Volume 8, Number 4—April 2002
Research
Genetic and Antigenic Analysis of the First A/New Caledonia/20/99-like H1N1 Influenza Isolates Reported in the Americas
Figure 2
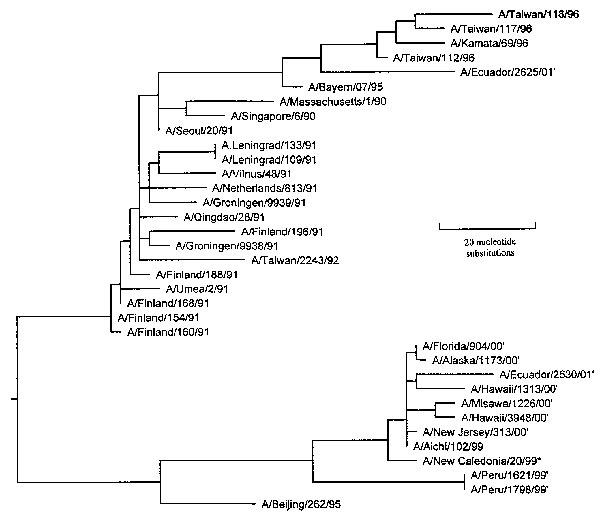
Figure 2. An unrooted phylogenetic analysis of the HA1 gene nucleotide sequence of influenza A H1N1 viruses isolated since 1990. Peru isolates 1621 and 1798 are within the Beijing/262/95 lineage but are more similar to A/New Caledonia/20/99. A number of isolates characterized by Brooks Air Force Base from 1999 to 2001 were also A/NC/20/99-like. The tree was generated by using the Jotun-Hein algorithm (6) in MegAlign software (version 3.18). Horizontal lines are proportional to the number of substitutions between branch points. Asterisk (*) denotes 2000/01 vaccine strain; (1) denotes isolates characterized by Brooks Air Force Base. Brooks Air Force Base isolates are available from Genbank under accession numbers AF268312, AF268313 and AY029287-AY029292.
References
- Centers for Disease Control and Prevention. Influenza 2000. Available from: URL: http://www.cdc.gov/ncidod/diseases/flu/fluinfo.htm
- Breschkin AM, Ahern J, White DO. Antigenic determinants of influenza virus hemagglutinin. Virology. 1981;113:130–40. DOIPubMedGoogle Scholar
- Caton A, Brownlee GG, Yewdell JW, Gerhard W. The antigenic structure of the influenza virus A/PR/8/34 hemagglutinin (H1 subtype). Cell. 1981;31:417–27. DOIGoogle Scholar
- Gerhard W, Yewdell J, Frankel ME. Antigenic structure of influenza virus haemagglutinin defined by hybridoma antibodies. Nature. 1981;290:713–7. DOIPubMedGoogle Scholar
- Caton AJ, Raymond FL, Brownlee GG, Yewdell JW, Gerhard W. Antigenic variation in influenza virus. Biochem Soc Trans. 1983;4:435–41.
- Canas LC. The Department of Defense laboratory-based global influenza surveillance system. Mil Med 2000;165:Suppl 2.
- Williams RJ, Cox NJ, Regnery HL, Noah DL, Khan AS, Miller JM, Meeting the challenge of emerging pathogens: the role of the United States Air Force in global influenza surveillance. Mil Med. 1997;162:82–6.PubMedGoogle Scholar
- World Health Organization. Recommended composition of influenza virus vaccines for use in the 1997-1998 season. Wkly Epidemiol Rec. 1997;72:57–64.PubMedGoogle Scholar
- World Health Organization. Recommended composition of influenza virus vaccines for use in the 1998-1999 season. Wkly Epidemiol Rec. 1998;73:56–63.PubMedGoogle Scholar
- World Health Organization. Recommended composition of influenza virus vaccines for use in the 1999-2000 season. Wkly Epidemiol Rec. 1999;74:321–5.PubMedGoogle Scholar
- Kendal AP, Pereura MS, Skehel J. Concepts and procedures for laboratory based influenza surveillance. Geneva, Switzerland: World Health Organization, 1982.
- Hein JJ. Unified approach to alignment and phylogenies. Methods Enzymol. 1990;183:626–45. DOIPubMedGoogle Scholar
- World Health Organization. Recommended composition of influenza virus vaccines for use in the 2000-2001 season. Wkly Epidemiol Rec. 2000;75:61–8.
- Wilson IA, Cox NJ. Structural basis of immune recognition of influenza virus hemagglutinin. Annu Rev Immunol. 1990;8:737–71. DOIPubMedGoogle Scholar
- Raymond FL, Caton AJ, Cox NJ, Kendal AP, Brownlee GG. Antigenicity and evolution of influenza J1 haemagglutinin, from 1950-1957 and 1977-1983: Two pathways form one gene. Virology. 1986;148:275–87. DOIPubMedGoogle Scholar
- Xu X, Rocha EP, Regenery HL, Kendal AP, Cox NJ. Genetic and antigenic analysis of influenza a (H1N1) viruses, 1986-1991. Virus Res. 1990;28:37–55.
- Centers for Disease Control and Prevention. Update: influenza activity-United States, 1999-2000. MMWR Morb Mortal Wkly Rep. 2000;283:13.
- Centers for Disease Control and Prevention. Influenza activity—United States and Worldwide, April-October. MMWR Morb Mortal Wkly Rep. 2000;49:1006–8.PubMedGoogle Scholar
- Winter G, Fields S, Brownlee GG. Nucleotide sequence of the haemagglutinin gene of influenza virus H1 subtype. Nature. 1981;292:72–5. DOIPubMedGoogle Scholar
- World Health Organization. Recommended composition of influenza virus vaccines for use in the 2001-2002 season. Wkly Epidemiol Rec. 2001;76:58–61.PubMedGoogle Scholar
- Bush RM, Bender CA, Subbarao K, Cox NJ, Fitch WM. Predicting the evolution of human influenza A. Science. 1999;286:1921–5. DOIPubMedGoogle Scholar
- Fitch WM, Bush RM, Bender CA, Cox NJ. Long term trends in the evolution of H(3) HA1 human influenza type A. Proc Natl Acad Sci U S A. 1997;94:7712–8. DOIPubMedGoogle Scholar