Volume 8, Number 2—February 2002
Dispatch
Cryptosporidium muris Infection in an HIV-Infected Adult, Kenya
Cite This Article
Citation for Media
Abstract
We describe a case of Cryptosporidium muris infection in an HIV-infected adult with diarrhea in Kenya. Sequence analysis of an 840-bp region of the 18S rRNA gene locus demonstrated the isolate had 100% nucleotide identity with C. muris recovered from a rock hyrax, 98.8% with a C. muris “calf” isolate, 95.5% with C. serpentis, but only 87.8% with C. parvum “human” type.
Tyzzer identified the first Cryptosporidium species, C. muris, in the gastric glands of mice (1). Thereafter, he identified C. parvum, which infects the small intestines of many mammals, and described the complete coccidian life cycle. Over the next 70 years, more than 23 different species of the genus were described on the basis of their morphology and natural hosts. However, when animals were experimentally infected, many of the described Cryptosporidium species were found to be identical. In the 1970s the classification was revised, and today only six to eight species are recognized as valid, with most human, zoonotic, and mammalian infections being attributed to the different C. parvum genotypes (2,3). C. muris, which is naturally a murine parasite, appears to have a more limited host range than C. parvum. Experimental transmission studies of C. muris have shown that the isolate from laboratory mice can infect other animals, including dogs, guinea pigs, rabbits, lambs, and gerbils, although it did not produce patent infections (4). The parasite has also been isolated from a rock hyrax (Procavia sp.) from a zoo and a Bactrian camel with chronic cryptosporidiosis (3,5). For many years, the parasite was thought to infect cattle; however, recent studies have shown that the C. muris that infects cattle is genetically distinct, and a new species name, C. andersoni, has been suggested (6).
Conventional diagnostic methods for Cryptosporidium do not differentiate the various species and genotypes, and most infections are diagnosed as C. parvum. C. parvum “human” and “bovine” genotypes remain the main causes of human cryptosporidiosis, but lately identification of infections with other genotypes and also Cryptosporidium species other than C. parvum has increased in both immunosuppressed and immunocompetent persons (7–10). Possible asymptomatic human infection with C. muris was reported in two healthy girls in Indonesia (11). Morphologic studies on the oocysts showed they were most likely to be C. muris, although there was no genotypic or experimental animal confirmation. Phylogenetic analysis has enabled more conclusive assignment to species and genotypes infecting humans and other animals (12,13). We report a case of C. muris infection, confirmed by morphology and genotyping, in an adult HIV-infected man from Kenya, hospitalized with diarrhea.
Fecal samples were collected from diarrheal patients from a hospital in Nairobi, Kenya, as part of a larger study. The patient described was an HIV-infected man who had clinical tuberculosis and diarrhea. Isospora belli was also detected in a fecal sample from the patient.
The fecal samples were preserved in both sodium acetate formalin and 2.5% potassium dichromate and kept at 4°C. They were stained with Kinyoun’s carbol fuchsin modified acid-fast stain and examined by oil immersion microscopy . An aliquot of 400 μL of the sample suspension in 2.5% potassium dichromate was processed for genotypic analysis. Potassium dichromate was washed 5 times with cold, distilled water until the yellow color cleared. Oocysts were ruptured by freeze-thaw, and DNA was extracted by using a QIAamp DNA Mini Kit (Qiagen, West Sussex, UK)) for stool DNA purification as per protocol.
A section of the SSU (18S) rRNA gene was amplified by nested polymerase chain reaction (PCR) as described (14), using the forward primers 5´-TTCTAGAGCTAATACATGCG-3´ and the reverse primer 5´-CCCTAATCCTTCGAAACAGGA-3´ for primary PCR. Secondary PCR used primers 5´-GGAAGGGTTGTATTTATTAGATAAAG-3´ and reverse primer 5´-AAGGAGTAAGGAACAACCTCCA-3´, employing the Techne (FTGENE2D Techne, Cambridge Ltd., UK) thermal cycler. Restriction fragment analysis of the secondary PCR product was done by digesting 15 μL of product in a 40-_L total reaction volume consisting of 15 U of Ssp1 and 3 μL of restriction buffer (Boehringer Mannheim Biochemicals, Indianapolis, IN) for species identification and Asn1 (Boehringer Mannheim) for genotyping in the same concentration at 37°C for 1 hour. Digestion products were separated on 2% agarose gel and visualized by ethidium bromide staining. The internal (secondary) fragment was purified by using the Prep-A-Gene DNA purification kit and cloned into PGEM-T Easy plasmid vector (Promega Corporation, Madison, WI) as described by the manufacturer. The cloned product was sequenced and aligned with previously published sequences of the 18S rRNA gene of Cryptosporidium species by using the CLUSTALX (EMBL, Heidelberg, Germany) program and manual adjustments. Multiple alignment was done with the Phylogeny Inference Package (PHYLIP version 3.5c, J. Felsenstein and the University of Washington, Seattle, WA). Sequences were analyzed by using DNADIST followed by neighbor joining (NEIGHBOR, PHYLIP package). One hundred replica samplings were analyzed for percentage bootstrap values. Accession numbers for Cryptosporidium 18S rRNA genes used were AF093498, AF093497, AF093496, AF108866 and AF093489, AF093499, AF112569 AF115377.
Microscopic examination of the acid-fast stained fecal smear revealed ovoid oocysts that were an average size of 7.5-9.8 x 5.5-7.0 μm (Figure 1). Cysts of I. belli were also identified in the stained smear.
Restriction endonuclease digestion with Ssp1 of the secondary PCR 18S rRNA product yielded two fragments of 385 bp and 448 bp in size, while Asn1 digestion yielded two visible bands that were 102 bp and 731 bp. The results match restriction fragment patterns observed following similar digestions of C. muris amplicons from a rock hyrax and Bactrian camel isolates (14).
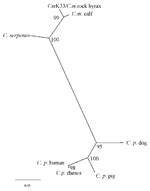
The resulting sequence 18S r RNA gene fragment of the C. muris human isolate was deposited in the EMBL Nucleotide Sequence Database (Accession no. AJ307669). Sequence analysis with ClustalX showed this human C. muris isolate had a 100% nucleotide identity to that of a C. muris isolate from a rock hyrax and a Bactrian camel (EMBL Accession no. AF093498, AF093497), 98.8% identity to a C. muris “calf” isolate (AF093496), 96.5% with C. serpentis (AF108866), and only 87.8% identity to C. parvum human type (AF093489). C. muris calf isolate (AF093496) has since been shown to be a different species from C. muris (“mouse” type, Accession no. AF093498 ) and has been given a new name, C. andersoni. The phylogenetic tree showed topology similar to that already published for Cryptosporidium, with C. parvum clustering in one clade, and our patient’s sample and published sequences of C. muris (rock hyrax isolate), C. andersoni (calf isolate), and C. serpentis clustered in another group (Figure 2).
Our study used genotypic analysis to confirm microscopic detection of Cryptosporidium oocysts in fecal samples and indicated that C. muris can indeed infect humans. Although immunosuppression has been observed to produce an increased susceptibility to cryptosporidiosis, the range of Cryptosporidia that can cause human cryptosporidiosis is still being elucidated (8,13,15). Lately, novel genotypes and non-C. parvum species such as C. meleagridis , C. felis, and C. parvum “dog” type have been identified not only in HIV-infected persons but also in HIV-uninfected patients (7,9,10). Genotypic analysis of Cryptosporidium organisms in fecal samples in the United Kingdom showed the occurrence of C. meleagridis, C. felis, and C. parvum “dog” type in immunocompetent and immunosuppressed persons (10,16). In another study in Peru, C. felis, C. parvum “dog” type, and C. meleagridis were identified in children not infected with HIV. In that study, C. meleagridis was as common as C. parvum “bovine” type; it appeared to be a stable part of the enteric pathogen mix causing cryptosporidiosis, perhaps only being identified with current definitive molecular methods (9).
C. muris infects the gastric glands of immunocompetent or immunocompromised (nude and SCID) mice (17); however, since our patient was co-infected with I. belli, the role of C. muris in our patient’s gastroenteritis and its possible site of infection in this patient are unclear.
A report of possible asymptomatic C. muris infection in healthy persons (11) and our finding of it in an immunosuppressed patient suggest that this may be yet another Cryptosporidium species with a zoonotic potential. The range of animal reservoir hosts in which C. muris has been identified or experimentally transmitted adds to the importance of Cryptosporidium species as a public health concern (3,4,15). The current genotypic analyses are making it possible to make more conclusive diagnoses and to speculate on possible sources of infection (14–16). These techniques will need to be applied more widely to identify and characterize isolates of Cryptosporidium for more definitive epidemiologic mapping.
Gatei Wangeci is a Commonwealth Universities Scholar, working toward a doctorate in parasitology in the Tropical and Medical Schools, University of Liverpool. She qualified in veterinary medicine in 1987 and since then has worked at the Kenya Medical Research Institute in Nairobi as a veterinary parasitologist.
Acknowledgments
We thank Winifred Dove, F.B. Kiliku, and Dr. Njeri Wamae for their assistance.
The study is part of doctoral research grant to Wangeci Gatei from the Association of Commonwealth Universities at the Liverpool School of Tropical Medicine, United Kingdom.
References
- Tyzzer EE. A sporozoan found in the peptic glands of the common mouse. Proc Soc Exp Biol Med. 1907;5:12–3.
- Levine ND. Taxonomy and review of the coccidian genus Cryptosporidium (Protozoa, Apicomplexa). J Protozool. 1984;31:94–8.PubMedGoogle Scholar
- Tzipori S, Griffiths JK. Natural history and biology of Cryptosporidium parvum. In: Baker JR, Muller R, Rollison D, Tzipori S. Advances in parasitology: Opportunistic protozoa in humans. Vol. 40. London: Academic Press; 1998:5-35.
- Aydin Y, Ozkul IA. Infectivity of Cryptosporidium muris directly isolated from the murine stomach for various laboratory animals. Vet Parasitol. 1996;66:257–62. DOIPubMedGoogle Scholar
- Fayer R, Phillips L, Anderson BC, Bus M. Chronic cryptosporidiosis in a bactrian camel (Camelus bactrianus). J Zoo Wildl Med. 1991;22:228–32.
- Lindsay DS, Upton SJ, Owens DS, Morgan UM, Mead JR, Blagburn BL. Cryptosporidium andersoni n. sp. (Apicomplexa: Cryptosporiidae) from cattle, Bos taurus. J Eukaryot Microbiol. 2000;47:91–5. DOIPubMedGoogle Scholar
- Morgan U, Weber R, Xiao L, Sulaiman I, Thompson RC, Ndiritu W, Molecular characterization of Cryptosporidium isolates obtained from human immunodeficiency virus-infected individuals living in Switzerland, Kenya, and the United States. J Clin Microbiol. 2000;38:1180–3.PubMedGoogle Scholar
- Pieniazek NJ, Bornay-Llinares FJ, Slemenda SB, da Silva AJ, Moura IN, Arrowood MJ, New Cryptosporidium genotypes in HIV-infected persons. Emerg Infect Dis. 1999;5:444–9.PubMedGoogle Scholar
- Xiao L, Bern C, Limor J, Sulaiman I, Roberts J, Checkley W, Identification of 5 types of Cryptosporidium parasites in children in Lima, Peru. J Infect Dis. 2001;183:492–7. DOIPubMedGoogle Scholar
- Pedraza-Diaz S, Amar C, Iversen AM, Stanley PJ, McLauchlin J. Unusual Cryptosporidium species recovered from human faeces: first description of Cryptosporidium felis and Cryptosporidium 'dog type' from patients in England. J Med Microbiol. 2001;50:293–6.PubMedGoogle Scholar
- Katsumata T, Hosea D, Ranuh IG, Uga S, Yanagi T, Kohno S. Short report: possible Cryptosporidium muris infection in humans. Am J Trop Med Hyg. 2000;62:70–2.PubMedGoogle Scholar
- Xiao L, Escalante L, Yang C, Sulaiman I, Escalante AA, Montali RJ, Phylogenetic analysis of Cryptosporidium parasites based on the small-subunit rRNA gene locus. Appl Environ Microbiol. 1999;65:1578–83.PubMedGoogle Scholar
- Morgan U, Xiao L, Sulaiman I, Weber R, Lal AA, Thompson RC, Which genotypes/species of Cryptosporidium are humans susceptible to? J Eukaryot Microbiol. 1999;46:42S–3S.PubMedGoogle Scholar
- Xiao L, Morgan UM, Limor J, Escalante A, Arrowood M, Shulaw W, Genetic diversity within Cryptosporidium parvum and related Cryptosporidium species. Appl Environ Microbiol. 1999;65:3386–91.PubMedGoogle Scholar
- Xiao L, Morgan UM, Fayer R, Thompson RC, Lal AA. Cryptosporidium systematics and implications for public health. [Review]. Parasitol Today. 2000;16:287–92. DOIPubMedGoogle Scholar
- McLauchlin J, Amar C, Pedraza-Diaz S, Nichols GL. Molecular epidemiological analysis of Cryptosporidium spp. in the United Kingdom: results of genotyping Cryptosporidium spp. in 1,705 fecal samples from humans and 105 fecal samples from livestock animals. J Clin Microbiol. 2000;38:3984–90.PubMedGoogle Scholar
- McDonald V, Deer R, Uni S, Iseki M, Bancroft GJ. Immune responses to Cryptosporidium muris and Cryptosporidium parvum in adult immunocompetent or immunocompromised (nude and SCID) mice. Infect Immun. 1992;60:3325–31.PubMedGoogle Scholar
Figures
Cite This ArticleTable of Contents – Volume 8, Number 2—February 2002
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
*CA Hart, Department of Medical Microbiology and Genito-Urinary Medicine, University of Liverpool, Duncan Building, Daulby Street, Liverpool, L69 3GA, UK; fax: 44-151-706-5805; e-mail:.
Top