Volume 10, Number 5—May 2004
Research
Acute Tick-borne Rickettsiosis Caused by Rickettsia heilongjiangensis in the Russian Far East
Cite This Article
Citation for Media
Abstract
An acute tick-borne rickettsiosis caused by Rickettsia heilongjiangensis was diagnosed in 13 patients from the Russian Far East in 2002. We amplified and sequenced four portions of three rickettsial genes from the patients' skin biopsy results and blood samples and showed that the amplified rickettsial genes belong to R. heilongjiangensis, which was recently isolated from Dermacentor sylvarum ticks in nearby regions of China. This rickettsia, belonging to subgroup of R. japonica, was previously suggested to be pathogenic for humans on the basis of serologic findings. We tested serum samples with different rickettsial antigens from 11 patients and confirmed increasing titers of immunoglobulin (Ig) G and IgM to spotted fever group rickettsiae, including R. heilongjiangensis. Clinical and epidemiologic data on these patients show that this disease is similar to other tick-borne rickettsioses.
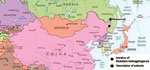
Russian Far East is a geographic, economic, and political unit within the Russian Federation. It consists of the smaller administrative areas (regions) located on or close to the Asian Pacific coast. The southern portion of Khabarovsk region, where this study was carried out, is situated alongside the Amur River down to the sea (Figure 1) and is characterized by peculiar combinations of subtropical and boreal biologic niches. Local experience suggests that tick-borne encephalitis, Siberian tick typhus, and, more recently, Lyme disease are prevalent in this territory, with marked seasonal disease peaks (1). In 2002, serologic evidence for acute granulocytic ehrlichiosis was found in the region (2).
In 1932, a disease described as tick-borne fever (tick typhus) was identified in the Russian Far East (3). In Central Siberia in 1935, the agent of tick-borne fever, Rickettsia sibirica, was isolated and described (4–6), and several rickettsial isolates from the ticks from the Far Eastern Russia have also been identified as this new species (7). Since 1935, the cause of acute tick-borne spotted fever in the Russian Far East (including Khabarovsk Region) is thought to be R. sibirica, and the antigen of R. sibirica has been used for serologic studies in clinical laboratories. Nevertheless, differences between clinical pictures of tick-borne fever and differences in serologic cross-reactivity of sera from patients with tick-borne rickettsiosis have been reported in Siberia and the Russian Far East (8). Dermacentor sylvarum ticks have been identified as a vector. In 1990s, several strains of R. sibirica were also isolated from ticks in Primorye Region, south of Khabarovsk Region (9).
In Siberia and China, several new rickettsiae were found. R. mongolotimonae found in Inner Mongolia is closely related to R. sibirica (10) and causes acute disease in humans. Clinical cases have now been described in France (11,12). R. heilongjiangensis (strain 054) was first isolated from D. sylvarum ticks collected around Suifenhe in the Heilongjiang Province of China in 1982 (13). Serologic studies indicated that the organism was the probable cause of an acute tick-borne disease in humans (14). R. hulinensis (strain HL-93) was isolated in 1993 from Haemaphysalis concinna ticks collected in Hulin County, Heilongjiang Province (10). The pathogenicity of this agent in humans is unknown. By using molecular evolutionary genetic analyses, these rickettsiae were shown to form a well-defined clade distinct from other spotted fever group rickettsiae, including R. japonica (15). Both D. sylvarum and H. concinna ticks inhabit the Russian Far East, although D. sylvarum is quite rare (1). In this study, we identify the cause of an acute, febrile, tick-transmitted disease in the Russian Far East and describe the clinical picture in these cases.
Sample Collection
During this study in 2002, we screened almost all patients who were suspected clinically to have a tick-borne rickettsiosis and who were hospitalized in the infectious diseases department of Khabarovsk Municipal Hospital No.10. Five to 20 days before onset of the disease, most patients reported tick bites or tick exposure. Some had typical eschars on the skin without a history of a tick bite. The illness was acute, febrile, and usually involved a macular or maculopapular rash. All patients were successfully treated with a 14-day course of doxycycline. Patients with clinically evident features of tick-borne encephalitis (neurologic signs) or Lyme disease (erythema migrans) were excluded from the study. In total, samples from 65 patients were investigated, 64 blood samples and 17 skin biopsy results. Paired sera from most of the patients were tested with conventional serologic methods. The first serum sample was taken immediately after admission to the hospital, usually 1–9 days after the onset of the disease. The second serum sample was taken 4–18 days later.
DNA Extraction, PCR, and Sequencing
Buffy coats were separated from fresh blood specimens immediately after collection at patient's admission. DNA was extracted from the buffy coat and skin biopsies samples with QIAmp DNA Blood Mini Kit and DNeasy Tissue Kit (Qiagen, Tokyo, Japan) according to manufacturer's instructions.
Oligonucleotide primers used in this study and annealing temperatures are shown in Table 1. We used the nested polymerase chain reaction (PCR) method for primary detection of the rickettsial DNA in human samples. The gltA (citrate synthase) gene was chosen as the target for amplification because of its genus specificity and conservativeness. The primer CS1d (16), which is used traditionally to amplify the full-length gltA gene, is complementary to the ultimate 5´-end of the open reading frame for this gene of R. prowazekii. Since we expected to find DNA of the spotted fever group rickettsiae, we modified the primer to be completely homologous to the corresponding portion of the gene of R. conorii (Table 1). R. conorii has been chosen as a template to design primer CS2d as the only spotted fever group rickettsia having a completely sequenced genome at the time of investigation. Comparison of these two primers indicated that the newly designed primer (CS2d) was at least 100 times more sensitive than CS1d in the amplification of the serially diluted DNA of R. montanensis, which was used as a positive control (data not shown). Primers CS2d and CSendR amplify the full-length of the gltA gene. Primers CS877f and CS1258r were used in the nested PCR assay. To amplify the full-length of the gltA gene, we used PCR followed by hemi-nested PCR using the product of the first PCR as a template. Two conservative regions of the outer membrane protein A (ompA) gene were amplified (base positions 91–680 and 3,608–6,789 with respect to the sequence published for R. rickettsii) by using previously described primers (17). Initial amplification of the 3,182-bp longer region was achieved with primers 190-3588f and 190-5044r and with primers 190-4859f and 190-6808r. Products of this reaction were used later in the nested and hemi-nested PCR reactions with corresponding primers. The ompB gene was amplified from clinical samples and DNA by using standard primers (18), except for the ultimate forward primer 120-M59, which was found to produce nonspecific amplicons with the human DNA in clinical samples. Instead, for clinical specimens, we used a newly designed primer, 120-M36. As with the ompA, the DNA from clinical samples was amplified in two steps. In the first step, we amplified two halves of the gene, with a small overlapping region with primers 120-M36 with 120-2988 and 120-2788 with 120-4879. The products of these reactions were used as templates for each of the specific nested or heminested reactions for seven regions. All primers were purchased from Eurogentec (4102 Seraing, Belgium).
Amplification has been carried out by routine methods in a final volume of 50 µL with appropriate negative controls (10,17). In all reactions, as a positive control, we used a mixture of DNA of the following microorganisms: Anaplasma phagocytophilum, Neorickettsia sennetsu, Wolbachia pipientis, Francisella tularensis, Bartonella henselae, Borrelia garinii, Coxiella burnetii, and R. montanensis. We found that the cocktail of DNA of tick-borne or phylogenetically close to tick-borne bacteria is convenient to use in PCR with potentially polyinfected human samples.
We checked all samples for other possible tick-borne bacterial and pathogens with the following primers under conditions suggested in published references: SL primers for ospA gene of Borrelia (19); BhCS.781p and BhCS.1137n primers for Bartonella citrate synthase (20); HE1, HE3, and HE4 primers for 16S rDNA of Ehrlichia chaffeensis (21); GE3A, GE10r, GE9f, and GE2 primers for 16S rRNA gene of A. phagocytophilum (22); and P3708 and p4257 primers for the gene encoding p44 protein of A. phagocytophilum (23) [data not shown]. No positive results have been obtained among patients described here.
The PCR products were purified for DNA sequencing with the QIAquick PCR purification kit (Qiagen) and then directly sequenced by using PCR primers. Sequencing reactions were carried out with a D-rhodamine terminator cycle DNA sequencing kit (Applied Biosystems, Foster City, CA) according to the manufacturer's instructions. Sequencing reaction products were resolved by electrophoresis with an ABI Prism 377 sequencer (Applied Biosystems). The results obtained were processed into sequence data with AutoAssembler software (Applied Biosystems). The sequences of the gltA, both regions of the ompA gene, and the ompB genes were aligned by using the software Genetix-Win 5.1 (Software Development Co., Ltd., Japan). Sequences in the ompB genes of R. heilongjiangensis and R. hulinensis were not available in the GenBank database so we amplified and sequenced the gene. The sequences used for comparison were obtained from the GenBank database, aligned, and then corrected manually to preserve codon alignment and conserved motifs. Sites with ambiguous alignments were removed before phylogenetic analysis. The phylogenetic tree was calculated by using neighbor-joining method with MEGA2 Version 2.1 software (available from http://megasoftware.net). Internal node support was verified by using the bootstrap method with 100 replicates.
Serologic Studies
Two serologic tests were performed in Khabarovsk Plague Control Station: immunofluorescence studies with a combined antigen consisted of two local strains of B. garinii and one local strain of B. afzelii, and an enzyme-linked immunosorbent assay detected antibodies against tick-borne encephalitis virus (24). For further investigations, sera were dried on blotting papers as described previously (25) and transported to Marseilles, where microimmunofluorescence testing (26) was performed by using in-house prepared antigens of R. heilongjiangensis (strain 054, ATCC VR-1524), R. hulinensis (strain HL-93, ATCC VR-1527), R. sibirica (strain 246, ATCC VR-151), R. conorii (strain Moroccan ATCC VR-141), C. burnetii (strain Nine Mile, ATCC VR-615), Orientia tsutsugamushi (strains Gilliam, Karp, Kato, and Kawazaki), E. chaffeensis (strain Arkansas), A.phagocytophilum (strain Webster), Bartonella henselae (Houston-1, ATCC-49882), and B. quintana (strain Oklahoma). Antigens were applied by pen point to 18-well microscope slides, dried for 30 min, and fixed. Appropriate positive- and negative-control serum samples were tested on each slide together with twofold dilutions of patients' serum samples made in 3% nonfat dry milk in phosphate-buffered saline (PBS). Slides were incubated in a moist chamber for 30 min at 37°C, washed twice in PBS and once in distilled water (10 min each); reactive antibodies showed fluorescein isothiocyanate–conjugated goat anti-human γ chain and µ-chain immunoglobulins (BioMérieux, Marcy l'Etoile, France). After the conjugate was added, slides were incubated for 30 min at 37°C, washed in two PBS for 10 min and for 5 min in distilled water, and mounted in buffered glycerol. Endpoints for each antigen were the lowest concentrations of serum that definitely conferred fluorescence on bacteria.
Nucleotides Accession Numbers
Nucleotide sequences obtained during this study were deposited in GenBank under the following numbers: AY260451 for ompB gene of R. heilongjiangensis, strain 054; AY260452 for ompB gene for R. hulinensis; AY280712 for ompB gene; AY280711 for previously tandemly repeated region portion of ompA gene; AY280710 for another portion; and AY280709 for gltA gene of Rickettsia heilongjiangensis found in this study.
We amplified and sequenced DNA of R. heilongjiangensis in samples from 16 patients. Serum samples from 11 were available for serologic studies, and clinical and epidemiologic data have been analyzed for 13 patients, including all patients with investigated serum samples.
Ten of 17 samples of DNA extracted from skin eschars and seven of 64 samples of DNA extracted from buffy coats were positive in the nested PCR for the gltA gene. In one patient, both the skin biopsy and the buffy coat were positive and had the same DNA sequence. Because we had limited amounts of extracted DNA, we attempted to amplify both the ompA and ompB genes from six samples (three skin biopsies and three blood samples), which were previously positive in nested PCR with primers for citrate synthase gene. All positive samples were also screened by PCR for other possible bacterial tick-borne pathogens and were found to be negative. Results from testing, serologic or PCR, that suggested double infection were excluded from the study. Clinical picture of the disease was analyzed in patients with R. heilongjiangensis infection to describe the disease associated with this organism.
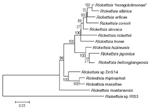
All 17 nested PCR amplicons of amplified gltA gene were directly sequenced and showed 100% homology. Six amplicons of ompA and ompB genes of corresponding samples were also identical with each other. We obtained full-length gltA gene sequence, 590 bp and 3,182 bp (excluding primer sequences) of 5´- and 3´-regions of the ompA gene, respectively, and a 4,852-bp length sequence of the ompB gene. A BLAST search showed that all sequences were completely homologous to correspondent genes of R.heilongjiangensis. Figure 2 shows the phylogenetic relationships of this Rickettsia and other species based on the analysis of both concatenated portions of the ompA gene.
Serum samples from 11 of 16 patients were studied; data are presented in Tables 2 and 3. None of the samples had serologic evidence of any other nonrickettsial acute, tick-borne disease. One previously vaccinated patient had a low titer of immunoglobulin (Ig) G against tick-borne encephalitis virus, and two had low titers to both B. henselae and B. quintana (data not shown). In 9 of 11 available paired sera from PCR-positive patients, we found serologic evidence for acute rickettsial infection, e.g., the presence of IgM antibodies, seroconversion, or a fourfold rise in antibody titer against rickettsial antigens. In samples from two patients, IgG and IgM antibody titers to R. heilongjiangensis were highest. In samples from five patients, the same titers were present against R. conorii and R. heilongjiangensis. In samples from one patient, we found the same levels of antibodies to all rickettsial antigens. Levels of antibodies against R. sibirica in all cases, except one, were lower than against R. heilongjiangensis. In four of nine cases, titers were identical against antigens of R. hulinensis and R. sibirica. Absence of specific antibodies against rickettsiae in patients 2 and 3 could be explained either by early treatment with a specific antimicrobial drug or probable errors in serum sample collection and treatment.
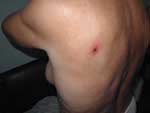
Epidemiologic, clinical, and laboratory data available in 13 of 16 PCR-positive patients are given in Table 4. Before the onset of the disease in the summer 2002 (from June to August), all patients had a history of tick bite, tick exposure, or a stay in an epidemiologically suspected location. After an incubation period of 4 to 7 days, the patient had a sudden onset with fever, but no specific symptoms appeared during the first several days. In 12 patients, a macular or maculopapular rash appeared but was usually faint. Twelve patients had a primary lesion (eschar) at the site of tick attachment (Figure 3). The eschar consisted of a necrotic central region (50–150 mm in diameter) surrounded by infiltrated and inflamed tissue (70–400 mm in diameter), and a zone of hyperemia (250–500 mm in diameter). The eschars were found on the waist and buttocks region (four); lower extremities (two); upper extremities and axillar region (two); and back, chest, neck, and abdomen (one case each). In two patients, we noticed subcutaneous lymphangitis and regional lymphadenopathy. Initial conventional treatment at home, with a combination of antipyretics, analgesics, and antibacterial therapy (oral penicillins) did not result in improvement; 5–9 days after the onset of symptoms, all patients were admitted to the hospital. They received oral doxycycline for 14 days and antihistamine therapy, and clinical symptoms resolved within 2 to 3 days. Laboratory tests in the hospital showed elevated levels of serum transaminases (alanine aminotransferase and aspartate aminotransferase) in 46% and 15% of patients, respectively, and these remained elevated even during convalescence but were normal at follow-up at 3 to 4 weeks.
Our findings suggest that patients had an acute infection with R. heilongjiangensis. Clinical data were typical for acute rickettsial infections. We successfully amplified four portions of three different rickettsial genes from human blood and skin samples, which suggested the presence of this microorganism. We did not amplify any of these genes in samples from healthy donors or from patients suffering from other infectious diseases (negative controls). The gltA gene is conservative, especially among Rickettsia species (16), and ompA and ompB genes encode main surface proteins of rickettsiae. These gene sequences were completely homologous to the spotted fever group R. heilongjiangensis. Previous GenBank sequences were recently corrected (Fournier PE et al., unpub. data). The identity of sequenced PCR products among patients, clinical data, and epidemiologic data suggest the focality of this rickettsiosis.
The serologic data support PCR and sequencing findings. We showed that Rickettsia produces a clear immunologic response in patients. We studied paired sera from patients for reactivity with antigens of several species in the Rickettsia genus, including R. heilongjiangensis and R. hulinensis. Seroconversion, presence of IgM, or increasing antibody titers were observed in most patients. In seven cases, sera reacted at higher titers with antigen of R. heilongjiangensis when compared with R. sibirica, the only currently identified tick-borne rickettsia in the Russian Far East. In 77%, the titers were identical against R. heilongjiangensis and R. conorii antigens. Although serologic cross-reactions are common among rickettsiae of the spotted fever group (26), the finding of lower titers against the phylogenetically more closely related R. hulinensis than the relatively more distant R. conorii was unexpected.
Epidemiologic evidence of tick bite or exposure, rash, primary lesion (eschar) at the site of tick bite, and rapid recovery after doxycycline treatment support a rickettsial cause for the disease. Some peculiarities were noticed when signs were compared with infection caused by R. sibirica in the Central Siberia. Seasonal peak of infections is in the end of June and July. For Siberian tick typhus, the seasonal peak is the end of April and May. The rash that accompanies tick-borne rickettsiosis in the Russian Far East is less obvious, and the disease apparently affects older people than Siberian tick typhus. Only 1 of 13 patients was >45 years of age. Generally, the disease is mild, with no serious complications or death recorded.
The epidemiology of the disease remains mostly unknown. Recently, DNA of the Rickettsia described in our report was amplified from H.concinna ticks collected in Siberia (S. Shpynov, unpub. data).
PCR-based technologies and direct sequencing provide a fast and precise diagnosis or rickettsiosis. The preferable method may be PCR on eschar biopsy samples because this technique has high sensitivity and probability of finding rickettsial DNA (27). Serologic studies of samples from Russian Far East area should include tests with antigens of R. heilongjiangensis.
Results of our studies showed that acute febrile tick-borne disease caused by R. heilongjiangensis is prevalent in the Russian Far East. Molecular biology approaches enabled us to identify the cause of an acute disease and to detect its bacterial origin. As no evidence of R. sibirica human infection was found in our study, further investigations are needed clarify its role in human pathology in the Russian Far East, especially the Khabarovsk Region.
Dr. Mediannikov is a postgraduate student in the Laboratory of Rickettsial Ecology, Gamaleya Research Institute of Epidemiology and Microbiology, Moscow, Russia. In 2002 to 2003, he spent 5 months in the Laboratory of Rickettsiology, Mediterranean University, Marseilles. His interests include the microbiology and epidemiology of tick-borne infections.
Acknowledgment
We are grateful to S. Dumler and M.M. Eremeeva for worthy advice, G.S. Tomilka and V.I. Zholondz for assistance in samples collection, and P. Kelly for thoroughly reviewing this manuscript.
References
- Yastrebov VK. Transmissible and natural infections in Siberia and the Far East. Probl Sotsialnoi Gig Istor Med. 1995;5:16–9.PubMedGoogle Scholar
- Sidelnikov YN, Mediannikov OY, Ivanov LI, Zdanovskaya NI. Clinical and laboratory features of human granulocytic ehrlichiosis in the south of Russian Far East. Epidemiologia i Infectsionnye Bolezni. 2002;3:28–31.
- Mill EI. Tick-borne fever in Primorye. Far Eastern Medical Journal. 1936;3:54–6.
- Shmatikov MD, Velik MA. Tick-borne spotted fever. Klinicheskaya medicina 1939;7:124–6.
- Korshunova OS. Etiology of tick-borne typhus in Krasnoyarsk region. Zh Mikrobiol Epidemiol Immunobiol. 1943;1–2:9–64.
- Zdrodovski PF, Galinevich EM. The Rickettsial diseases. London: Pergamon Press; 1960.
- Savitskaya EM. On etiology of tick-borne typhus in Khabarovsk region. Zh Mikrobiol Epidemiol Immunobiol. 1943;10–11:87.
- Mayevsky MM. Recent news and findings in rickettsiology. Zh Mikrobiol Epidemiol Immunobiol. 1947;4:3–14.
- Balayeva NM, Eremeeva ME, Ignatovich VE, Rudakov NV, Reshetnikova TA, Samoilenko IE, Biological and genetic characterization of Rickettsia sibirica strains isolated in the endemic area of the North Asian tick typhus. Am J Trop Med Hyg. 1996;55:685–92.PubMedGoogle Scholar
- Yu X, Jin Y, Fan M, Xu G, Liu Q, Raoult D. Genotypic and antigenic identification of two new strains of spotted fever group rickettsiae isolated from China. J Clin Microbiol. 1993;31:83–8.PubMedGoogle Scholar
- Raoult D, Brouqui P, Roux V. A new spotted-fever-group rickettsiosis. Lancet. 1996;348:412. DOIPubMedGoogle Scholar
- Fournier PE, Tissot-Dupont H, Gallais H, Raoult D. Rickettsia mongolotimonae, a rare pathogen in France. Emerg Infect Dis. 2000;6:290–2. DOIPubMedGoogle Scholar
- Fan MY, Zhang JZ, Chen M, Yu XJ. Spotted fever group rickettsioses in China. In: Raoult D, Brouqui P, editors. Rickettsiae and rickettsial diseases at the turn of the third millennium. Paris: Elsevier; 1999. p. 247–57.
- Lou D, Wu YM, Wang B, Wang W, Zhang Z, Zhang XW. Confirmation of patients with tick-borne spotted fever caused by Rickettsia heilongjiangi. Zhonghua Liu Xing Bing Xue Za Zhi. 1989;10:128–32.
- Zhang JZ, Fan Y, Wu YM, Fournier PE, Roux V, Raoult D. Genetic classification of "Rickettsia heilongjiangii" and "Rickettsia hulinii," two Chinese spotted fever group rickettsiae. J Clin Microbiol. 2000;38:3498–501.PubMedGoogle Scholar
- Roux V, Rydkina E, Eremeeva M, Raoult D. Citrate synthase gene comparison, a new tool for phylogenetic analysis, and its application for the rickettsiae. Int J Syst Bacteriol. 1997;47:252–61. DOIPubMedGoogle Scholar
- Fournier PE, Roux V, Raoult D. Phylogenetic analysis of spotted fever group rickettsiae by study of the outer surface protein rOmpA. Int J Syst Bacteriol. 1998;48:839–49. DOIPubMedGoogle Scholar
- Roux V, Raoult D. Phylogenetic analysis of members of the genus Rickettsia using the gene encoding the outer-membrane protein rOmpB (ompB). Int J Syst Evol Microbiol. 2000;50:1449–55. DOIPubMedGoogle Scholar
- Demaerschalck I, Ben Messaoud A, De Kesel M, Hoyois B, Lobet Y, Hoet P, Simultaneous presence of different Borrelia burgdorferi genospecies in biological fluids of Lyme disease patients. J Clin Microbiol. 1995;33:602–8.PubMedGoogle Scholar
- Norman AF, Regnery R, Jameson P, Greene C, Krause DC. Differentiation of Bartonella-like isolates at the species level by PCR-restriction fragment length polymorphism in the citrate synthase gene. J Clin Microbiol. 1995;33:1797–803.PubMedGoogle Scholar
- Anderson BE, Sumner JW, Dawson JE, Tzianabos T, Greene CR, Olson JG, Detection of the etiologic agent of human ehrlichiosis by polymerase chain reaction. J Clin Microbiol. 1992;30:775–80.PubMedGoogle Scholar
- Massung RF, Slater K, Owens JH, Nicholson WL, Mather TN, Solberg VB, Nested PCR assay for detection of granulocytic ehrlichiae. J Clin Microbiol. 1998;36:1090–5.PubMedGoogle Scholar
- Zhi N, Ohashi N, Rikihisa Y. Multiple p44 genes encoding major outer membrane proteins are expressed in the human granulocytic ehrlichiosis agent. J Biol Chem. 1999;274:17828–36. DOIPubMedGoogle Scholar
- Vorobyeva NN, Korenberg EI, Grigoryan YV. Diagnostics of tick-borne diseases in the endemic region of Russia. Wien Klin Wochenschr. 2002;114:610–2.PubMedGoogle Scholar
- Fenollar F, Raoult D. Diagnosis of rickettsial diseases using samples dried on blotting paper. Clin Diagn Lab Immunol. 1999;6:483–8.PubMedGoogle Scholar
- La Scola B, Raoult D. Laboratory diagnosis of rickettsioses: current approaches to diagnoses of old and new rickettsial diseases. J Clin Microbiol. 1997;35:2715–27.PubMedGoogle Scholar
- Raoult D, Roux V. Rickettsioses as paradigms of new or emerging infectious diseases. Clin Microbiol Rev. 1997;10:694–719.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 10, Number 5—May 2004
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Oleg Y. Mediannikov, Unité des Rickettsies, Faculté de Médecine, CNRS UMR 6020 27, Université de la Méditerranée, Bd Jean Moulin, 27, 13385 Marseille, Cedex 05, France; fax: +33-04-91-83-03 90
Top