Volume 9, Number 3—March 2003
Research
Emergence of Ceftriaxone-Resistant Salmonella Isolates and Rapid Spread of Plasmid-Encoded CMY-2–Like Cephalosporinase, Taiwan
Cite This Article
Citation for Media
Abstract
Of 384 Salmonella isolates collected from 1997 to 2000 in a university hospital in Taiwan, six ceftriaxone-resistant isolates of Salmonella enterica serovar Typhimurium were found in two patients in 2000. The resistance determinants were on conjugative plasmids that encoded a CMY-2–like cephalosporinase. During the study period, the proportion of CMY-2–like enzyme producers among Escherichia coli increased rapidly from 0.2% in early 1999 to >4.0% in late 2000. Klebsiella pneumoniae isolates producing a CMY-2–like β-lactamase did not emerge until 2000. The presence of blaCMY-containing plasmids with an identical restriction pattern from Salmonella, E. coli, and K. pneumoniae isolates was found, which suggests interspecies spread and horizontal transfer of the resistance determinant. Various nosocomial and community-acquired infections were associated with the CMY-2–like enzyme producers. Our study suggests that the spread of plasmid-mediated CMY-2–like β-lactamases is an emerging threat to hospitalized patients and the public in Taiwan.
Because of increasing rates of antimicrobial resistance in salmonellae worldwide (1–4), extended-spectrum cephalosporins, especially ceftriaxone, are frequently used to treat invasive salmonellosis. Since the early 1990s, ceftriaxone-resistant salmonellae have been noted in many countries, including France, Argentina, Algeria, Tunisia, Turkey, Spain, Latvia, the United States, and Hungary (5–17), with resistance conferred by various class A extended-spectrum β-lactamases or class C cephalosporinases (18). These β-lactamases in salmonellae are usually encoded on transmissible plasmids (5–11,13–17), which could be acquired from other multidrug-resistant Enterobacteriaceae, such as Klebsiella pneumoniae or Escherichia coli (10,19).
Although the prevalence of multidrug-resistant salmonellae is a major public health concern, ceftriaxone-resistant salmonellae have not yet been reported in Taiwan (4,20,21). We conducted a retrospective survey of clinical Salmonella isolates collected over a 4-year period in a teaching hospital to investigate whether ceftriaxone-resistant salmonellae have emerged in Taiwan. Two ceftriaxone-resistant strains producing a CMY-2–like class C cephalosporinase were found. We also established a connection between the appearance of the resistant strains and the rapid spread of the blaCMY-2-like gene in this area.
Bacterial Isolates and Patients
From January 1997 to December 2000, a total of 384 Salmonella isolates from 324 patients were collected at the National Cheng Kung University Hospital, a tertiary-care referral center with 900 beds in southern Taiwan. According to the criteria of the National Committee for Clinical Laboratory Standards (NCCLS) for the disk diffusion method, we selected for further investigation the isolates that exhibited resistance or intermediate resistance to cefpodoxime, ceftazidime, aztreonam, cefotaxime, ceftriaxone, cefoperazone, or cefixime (22). For comparison, 5,520 E. coli isolates and 3,680 K. pneumoniae isolates collected during the same period were included. Of these isolates, 1,210 nonrepetitive E. coli isolates collected from January to September 1999 were investigated previously (23). The 1997 and 1998 isolates were randomly collected and the 1999 and 2000 isolates were consecutively collected. Salmonella isolates were serotyped according to the Kauffman and White scheme (24) by using somatic and flagellar antigens (Becton Dickinson Microbiology, Cockeysville, MD).
We reviewed the medical records of patients infected with or colonized by the organisms being studied. Patients who provided samples positive for the organisms collected from any body site but who had no related signs or symptoms of infections were considered colonized. Nosocomial acquisition of infections in the teaching hospital was defined according to the 1988 definitions from the Centers for Disease Control (25). Patients transferred from other hospitals or nursing homes with infections occurring <48 h after admission were considered to have acquired the infections at the other locations. Hospitalization histories for the previous locations were recorded for outpatients and for inpatients who were colonized by the studied organisms and who provided samples within 48 h after admission.
Susceptibility Testing
The susceptibilities of isolates to antimicrobial agents were determined by using the agar dilution or disk diffusion method according to the NCCLS guidelines (22,26). The antimicrobial agents used for the agar dilution test included amoxicillin, clavulanic acid, ceftriaxone, cefoxitin, ceftazidime, cefotaxime, and imipenem. Sources of antimicrobial agents used in this study are described elsewhere (23). Breakpoints used for susceptibility meet NCCLS criteria; the ceftazidime, cefotaxime, and ceftriaxone breakpoint was 8 µg/mL (26).
Isoelectric Focusing
Crude β-lactamase extracts were prepared by using sonication (27) as described previously (23). We performed isoelectric focusing by the method of Matthew et al. (28) with an LKB Multiphor apparatus on prepared PAGplate gels (pH 3.5 to 9.5; Amersham Pharmacia Biotech, Hong Kong, China). Enzyme activities of β-lactamases were detected by overlaying the gel with 0.5 mM nitrocefin in 0.1 M phosphate buffer, pH 7.0. We used TEM-1, TEM-10, SHV-1, SHV-5, CMY-1, CTX-M-3, and CMY-2 β-lactamases as standards (18,23).
Conjugation Experiments and Plasmid Analysis
Conjugation experiments were performed by the liquid mating-out assay as described (29) with streptomycin-resistant E. coli C600 as the recipient (30). Transconjugants were selected on tryptic soy agar plates supplemented by 500 µg of streptomycin and 10 µg of ceftazidime per milliliter. Plasmids from transconjugants were extracted by using a rapid alkaline lysis procedure (31). We analyzed restriction fragment length polymorphism of transferred plasmids using agarose gel electrophoresis of plasmid DNA samples treated with the restriction endonuclease EcoRI (Roche Molecular Biochemicals, Mannheim, Germany). The restricted plasmid DNA samples were then transferred to a nylon membrane (Amersham Pharmacia Biotech) and subjected to Southern hybridization. The plasmid sizes of transconjugants were estimated by adding restriction fragments.
Molecular Techniques
Plasmid preparations from clinical isolates and their transconjugants were used as templates in polymerase chain reaction (PCR) assays. Genes related to blaTEM, blaSHV, blaCMY-1, blacmy-2, and blaCTX-M-3 were amplified with the oligonucleotide primers as described (23). Primers 5´-ATAAAATTCTTGAAGACGAAA-3´ and 5´-GACAGTTACCAATGCTTAATCA-3´, corresponding to nucleotides –5 to 15 and 1,074 to 1,053, respectively, of the blaTEM-1 structural gene (32), were used to amplify the entire sequences of blaTEM-related genes. Primers AmpC-1C (5´-CTGCTGCTGACAGCCTCTTT-3´) and AmpC-1B (5´-TTTTCAAGAATGCGCCAGGC-3´) (23), which correspond to nucleotides 28–47 and 1,136–1,117, respectively, of the blaCMY-2 structural gene (33), were used to amplify an internal fragment of approximately 95% of blaCMY-2–related genes. Both strands of the amplified products were sequenced on an ABI PRISM 310 automated sequencer (Applied Biosystems, Foster City, CA). Colony hybridization and Southern hybridization were performed as described (34,35) with DNA probes prepared from the PCR-generated amplicons. The probes were labeled with [α-32P]dCTP (Amersham Pharmacia Biotech) by using the random priming technique with a commercial kit (GibcoBRL Life Technologies, Gaithersburg, MD).
The genetic relatedness of ceftriaxone-resistant Salmonella isolates was investigated by ribotyping by using the method described by Popovic et al. (36). The chromosomal DNA was extracted and digested overnight with 10 U of SphI and PstI or EcoRI (Roche Molecular Biochemicals) (36,37). A cDNA probe was prepared by reverse transcription of 16S plus 23S rRNA (Roche Molecular Biochemicals) and was labeled with [α-32P]dCTP. DNA molecular marker II (Roche Molecular Biochemicals) and 1-kb molecular marker (Promega Corp., Madison, WI) were used as size standards.
Emergence of Salmonella Isolates Producing CMY-2–Like Enzymes
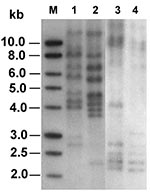
Six of 384 Salmonella isolates displayed intermediate resistance to ceftriaxone by the disk diffusion method. The isolates were recovered from stool samples of two patients who had community-acquired enteric infections in August and November 2000, respectively, which were identified as S. enterica serovar Typhimurium. One isolate from each patient (isolates ST275/00 and ST595/00) was investigated further. Both isolates demonstrated reduced susceptibilities to ceftazidime (MIC 64 µg/mL), cefotaxime (MIC 16 µg/mL), ceftriaxone (MIC 32 µg/mL), cefoxitin (MIC 128 µg/mL), and amoxicillin-clavulanic acid (MIC 64 µg/mL) and were susceptible to imipenem (MIC 0.25 µg/mL). Both isolates were susceptible to ciprofloxacin and trimethoprim-sulfamethoxazole by the disk diffusion method. Isolate ST275/00 was resistant to chloramphenicol, while isolate ST595/00 was susceptible. Isoelectric focusing showed that both isolates expressed two β-lactamases focusing at pIs 5.4 and 9.0, suggesting that they produced a TEM-1–like enzyme and an AmpC-like β-lactamase. Both isolates yielded an approximately 1.1-kb DNA fragment in PCR with primers for blaCMY-2–like genes, and the amplified sequences obtained by nucleotide sequencing were identical to the homologous region of blaCMY-2, which encodes a class C extended-spectrum cephalosporinase (33). A narrow-spectrum β-lactamase gene, blaTEM-1 (18) was also detected by PCR and nucleotide sequencing in both isolates. The two ceftriaxone-resistant Salmonella isolates had different ribotypes, suggesting that they are of different clones (Figure 1).
Prevalence of the blaCMY-2–Like Gene in E. coli and K. pneumoniae Isolates
We found that 659 of 5,520 E. coli and 409 of 3,680 K. pneumoniae isolates showed resistance to at least one extended-spectrum cephalosporin by the disk diffusion test. These isolates were subjected to colony hybridization and PCR assays. Of these isolates, 97 E. coli isolates from 48 patients and 4 K. pneumoniae isolates from 2 patients gave a strong signal in colony hybridization with the blaCMY-2 probe and gave positive results in PCR with the primers for blaCMY-2. Sequence analysis indicated that the sequences of all amplicons were identical to the homologous region of blaCMY-2. The prevalence rates of CMY-2–like enzyme producers in E. coli increased from 0% in 1997 and 1998 to >4% in late 2000 (Figure 2). Among the patients with E. coli isolates, the incidence of patients with blaCMY-positive isolates increased from 0.0% in 1997 and 1998 to 3.6% in late 2000 (Figure 2).
The sources of E. coli isolates harboring a blaCMY-2–like gene and the infections associated with these isolates are summarized in the Table. The isolates and patients are distributed according to the likely locations of infection or colonization. Of the 97 blaCMY-positive E. coli isolates, 8 isolates were likely acquired in the community by eight patients. Six of these patients had never been hospitalized, and two had been hospitalized 10 months or 2 years before isolation. K. pneumoniae isolates producing the CMY-2–like enzyme were associated with nosocomial bloodstream infections in two patients. Notably, E. coli (EC811/00) and K. pneumoniae (KP218/00) isolates were recovered from the blood sample of a single patient.
Conjugation Experiments and Plasmid Analysis
One isolate from each patient was subjected to conjugation experiments and plasmid analysis. The blaCMY-positive plasmids were successfully transferred to E. coli C600 from 2 Salmonella isolates, 40 of 48 E. coli isolates, and 2 K. pneumoniae isolates. All E. coli transconjugants and their plasmid donors showed decreased susceptibilities to ceftazidime (MIC >32 µg/mL), cefotaxime (MIC >16 µg/mL), ceftriaxone (MIC >32 µg/mL), and cefoxitin (MIC >64 µg/mL). The sizes of the transferred plasmids ranged from approximately 65 kb to >100 kb.
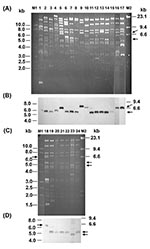
Restricted by EcoRI, the plasmids from transconjugants of E. coli isolates showed 19 restriction patterns, designated TP1–TP19 (Figures 3A and 3C). Patterns TP17 (lane 17) and TP19 (lane 19), the most common patterns, were shown by 6 and 17 transferred plasmids, respectively. Of the 17 isolates with a TP19 plasmid, 3 were acquired from nursing homes and 5 from the community. Of the six isolates with a TP17 plasmid, two could have been acquired from the community. We considered that the remaining isolates with a TP17 or TP19 plasmid were acquired in the university hospital. Each of the remaining 17 patterns was shown by a single transferred plasmid. The TP19 pattern is the only restriction pattern displayed by transconjugants of all three studied bacterial species: Salmonella (lane 21), K. pneumoniae (lane 22), and E. coli (lane 24). Notably, K. pneumoniae isolate KP218/00 and E. coli isolate EC811/00, from the same patient, both had a TP19-type plasmid. The plasmids from transconjugants of Salmonella isolate ST275/00 (lane 20) and another K. pneumoniae isolate KP1905/00 (lane 21) showed two distinct restriction patterns. The presence of the blaCMY-2–like gene on plasmids was confirmed by Southern hybridization with the blaCMY-2 probe (Figures 3B and 3D).
Emergence of ceftriaxone-resistant salmonellae has become a great public health concern worldwide (5–17). In Taiwan, no ceftriaxone-resistant isolates were detected from several surveys, which included isolates collected from 1989 to 1998 (4,20,21). One of the surveys was conducted in National Cheng Kung University Hospital (4), and one survey conducted in 1998 included isolates from 22 hospitals (21). In the present study, Salmonella isolates resistant to extended-spectrum β-lactams were not detected until August 2000. Thus, the appearance of ceftriaxone-resistant Salmonella strains is likely a recent event.
Production of a CMY-2–like β-lactamase was responsible for resistance to extended-spectrum β-lactams in the Salmonella strains we isolated. The spread of blaCMY-2 in salmonellae recently was reported to be an emerging problem in the United States (14,15). In Taiwan, the blaCMY-2–like gene was first detected in E. coli isolates collected in 1999 in the university hospital (23). The fact that the blaCMY-2–like gene was found in few isolates from patients with community-acquired infections suggested that the genetic determinant had spread in the community environment (23). The appearance of the blaCMY-2–like gene in the Salmonella isolates supports our previous speculation on the spread of the genetic determinant in the community environment. Moreover, the discovery of patients who might have acquired E. coli isolates with the CMY-2–like enzyme from nursing homes and in the community (Table) suggests widespread distribution of the blaCMY-2–like gene in southern Taiwan.
Conjugation experiments and plasmid analysis demonstrated the prevalence of conjugative resistance plasmids TP17 and TP19 among E. coli isolates. These E. coli isolates were recovered from patients hospitalized in the teaching hospital, transferred from nursing homes, or without recent hospitalization histories (Figure 3). Furthermore, the TP19 plasmid was found in a Salmonella strain and a K. pneumoniae strain, which suggests the interspecies spread of the blaCMY-2–like gene among different health-care settings and the community in Taiwan. The dissemination of the resistance determinant is probably partly because of horizontal transfer of endemic resistance plasmids.
All K. pneumoniae isolates producing the CMY-2–like enzyme were acquired by patients hospitalized in the university hospital. Producers of the CMY-2–like enzyme in K. pneumoniae were not found until 2000 and, even then, remained very rare (38). Isolate KP218/00 (lane 22, Figure 3C) and E. coli isolate EC811/00 (lane 24, Figure 3C) were obtained from the same patient, and both isolates had a TP19 plasmid, suggesting interspecies spread of a resistance plasmid. Thus, the acquisition of the blaCMY-2 gene by K. pneumoniae in this hospital was likely also a recent event, which occurred after spread of the resistance determinant.
Enterobacteriaceae with plasmid-encoded class C cephalosporinases are typically resistant to cephamycins, extended-spectrum cephalosporins, and monobactams (19). No standard methods exist to detect class C cephalosporinases (39). Failure to detect and report CMY-2–like enzyme producers, and a lack of infection control measures against such organisms might be partially responsible for the rapid spread of the blaCMY-2–like gene in E. coli and the increased cases of infections caused by such organisms in our hospital (Table). With the discovery of Salmonella isolates producing the CMY-2–like enzyme in Taiwan, our study suggests that the cephalosporinase could become an emerging threat, not only to hospitalized patients, but also to public health. Constant and consistent surveillance is needed to prevent its further spread.
Dr. Yan is an assistant professor, Department of Pathology, National Chen Kung University Medical College, Tainan, Taiwan. His major research interests are epidemiology and mechanisms of antibiotic resistance, especially beta-lactamases in gram-negative bacteria.
Acknowledgment
The National Science Council, Taiwan (grant NSC 91-2314-B-006-002) and the Centers for Disease Control and Prevention, the Department of Health, and the Executive Yuan, Taiwan (grant DOH 91-DC1033) provided financial support for this research.
References
- Lee LA, Puhr ND, Maloney EK, Bean NH, Tauxe RV. Increase in antimicrobial-resistant Salmonella infections in the United States, 1989–1990. J Infect Dis. 1994;170:128–34.PubMedGoogle Scholar
- Threlfall EJ, Ward LR, Skinner JA, Rowe B. Increase in multiple antibiotic resistance in nontyphoidal salmonellas from humans in England and Wales: a comparison of data for 1994 and 1996. Microb Drug Resist. 1997;3:263–6. DOIPubMedGoogle Scholar
- Reina J, Gomez J, Serra A, Borell N. Analysis of the antibiotic resistance detected in 2,043 strains of Salmonella enterica subsp. enterica isolated in stool cultures of Spanish patients with acute diarrhea (1986–1991). J Antimicrob Chemother. 1993;32:765–9. DOIPubMedGoogle Scholar
- Yang YJ, Liu CC, Wang SM, Wu JJ, Huang AH, Cheng CP. High rates of antimicrobial resistance among clinical isolates of nontyphoidal Salmonella in Taiwan. Eur J Clin Microbiol Infect Dis. 1998;17:880–3. DOIPubMedGoogle Scholar
- Arachambaud M, Gerbaud G, Labau E, Marty N, Courvalin P. Possible in-vivo transfer of β-lactamase TEM-3 from Klebsiella pneumoniae to Salmonella kedougou. J Antimicrob Chemother. 1991;27:427–36. DOIPubMedGoogle Scholar
- Poupart M-C, Chanal C, Sirot D, Labia R, Sirot J. Identification of CTX-2, a novel cefotaximase from a Salmonella mbandaka isolate. Antimicrob Agents Chemother. 1991;35:1498–500.PubMedGoogle Scholar
- Hammami A, Arlet G, Ben Redjeb S, Grimont F, Ben Hassen A, Rekik A, Nosocomial outbreak of acute gastroenteritis in a neonatal intensive care unit in Tunisia caused by multiply drug resistant Salmonella wien producing SHV-2 β-lactamase. Eur J Clin Microbiol Infect Dis. 1991;10:641–6. DOIPubMedGoogle Scholar
- Bauernfeind A, Casellas JM, Goldberg M, Holley M, Jungwirth R, Mangold P, A new plasmidic cefotaximase from patients infected with Salmonella typhimurium. Infection. 1992;20:158–63. DOIPubMedGoogle Scholar
- Vahaboglu H, Hall LMC, Mulazimoglu L, Dodanli S, Yildirim I, Livermore DM. Resistance to extended-spectrum cephalosporins, caused by PER-1, β-lactamase, in Salmonella typhimurium from Istanbul, Turkey. J Med Microbiol. 1995;43:294–9. DOIPubMedGoogle Scholar
- Morosini MI, Blázquez J, Negri MC, Cantón R, Loza E, Baquero F. Characterization of a nosocomial outbreak involving an epidemic plasmid encoding for TEM-27 in Salmonella enterica subspecies enterica serotype Othmarschen. J Infect Dis. 1996;174:1015–20.PubMedGoogle Scholar
- Koeck JL, Arlet G, Philippon A, Basmaciogullari S, Thien HV, Buisson Y, A plasmid-mediated CMY-2 β-lactamase from an Algerian clinical isolate of Salmonella senftenberg. FEMS Microbiol Lett. 1997;152:255–60.PubMedGoogle Scholar
- Bradford PA, Yang Y, Sahm D, Grope I, Gardovska D, Storch G. CTX-M-5, a novel cefotaxime-hydrolyzing β-lactamase from an outbreak of Salmonella typhimurium in Lativia. Antimicrob Agents Chemother. 1998;42:1980–4.PubMedGoogle Scholar
- Barnaud G, Arlet G, Verdet C, Gaillot O, Lagrange PH, Philippon A. Salmonella enteritidis: AmpC plasmid-mediated inducible β-lactamase (DHA-1) with an ampR gene from Morganella morganii. Antimicrob Agents Chemother. 1998;42:2352–8.PubMedGoogle Scholar
- Fey PD, Safranek TJ, Rupp ME, Dunne EF, Ribot E, Iwen PC, Ceftriaxone-resistant Salmonella infection acquired by a child from cattle. N Engl J Med. 2000;342:1242–9. DOIPubMedGoogle Scholar
- Winokur PL, Brueggemann A, Desalvo DL, Hoffmann L, Apley MD, Uhlenhopp EK, Animal and human multidrug-resistant, cephalosporin-resistant Salmonella isolates expressing a plasmid-mediated CMY-2 AmpC β-lactamase. Antimicrob Agents Chemother. 2000;44:2777–83. DOIPubMedGoogle Scholar
- Vahaboglu H, Fuzi M, Cetin S, Gundes S, Ujhelyi E, Coskunkan F, Characterization of extended-spectrum β-lactamase (TEM-52)-producing strains of Salmonella enterica serovar Typhimurium with diverse resistance phenotypes. J Clin Microbiol. 2001;39:791–3. DOIPubMedGoogle Scholar
- Hanson ND, Moland ES, Hossain A, Neville SA, Gosbell IB, Thomson KS. Unusual Salmonella enterica serotype Typhimurium isolate producing CMY-7, SHV-9 and OXA-10 β-lactamases. J Antimicrob Chemother. 2002;49:1011–4. DOIPubMedGoogle Scholar
- Bush K, Jacoby GA, Medeiros AA. A functional classification scheme for β-lactamases and its correlation with molecular structure. Antimicrob Agents Chemother. 1995;39:1211–33.PubMedGoogle Scholar
- Livermore DM. β-Lactamase-mediated resistance and opportunities for its control. J Antimicrob Chemother. 1998;41:25–41. DOIPubMedGoogle Scholar
- Chen YH, Chen TP, Tsai JJ, Hwang KP, Lu PL, Cheng HH, Epidemiological study of human salmonellosis during 1991–1996 in southern Taiwan. Kaohsiung J Med Sci. 1999;15:127–36.PubMedGoogle Scholar
- Ho M, McDonald C, Lauderdale TL, Yeh LLL, Chen PC, Shiau YR, Surveillance of antibiotic resistance in Taiwan, 1998. J Microbiol Immunol Infect. 1999;32:239–49.PubMedGoogle Scholar
- National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial disk susceptibility tests, 7th ed. Approved standard M2-A7. National Committee for Clinical Laboratory Standards. Wayne (PA): The Committee; 2000.
- Yan JJ, Ko WC, Tsai SH, Wu HM, Jin YT, Wu JJ. Dissemination of CTX-M-3 and CMY-2 β-lactamases among clinical isolates of Escherichia coli in southern Taiwan. J Clin Microbiol. 2000;38:4320–5.PubMedGoogle Scholar
- Brenner FW, Villar RG, Angulo FJ, Tauxe RV, Swaminathan B. Salmonella nomenclature. J Clin Microbiol. 2000;38:2465–7.PubMedGoogle Scholar
- Garner JS, Jarvis WR, Emori TG, Horau TC, Hughes JM. CDC definitions for nosocomial infections, 1988. Am J Infect Control. 1988;16:128–40. DOIPubMedGoogle Scholar
- National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 5th ed. Approved standard M7-A5. National Committee for Clinical Laboratory Standards. Wayne (PA): The Committee; 2000.
- Bauernfeind A, Grimm H, Schweighart S. A new plasmidic cefotaximase in a clinical isolate of Escherichia coli. Infection. 1990;18:294–8. DOIPubMedGoogle Scholar
- Matthew M, Harris M, Marshall MJ, Rose GW. The use of analytical isoelectric focusing for detection and identification of β-lactamases. J Gen Microbiol. 1975;88:169–78.PubMedGoogle Scholar
- Provence DL, Curtiss R III. Gene transfer in gram-negative bacteria. In: Gerhardt P, Murray RGE, Wood WA, Krieg NR, editors. Methods for general and molecular bacteriology. Washington: American Society for Microbiology; 1994. p. 319–47.
- Bachmann BJ, Low KB. Linkage map of Escherichia coli K-12, edition 6. Microbiol Rev. 1980;44:1–56.PubMedGoogle Scholar
- Takahashi S, Nagano Y. Rapid procedure for isolation of plasmid DNA and application to epidemiological analysis. J Clin Microbiol. 1984;20:608–13.PubMedGoogle Scholar
- Mabilat C, Goussard S. PCR detection and identification of genes for extended-spectrum β-lactamases. In: Persing DH, Smith TF, Tenover FC, White TJ, editors. Diagnostic molecular microbiology: principles and applications. Washington: American Society for Microbiology; 1993. p. 553–9.
- Bauernfeind A, Stemplinger I, Jungwirth R. Giamarellou. Characterization of the plasmidic β-lactamase CMY-2, which is responsible for cephamycin resistance. Antimicrob Agents Chemother. 1996;40:221–4.PubMedGoogle Scholar
- Grunstein M, Hogness DS. Colony hybridization: a method for the isolation of cloned DNAs that contain a specific gene. Proc Natl Acad Sci U S A. 1975;72:3961–5. DOIPubMedGoogle Scholar
- Southern EM. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975;98:503–17. DOIPubMedGoogle Scholar
- Popovic T, Bopp CA, Olsvik Ø, Kiehlbauch JA. Ribotyping in molecular epidemiology. In: Persing DH, Smith TF, Tenover FC, White TJ, editors. Diagnostic molecular microbiology: principles and applications. Washington: American Society for Microbiology; 1993. p. 573–89.
- Liebana E, Garcia-Migura L, Breslin MF, Davies RH, Woodward MJ. Diversity of strains of Salmonella enterica serotype Enteritidis from English poultry farms assessed by multiple genetic fingerprinting. J Clin Microbiol. 2001;39:154–61. DOIPubMedGoogle Scholar
- Yan JJ, Wu SM, Tsai SH, Wu JJ, Su IJ. Prevalence of SHV-12 among clinical isolates of Klebsiella pneumoniae producing extended-spectrum β-lactamase and identification of a novel AmpC Enzyme (CMY-8) in southern Taiwan. Antimicrob Agents Chemother. 2000;44:1438–42. DOIPubMedGoogle Scholar
- Thomson KS. Controversies about extended-spectrum and AmpC beta-lactamases. Emerg Infect Dis. 2001;7:333–6. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 9, Number 3—March 2003
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Jiunn-Jong Wu, Department of Medical Technology, College of Medicine, National Cheng Kung University, No. 1 University Road, Tainan, Taiwan; fax: 886-6-2363956
Top