly, cDNA (10µ Cl) was amplified in a PCR mix volume of 50 µl, containing 100 ng of each primer, 250 µl of each dNTP, 1.5 mM MgCl2, 2.5 units Taq polymerase, and 5 µl of 10X PCR buffer. The tubes were subjected to 30 cycles at 94°C for 1 minute, 58°C for 1 minute, and 72°C for 2 minutes, respectively, followed by a final extension for 7 minutes at 72°C. A 154-bp sized band of the PCR-amplified product was visualized under UV illumination on a 2% agarose gel after ethidium bromide staining.
References
- Pass RF, Britt WJ, Stagno S. Cytomegalovirus. Diagnostic procedures for viral, rickettsial and chlamydial infections, 7th ed. In: Lennette EH, Lennette DA, Lennette ET, editors. Washington: American Public Health Association; 1995.
- Chomczynski P, Sacchi N. Single step method of RNA isolation by acid guanidinium thiocyanate phenol chloroform extraction. Ann Clin Biochem. 1987;162:156–9.
- Rotbart HA. Enzymatic amplification of the enteroviruses. J Clin Microbiol. 1990;28:438–42.PubMedGoogle Scholar
- Kono R, Miyamura K, Tajiri E, Shiga S, Sasagawa A, Irani PF, Neurological complications associated with acute haemorrhagic conjunctivitis virus infection and its serological confirmation. J Infect Dis. 1974;129:590–3.PubMedGoogle Scholar
- Manjunath N, Balaya S, Mahajan VM. Isolation of enterovirus 70 during conjunctivitis epidemic in Delhi in 1981. Indian J Med Res. 1982;76:653–5.PubMedGoogle Scholar
- Chatterjee S, Quarcoopome CO, Apenteng A. Unusual type of conjunctivitis in Ghana. Br J Ophthalmol. 1970;54:628–30. DOIPubMedGoogle Scholar
- Kono R, Sasagawa A, Ishii K, Sugiura S, Ochi M, Matsumiya H, Pandemic of new type of conjunctivitis. Lancet. 1972;i:1191–4. DOIGoogle Scholar
- Christopher S, John J, Charles V, Ray S. Coxsackie virus A24 variant EH 24/70 and enterovirus type 70 in an epidemic of acute haemorrhagic conjunctivitis-a preliminary report. Indian J Med Res. 1977;65:593–5.PubMedGoogle Scholar
- Pal SR, Szucs Gy, Melnick JL, Kaiwar R, Bharadwaj G, Singh R, . Immunofluorescence test for the epidemiological monitoring of acute haemorrhagic conjunctivitis cases. Bull World Health Organ. 1983;61:485–90.PubMedGoogle Scholar
- Kishore J, Manjunath N, Bareja U, Verma LK, Broor S, Seth P. Study of an outbreak of epidemic conjunctivitis in Delhi in 1986. Indian J Pathol Microbiol. 1989;32:266–9.PubMedGoogle Scholar
- Broor S, Kishore J, Dogra V, Satapathy G, Seth P. An epidemic of acute haemorrhagic conjunctivitis caused by Coxsackie A24 variant. Indian J Med Res. 1992;95:253–5.PubMedGoogle Scholar
- Bhide VS, Prasad SR, Gogate SS. Isolation of a variant of enterovirus 70 from a patient during an epidemic of acute haemorrhagic conjunctivitis in Pune in 1991. Acta Virol. 1994;38:245–6.PubMedGoogle Scholar
- Uchio E, Yamazaki K, Aoki K, Ohno S. Detection of enterovirus 70 by polymerase chain reaction in acute haemorrhagic conjunctivitis. Am J Ophthalmol. 1996;122:273–5.PubMedGoogle Scholar