Volume 7, Number 2—April 2001
THEME ISSUE
4th Decennial International Conference on Nosocomial and Healthcare-Associated Infections
Prevention is Primary
Preventing Nosocomial Mycobacterium tuberculosis Transmission in International Settings
Integrating Important Principles from Existing Guidelines
Collecting Local Epidemiologic Data
Emphasizing Local Capabilities and Priorities
Administrative Control
Patients
Contacts
Staff
Engineering Controls
Respirator Protection
Ongoing Monitoring for Efficacy
Conclusions
Cite This Article
Cite This Article
Citation for Media
Abstract
Tuberculosis (TB) is a worldwide disease, and nosocomial transmission is known to occur. Authoritative preventive guidelines such as the one developed by the Centers for Disease Control have been published, but the expenses for implementing them can be prohibitive. Each country needs to develop its own protocol to prevent nosocomial transmission of TB. This article describes the key elements of a protocol undertaken for all public hospitals in Hong Kong, where TB is endemic.
Tuberculosis (TB) is an international disease of epidemic proportions. More than 3 million reported cases occur worldwide each year (1), and the actual incidence is estimated to be >10 million cases (2). The World Health Organization (WHO) has published a global strategy for TB control in the community (3) and has called on all nations to develop national TB programs. However, preventing TB in the hospital is just as critical internationally. This report focuses on issues related to preventing nosocomial TB in the international setting.
Numerous guidelines for preventing nosocomial TB have been introduced in the industrialized world. One of the most authoritative protocols is the guideline formulated by the Centers for Disease Control and Prevention (CDC) (4). Implementating this guideline, however, can be expensive. Various studies have estimated that the cost of preventing one case of occupational TB in a hospital, using the CDC guideline, could run into millions of U.S. dollars (5,6). This expense is a heavy burden for hospitals and beyond the capability of many developing countries.
The expense is related to the elaborate demands in the CDC guideline, which was developed in 1994 specifically for the United States after a serious resurgence of TB. The urgency of the matter was summarized succinctly in the 1993 document of the U.S. Occupational Safety and Health Administration (OSHA) (7). New TB cases had increased by 18%, reversing an 18-year downward trend. Outbreaks had occurred in many hospitals, and at least five health-care workers had died. Under such a cloud, making impeccable recommendations in spite of high expenses in cost and manpower seemed reasonable.
The situation can be entirely different in other countries, and therefore guidelines should be tailored to meet local needs. This paper discusses the approach needed to formulate a local TB prevention guideline for hospitals, using a guideline for public hospitals developed in Hong Kong. The challenge is to develop a tool that will be effective locally and yet remain consistent with established scientific principles. At least four elements are needed for a successful local program: 1) integrating important principles from existing guidelines; 2) collecting local epidemiologic data; 3) taking into account local capabilities and priorities; and 4) ongoing monitoring for efficacy.
The first element of a successful local TB prevention program is to integrate important principles from existing guidelines. Building on the work of others is critical. The CDC guideline is an important source, as is a guideline for health-care facilities formulated by WHO (8).
A useful concept in these guidelines is three levels of control measures, ordered according to their importance and priority for implementation: 1) administrative controls, which are aimed at reducing the TB exposures of health-care workers; 2) engineering controls, which are environmental methods to reduce the concentration of droplet nuclei in the air; and 3) personal respiratory protection for health-care workers who are exposed to TB in patient care (4). The protocol we developed in Hong Kong adopted these three levels as its basic format.
A second element of a successful local TB prevention protocol is collection of local epidemiologic data. Accurate local data on the incidence of TB can be difficult to obtain. Fortunately, most countries do have case notification data. In cities like Hong Kong, which have effective TB control programs, case reports approximate the true incidence of TB (9).
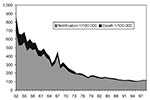
In Hong Kong, the incidence of TB peaked in 1952, and BCG vaccine was made mandatory at birth. Subsequently, the incidence and crude death rate dropped dramatically (Figure 1). Nevertheless, TB remains endemic in Hong Kong, with an incidence rate of 1/1,000 population for the past decade.
Figure 2 shows the antimicrobial drug-resistance rate for TB strains isolated in the government laboratory in 1998. Multidrug-resistant (MDR)-TB is still relatively low, at 1.3%. One reason may be the effective use of short-course therapy (five drugs), provided free to the public for the past 20 years.
Finally, we collected data from large, acute-care public hospitals that participated in the surveillance network of health-care workers who had nosocomial TB. In Hong Kong, infection control units are in place in most public hospitals, and, with the help of the hospital laboratory, staff clinic, and human resource departments, they regularly identify staff diagnosed with active TB. Data should be especially accurate after 1996, when a new law, the Occupational Safety and Health Ordinance, made reporting of employees with active TB mandatory. There is also a strong personal incentive for reporting because the ordinance stipulates compensation for verified TB cases. The incidence of health-care workers with active TB was found to be consistently below that of the general populace, even when the rates were adjusted for the younger ages of the health-care workers from 1994 (Table). This trend persisted even after the ordinance was introduced, making underreporting unlikely.
Surveys of health-care workers to identify tuberculin skin-test conversions are not conducted in Hong Kong. Such surveys would not be accurate for detecting active infections because BCG is given at birth and repeated if needed in the school health system. Furthermore, if the incidence of active TB in health-care workers is clearly below the general populace and the first prerogative of infection control is preventing active disease (10), the value of surveys that identify only immune responses is questionable.
In summary, TB is still endemic in Hong Kong, but the incidence has been stable for more than a decade. The percentage of MDR-TB cases is small, and the incidence of active TB in health-care workers is lower than in the general population. This low incidence is probably due to a high herd immunity. The mandatory BCG vaccination with repeated challenges from a TB- endemic environment and a robust general health must certainly be contributing factors. Nevertheless, local data indicate that, unlike the United States in 1993, no TB crisis confronts Hong Kong.
The third element in a successful local TB-prevention program is taking into account local capabilities and priorities. A guideline for preventing TB in the hospital was introduced in 1996 in Queen Mary Hospital, the teaching hospital for the University of Hong Kong. The guideline was then formally endorsed by the authorities as the reference guideline for all public hospitals in the territory.
The underlying assumption was that no crisis situation was at hand in Hong Kong; thus, drastic measures were probably not required. Nevertheless, best possible practice within the allocated resources ought to be promoted. The salient points of this guideline are summarized below.
Administration control is focused on three sectors of the hospital: patients, contacts, and staff.
The first strategy is to minimize hospitalization of TB patients. Pulmonary TB patients are generally treated as outpatients in Hong Kong. For those admitted, a 24-hour laboratory service for sputum microscopy is provided. The infection control nurse reviews all TB cases diagnosed by the laboratory (both smears and cultures) and facilitates their discharge or transfer to designated TB hospitals. In Queen Mary Hospital, under such a system, 95% of TB patients are discharged from the hospital within 4 days of a positive microbiology report.
An attempt is made to isolate patients with active disease for 2 weeks, but since facilities are limited, priority is given to those who are strongly (+++) smear positive, AIDS patients, and those suspected of having MDR-TB. If isolation cannot be maintained for 2 weeks, it is maintained for up to 5 days after effective chemotherapy has begun. Even when isolation is not possible, exposure of patients to neonates, young children, and immunocompromised hosts is not permitted for 4 weeks.
The admission rates for TB patients in Hong Kong hospitals are rather high and in Queen Mary Hospital, more than 200 inpatients are seen each year. In spite of this, the low incidence of health-care workers with active TB suggests that the risk of active infection in contacts is not overly high. Therefore, draconian measures to investigate contacts are not recommended.
However, when a strongly (+++) smear-positive patient is seen in a high-risk area (with neutropenic patients or neonates), a list of contacts in the same cubicle is generated. Those who have had prolonged contact (>3 weeks) or who have symptoms suggestive of TB are given a chest X ray. All contacts of a strongly smear-positive case who are immunocompromised or children <3 years old are followed up for 3 months.
Chemoprophylaxis is generally not recommended for contacts but may be considered for infants who are exposed. All contacts are counseled to obtain a chest X ray if they develop symptoms suggestive of pulmonary TB that last for 3 weeks.
The infection control nurse conducts surveillance for active TB in health-care workers. Physical therapists are to avoid chest drainage on patients who are smear positive unless they are connected to a closed suction system. A respirator mask is provided for a health-care worker if intubation is needed for patients who are smear positive.
Some strategies routinely recommended elsewhere were not included in the Hong Kong guideline. An assessment of transmission risk at all sites is not conducted. The admission rate for TB is so high that it seems reasonable to assume that the frequency of exposure is probably high in most departments. This high number of admissions also makes routine education of contacts and staff difficult. The suggestion of triage and special precautions in departments such as accident and emergency and radiology was proposed, but not adopted by the respective departments because they never had nosocomial TB reported nor encountered difficulties with their present arrangements. As stated above, surveys of health-care workers for TST conversion is not done, nor are surveys of chest X rays or symptoms because these are reported to be inaccurate (8).
Engineering controls are another major point of our guideline. In hospitals with no central air conditioning, a specially designed isolation room is not provided. In fact, WHO has stated that hospitals ought to "maximize natural ventilation through open windows" (8). Negative-pressure isolation rooms are usually installed in hospitals with central air conditioning. The locations of these isolation rooms, as with the 10 available in Queen Mary Hospital, must be clearly listed in the guideline for the hospital. The number of isolation rooms provided is generally insufficient, and therefore contingency plans with a priority list for isolation are included as recommendations in the guideline.
Other control measures for proven TB cases are included in the guideline. Filters are used on ventilated patients and changed daily. Heat mist exchangers are recommended to avert frequent tubing change. Finally, for patients in the intensive care unit, a closed suction system with disposable suction canisters and tubings is recommended. UV lights and portable HEPA filters are not recommended in Hong Kong.
Respirator protection is another feature of our guideline. Special N95 masks are provided only for bronchoscopists and staff with substantial contact (e.g., during intubation) with patients who have active TB and are not on effective chemotherapy. For other patient-care activities, only the surgical mask is recommended. There is no evidence that the N95 is better than the surgical mask in preventing employee skin-test conversion in the United States (11). Routine fit testing and medical screening, as mandated by OSHA in America (7), are not conducted, as even U.S. specialists have questioned their benefit (11).
The efficacy of the preventive measures should be monitored. In Hong Kong, this is made possible by the ongoing surveillance program for TB in health-care workers. Our guideline was introduced in 1996. Surveillance data in 1997 and 1998 (Table) should offer an evaluation on its effectiveness.
With the resurgence of TB as a global problem, due attention needs to be given to this disease in the health-care setting. Although authoritative guidelines for preventing nosocomial TB are available, each country needs to develop its own specific protocol because, to be effective, guidelines must address local issues such as disease patterns and resource availability. The Hong Kong experience hopefully can be a model for other hospitals engaged in similar undertakings.
Dr. Seto Wing Hong is the chief of service for microbiology in Queen Mary Hospital, the teaching hospital for the University of Hong Kong. He is also chairman of infection control and director of the Quality Improvement Unit in the hospital.
Acknowledgment
Special thanks to Dr. C. M. Tam and the Chest Service of the Department of Health, Hong Kong, for assistance in preparing this report.
References
- World Health Organization. Global Tuberculosis Programme - global tuberculosis control. WHO Report 1997. Geneva: World Health Organization; 1997.
- Dolin PJ, Raviglione MC, Kochi A. Estimates of future global tuberculosis incidence and mortality during 1990-2000. Bull World Health Organ. 1994;72:213–20.PubMedGoogle Scholar
- Maher D, Chaulet P, Spinaci S, Harries A. Treatment of tuberculosis: guidelines for national programmes. 2nd ed. Geneva: World Health Organization; 1997.
- Centers for Disease Control and Prevention. Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care facilities, 1994. MMWR Morb Mortal Wkly Rep. 1994;43(RR-13).
- Adal KA, Anglim AM, Palumbo CL, Titus MG, Coyner BJ, Farr BM. The use of high-efficiency particulate air-filter respirators to protect hospital workers from tuberculosis. N Engl J Med. 1994;331:169–73. DOIPubMedGoogle Scholar
- Nettleman MD, Frederickson M, Good NL, Hunter SA. Tuberculosis control strategies: The cost of particulate respirators. Ann Intern Med. 1994;121:37–40.PubMedGoogle Scholar
- US Department of Labor. OSHA. Enforcement procedures and sheduling for ocupational eposure to tuberculosis. OSHA Instruction CPL 2.106. Washington: Occupational Safety and Health Administration; 1996.p.1-21.
- World Health Organization. Guidelines for the prevention of tuberculosis in health care facilities in resource-limited settings. WHO/TB/99.269. Geneva: World Health Organization; 1999.
- Maher D, Raviglione M. The global epidemic of tuberculosis: A WHO organization perspective. In: Tuberculosis and nontuberculosis mycobacterial infections. Schlossberg D, editor. Philadelphia: WB Saunders; 1999.p. 104-15.
- Gerberding JL. Occupational infectious diseases or infectious occupational diseases? Bridging the view on tuberculosis control. Infect Control Hosp Epidemiol. 1993;14:686–8. DOIPubMedGoogle Scholar
- Woeltje KF. Tuberculosis: What you don't know can hurt you. Infect Control Hosp Epidemiol. 1998;19:626–8. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 7, Number 2—April 2001
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Seto Wing Hong, Dept. of Microbiology, Queen Mary Hospital, Pokfulam Road, Hong Kong, SAR, China; fax: 852-2872-4555
Top