Volume 7, Number 4—August 2001
THEME ISSUE
West Nile Virus
West Nile Virus
Clinical Characteristics of the West Nile Fever Outbreak, Israel, 2000
Cite This Article
Citation for Media
Abstract
West Nile (WN) virus is endemic in Israel. The last reported outbreak had occurred in 1981. From August to October 2000, a large-scale epidemic of WN fever occurred in Israel; 417 cases were confirmed, with 326 hospitalizations. The main clinical presentations were encephalitis (57.9%), febrile disease (24.4%), and meningitis (15.9%). Within the study group, 33 (14.1%) hospitalized patients died. Mortality was higher among patients >70 years (29.3%). On multivariate regressional analysis, independent predictors of death were age >70 years (odds ratio [OR] 7.7), change in level of consciousness (OR 9.0), and anemia (OR 2.7). In contrast to prior reports, WN fever appears to be a severe illness with high rate of central nervous system involvement and a particularly grim outcome in the elderly.
West Nile (WN) virus was first isolated and identified from the blood of a febrile woman in Uganda in 1937 (1); fever was her only known symptom. In the early 1950s, several reports from outbreaks in Israel were the first to detail the clinical characteristic of this illness; the clinical picture that emerged was that of a benign febrile disease in young adults (2,3). In a later outbreak from Israel in 1957, a linkage between WN virus infection and severe central nervous system (CNS) disease was first noted, and a correlation between the age of patients and severity of disease was established (4).
In the last decade, two outbreaks gained attention: the first from Romania in 1996, where a high percentage of CNS involvement was noted, and the second from New York in 1999, in which several cases of flaccid paralysis were described (5,6). The outbreak in New York was the first time WN fever was reported in the Western Hemisphere (7).
From August to October 2000, 417 laboratory-confirmed WN fever cases occurred in Israel; 326 were hospitalized cases. We collected clinical data on 233 of the hospitalized population from 12 different hospitals throughout the country. We report the clinical characteristics of these patients.
Study Population
A case of WN fever was defined as illness in a patient with a clinical picture consistent with WN fever and with anti-WN virus immunoglobulin (Ig) M antibodies detected in either a serum or cerebrospinal fluid (CSF) specimen. The study population was persons who were hospitalized and diagnosed from August 1, 2000, to October 31, 2000. Patients with headache and abnormal CSF findings, with no confusion or change in level of consciousness (i.e., drowsiness to coma), were diagnosed as having meningitis. Patients with an altered level of consciousness, confusion, or focal neurologic signs were diagnosed as having encephalitis, regardless of CSF findings. Patients with severe weakness of limbs or flaccid paralysis were diagnosed as having myelitis. Patients with fever and no CNS symptoms were diagnosed as having febrile disease.
A total of 325 WN fever patients were hospitalized in 20 hospitals. Data were obtained from 12 (60%) of the hospitals and 233 (71%) of the 326 patients. The geographic location of hospitals that participated in the study was similar to that of nonparticipating hospitals. No exclusion was made based on patient characteristics.
Demographic, epidemiologic, and clinical data were collected by infectious disease specialists in each hospital, using a structured questionnaire. Information was obtained by interviewing the patients and reviewing medical records or by reviewing the charts in cases in which the diagnosis was confirmed after the discharge of the patients.
Serologic Testing
Serologic diagnosis of WN virus infection was based on an IgM-capture enzyme-linked immunosorbent assay (ELISA), performed in serum or CSF samples. The assay, which was developed in the Central Virology Laboratory during 1999 and 2000, includes the following steps: coating of ELISA plate with goat anti-human IgM and stepwise incubation with the patient's serum (diluted 1:100 and 1:2,000) or CSF (diluted 1:10 and 1:100 for CSF); WN virus antigen was prepared from Vero cells infected with an Israeli gull isolate from 1999; mouse anti-flavivirus monoclonal antibodies (TropBio, Australia); horseradish peroxidase conjugated goat anti-mouse antibody (Jackson Imunoresearch Laboratories, West Grove, PA, USA),and o-phenylenediamine as substrate. Cross-reaction with other flaviviruses was not thoroughly evaluated; however, no other known endemic cross-reacting Flavivirus infects humans in Israel.
Statistical Analysis
The Fisher exact test was applied to compare differences between diagnosis by age--<70 years or >70 years. A two-step process was used to determine which of the proposed risk factors were independently associated with death. A bivariate logistic regression analysis was used to determine the association of individual risk factors with death. Subsequently, a multivariate logistic regression analyses was performed, with a forward elimination model. Variables for this analysis were selected if they fulfilled the following criteria: 1) level of significance of p<0.15 in the bivariate analysis; 2) data on variables were available in >92% of patients; and 3) variables on which sufficient information was available and occurred in more than 10% of the cases. Data were analyzed with the SAS program (version 6.12).
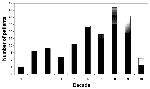
The age distribution of the 233 patients for whom data were available is delineated in Figure 1. The mean age of the patients was 59 (±23.5 standard deviations) years old. The median age was 65 years (range 3 to 95 years). A large percentage (79.8%) of patients had one or more coexisting illnesses or conditions, including hypertension, diabetes mellitus, ischemic heart disease, renal failure, obstructive lung disease, and immunodeficiency such as organ transplantation, malignancy, and chemotherapy.
Three main clinical presentations were prominent in hospitalized patients with WN fever: encephalitis (57.9%), febrile illness (24.4%), and meningitis (15.9%). Compared with persons <70 years of age (n=134), older persons (n=99) were more likely to have encephalitis (80% vs. 41%) and were less likely to have febrile illness (18% vs. 30%) or meningitis (1% vs. 27%) (p<0.00001).
Table 1 presents the symptoms and signs during the disease course. Fever (>38°C) was almost universally present. Sixty percent of the patients had fever above 39°C and 20% had fever above 40°C. Headache, myalgia, chills, and rash were common, as were gastrointestinal complains such as abdominal pain and diarrhea. Lymphadenopathy was infrequent (Table 1).
Symptoms and signs that suggested CNS involvement were common: nuchal rigidity was present in 28.7%, confusion in 39.5%, and change in level of consciousness in 46.8%; 17.7% percent of the hospitalized patients deteriorated into coma. Thirty-seven patients received enteral ribavirin as an experimental therapy
Lumbar puncture was performed in 153 patients (65.6%). The findings were characteristic of viral infection and demonstrated mild leukocytosis (median 77 leukocytes/mm3; range 0 to 1,750), high protein (median 85 mg/dL; range 18 to 1,900), and normal glucose (median 67 mg/dL; range 2.8 to 197).
Brain computerized tomography (CT) scan was performed for 105 patients. The findings were interpreted as normal in 62 patients (58%). Abnormal findings included old infarcts (18%), cortical atrophy (13%), meningioma (4%), old hemorrhages (2%), and multiple brain metastasis (1%). None of these findings were attributable to WN virus infection. Electroencephalogram (EEG), performed in 43 patients, was consistent with encephalitis in 34 (79%). No specific pattern for WN virus infection was found.
Laboratory results are depicted in Table 2. Anemia was documented in 41.1% of the patients. Leukocytosis, thrombocytopenia, and leukopenia were documented in 35.9%, 14.9%, and 8.6% of patients, respectively. Liver function tests were mildly impaired in 20% of patients. Mild electrolyte abnormalities and elevated urea were noted.
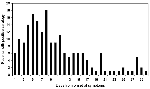
Anti-WN virus IgM antibodies were detected in serum (143 cases), CSF (27 cases) or both (63 cases). Of note, many of these samples were obtained on the first week of symptoms (83 of serum samples and 50 of CSF samples) and were found to be already positive at that time (Figure 2).
Thirty-three patients (14.1%) in the study group died during hospitalization. Their median age was 80 years (range 54 to 95). The mortality rate among patients >70 years increased to 29.3%. All fatal cases but one were >68 years. A 54-year-old woman with myasthenia gravis, treated with azathioprine, was the youngest to die from WN virus encephalitis.
We tried to identify clinical and laboratory variables associated with death. By bivariate logistic regression analysis, age >70 years, headache, confusion, changes in level of consciousness, anemia on admission, and ribavirin therapy were found to be associated with death. Lack of a coexisting condition was protective (p<0.05). No single coexisting condition was found to be a significant predictor of death.
Sixteen patients were immunocompromised because of heterogeneous conditions, i.e., organ transplantation, malignancy, and chemotherapy. Only four of these patients were >70 years. Mortality rate was higher among the immunocompromised (5/16) than among those not immunocompromised (28/217); p=0.052, odds ratio [OR] 3.1, 95% confidence intervals 1.0-9.5).
Multivariate logistic regression analysis was performed with all variables in Table 3. Three variables were independently correlated with death: age >70 years (OR 7.7, p= 0.0004), change in level of consciousness (OR 9.0, p=0.0007), and anemia at presentation (OR 2.7, p=0.028).
In this report we summarized the clinical characteristics of 233 hospitalized patients during a large-scale outbreak of WN fever in Israel. Our data indicated that WN fever was a severe disease with significant CNS involvement and high mortality (14.1% death rate in the study group). Furthermore, a clear correlation between age and increased severity and death was noted. Eighty percent of hospitalized patients >70 years of age had encephalitis, and deaths in this age group rose to 29.3%. Such findings raise the question of whether old age was a risk factor in itself or whether coexisting conditions associated with advanced age account for the observed increased risk. Importantly, in the bivariate regression analysis no single coexisting condition or illness was correlated with death. Moreover, in the multivariate regression analysis, age >70 years was an independent risk factor. These findings agree with the results of a case-control study in Romania in which no correlation between an underlying medical condition and meningoencephalitis was found (8).
Immunosuppressive therapy, which nearly reached statistical significance, may be the exception to this finding. Notably, the only fatality in a patient <68 years occurred in a woman treated by immunosuppressive therapy.
The reasons for the high death rates observed in the present epidemic are not clear. Mortality was higher than that described in the outbreak in Romania in 1996 (14.3% vs. 4.3%, respectively), despite the fact that a high percentage of CNS involvement (89%) was noted in Romania as well (9). High background immunity may account for lower morbidity and deaths. However, the outbreak in Romania was the first reported from that area, and therefore occurred in a population with low background immunity. In fact, the seroprevalence after the outbreak in Romania was reported to be 4.1% (5), compared with seroprevalence of 18.6% in a healthy adult population in Israel in the 1980s (10). Thus, background immunity of the population could not explain the difference in death rates.
Introduction of a new, more virulent strain of WN virus is another possible explanation for the high death rate. Studies in North America support this hypothesis. Mortality during the outbreak in New York (11.4%) was closer to that from Israel. Viral isolates from the outbreak in New York showed a 99.8% genomic similarity to WN virus strain from the brain of a dead goose in Israel in 1998, as well as a fatal human case from Israel in 1999 (11,12). In addition, avian deaths caused by WN viral infection were reported only from North America and Israel, a phenomenon that had not been reported previously. Taken together, these findings suggest that a new strain of WN virus identified in the United States and Israel may be responsible for the higher death rates in these countries.
The signs and symptoms of the disease are not specific, and their percentages seem to differ considerably in the outbreaks reported. Data from confirmed cases of outbreaks in Israel in the early 1950s indicate that fever, headache, and lymphadenopathy were almost universal findings (80% to 100%), with rash in 50% and gastrointestinal complaints in 20% to 30% (2). The study from Romania showed high prevalence of fever and headache but noted only 5% rash, 11% gastrointestinal symptoms, and 2.4% lymphadenopathy (9). In our series, fever, headache, and change in level of consciousness were the most frequent findings; rash was less frequent (28.1%). Differences in study population and case definitions, as well as methods of data collection, may account for such discrepancies. Alternatively, the variation in clinical manifestations may have occurred because of differences between viral strains.
Laboratory results were mostly normal. The mild changes in electrolyte and urea could be attributed to changes in fluid balance in patients with vomiting and diarrhea. Anemia on admission was reported in a high proportion of our patients and was found to be independently associated with fatal outcome. Anemia has not been reported as an important laboratory abnormality in other flaviviral infections (13); with the limited data available, determining whether it was caused by the WN fever or whether it antedated the infection could not be assessed.
A large number of patients underwent brain CT and EEG examinations. CT showed abnormalities in 40% of the patients, but none were specific to the disease and reflected mostly the age of the patients. These results agree with published findings from New York (6).
Generally, the signs, symptoms, laboratory findings, and imaging results in WN fever are nonspecific. Similar to reports from Romania, we found that a high number of blood and CSF specimens obtained during the first week of symptoms were already positive by IgM serology (14). Therefore, this specific test is important for timely diagnosis.
Our study had some limitations. Because the patient population was distributed in multiple hospitals, the physical examination, clinical assessment, and chart review were performed by different physicians. This may have resulted in some skewing of the percentages of the different signs and symptoms attributed to the disease.
Despite a detailed analysis of the clinical manifestations, signs, symptoms, and laboratory results of patients with WN fever, no findings are diagnostic. A febrile illness with neurologic manifestations in elderly patients hospitalized in the summer or fall should raise the possibility of WN fever and prompt a work-up to establish the diagnosis.
Finally, this study identified a specific group with increased risk for death from WN fever. The elderly might be a suitable target group for protective vaccines.
Dr. Chowers is an infectious disease specialist in Meir Medical Center, Kfar Sava, Israel, which is affiliated with Tel Aviv University.
References
- Smithburn JS, Hughes TP, Burke AW, Paul JH. A neurotropic virus isolated from the blood of a native of Uganda. Am J Trop Med Hyg. 1940;20:471–92.
- Marberg K, Goldblum N, Sterk VV, Jasinska-Klinberg W, Klinberg MA. The natural history of West Nile fever: clinical observations during an epidemic in Israel. Am J Hyg. 1956;64:259–69.PubMedGoogle Scholar
- Goldblum N, Sterk VV, Paderski B. West Nile fever. The clinical features of the desease and the isolation of West Nile virus from the blood of nine human cases. Am J Trop Med Hyg. 1954;59:89–103.
- Spigland W, Jasinska-Klingberg W, Hofshi E, Goldblum N. Clinical and laboratory observations in an outbreak of West Nile fever in Israel in 1957. Harefua. 1958;54:275–81.PubMedGoogle Scholar
- Tsai TF, Popovici F, Cernescu C, Campbell GL, Nedelcu NI. West Nile encephalitis epidemic in southeastern Romania. Lancet. 1998;352:767–71. DOIPubMedGoogle Scholar
- Asnis DS, Conetta R, Teixeira AA, Waldman G, Sampson BA. The West Nile virus outbreak of 1999 in New York: the Flushing Hospital experience [published erratum appears in Clin Infect Dis 2000 May;30:841]. Clin Infect Dis. 2000;30:413–8. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: West Nile-like viral encephalitis--New York, 1999. MMWR Morb Mortal Wkly Rep. 1999;48:890–2.PubMedGoogle Scholar
- Han LL, Popovici F, Alexander JJ, Laurentia V, Tengelsen LA, Cernescu C, Risk factors for West Nile virus infection and meningoencephalitis, Romania, 1996. J Infect Dis. 1999;179:230–3. DOIPubMedGoogle Scholar
- Ceausu E, Erscoiu S, Calistru P, Ispas D, Dorobat O, Homos M, Clinical manifestations in the West Nile virus outbreak. Rom J Virol. 1997;48:3–11.PubMedGoogle Scholar
- Cohen D, Zaide Y, Karasenty E, Schwartz M, LeDuc JW, Slepon R, Prevalence of antibodies to West Nile fever, sandfly fever Sicilian, and sandfly fever Naples viruses in healthy adults in Israel. Public Health Rev. 1999;27:217–30.PubMedGoogle Scholar
- Lanciotti RS, Roehrig JT, Deubel V, Smith J, Parker M, Steele K, Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science. 1999;286:2333–7. DOIPubMedGoogle Scholar
- Giladi M, Metzkor-Cotter E, Martin DA, Siegman-Igra Y, Korczyn A, Rosso R, West Nile encephalitis in Israel, 1999: The New York connection. Emerg Infect Dis. 2001;7:659–61. DOIPubMedGoogle Scholar
- Tsai T. Flaviviruses. In: Mandell G, Bennett J, Dolin R, editors. Principles and practice of infectious diseases. Fifth ed. Philadelphia: Churchill Livingstone; 2001:1714-36.
- Cernescu C, Ruta SM, Tardei G, Grancea C, Moldoveanu L, Spulbar E, A high number of severe neurologic clinical forms during an epidemic of West Nile virus infection. Rom J Virol. 1997;48:13–25.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 7, Number 4—August 2001
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Michal Chowers, Infectious Disease Unit, Meir Medical Center, Kfar Sava, Israel; fax: 972-9747-1187
Top