Volume 11, Number 12—December 2005
Dispatch
Human Rickettsia felis Infection, Canary Islands, Spain
Cite This Article
Citation for Media
Abstract
We report the first cases of human infection by Rickettsia felis in the Canary Islands. Antibodies against R. felis were found in 5 adsorbed serum samples from 44 patients with clinically suspected rickettsiosis by Western blot serology. Fleas from 1 patient's dog were positive for R. felis by polymerase chain reaction.
Rickettsia felis is an intracellular bacterium (genus Rickettsia, spotted fever group [SFG]) (1,2). Its biological cycle involves the cat flea (Ctenocephalides felis) as the main vector (3). R. felis has been found in C. felis and C. canis in the Americas, Europe, Africa, Asia, and Oceania (1,3–6). Human disease caused by R. felis was unknown until 1994 (4). Since then, R. felis infection has been reported in Mexico (3 patients) (7), Germany (1 patient) (8), Brazil (2 patients) (1), and France (2 patients) (1). The clinical manifestations of the disease include high fever, rash, and elevation of liver enzymes (1,4,7). Exposure to fleas or to flea-prone animals is sometimes recorded (7,8).
On the Canary Islands (Atlantic islands of Spain), autochthonous cases of murine typhus have been reported (9). Although we suspected that some patients with a clinical picture of murine typhus actually had R. felis infection, we were not able to confirm this hypothesis. Therefore, 44 serum samples from 44 patients from the Canary Islands with suspected murine typhus were sent to the Unité des Rickettsies in Marseille, France, for specific serologic tests. Here, we describe the first 5 human infections caused by R. felis on the Canary Islands.
Forty-four patients were recruited for a prospective study of fever of intermediate duration (i.e., fever without focal symptoms lasting 7–28 days). Demographic, clinical, and laboratory data were collected for all patients. Chest radiographs and blood and urine cultures were taken. Antibodies against R. typhi were tested by direct immunofluorescence test (bioMérieux, Marcy L'Etoile, France) in the Canary Islands. Among the 44 patients, 24 showed a positive serologic result. Antibodies against other agents (Coxiella burnetii, R. conorii, Leptospira spp., Epstein-Barr virus, cytomegalovirus, HIV, and hepatitis B virus) were also tested; all were negative.
To search for evidence of infection with R. felis, all serologic results were confirmed by microimmunofluorescence (MIF) in France, as previously described (10). Systematic testing of SFG rickettsia antigens present in Europe and Africa was performed in parallel. The MIF procedure was followed by the use of Western blot and cross-adsorption studies. An immunofluorescence assay was considered positive if immunoglobulin G (IgG) titers were >1:64 or if IgM titers were >1:32. When cross-reactions were noted between the rickettsial antigens, the analysis comprised 3 steps. First, a rickettsial antigen was considered to represent the agent of infection when IgG or IgM antibody titers against this antigen were >2 serial dilutions higher than titers of IgG or IgM antibody against other rickettsial antigens (11). Second, when the difference in titers between R. felis and other antigens was <2 dilutions, Western blot assays were performed. A rickettsial antigen was considered the agent of infection when sera reacted only against the specific protein of this antigen. Expected molecular masses of the specific proteins were ≈125 kDa for R. typhi and 31 kDa for R. felis. Finally, when Western blot assays were not diagnostic, cross-adsorption studies were performed, as previously described (12). Specific diagnosis criteria after cross-adsorptions studies included a Western blot assay that showed exclusive reactivity with specific proteins of a sole agent. If reactivity with the 2 tested agents was still observed, diagnosis of an indeterminate rickettsial disease was made. With this strategy, patients were classified by 3 types: R. felis infection, R. typhi infection, and indeterminate rickettsial disease.
Five fleas from the dog of 1 R. felis–infected patient were tested by polymerase chain reaction (PCR) (3). DNA was extracted and amplified with primers that targeted the citrate synthase sequence, as previously described (3). For negative controls, we used sterile water and infection-free fleas previously tested in our laboratory; both negative controls were tested after every 7 samples. Amplicons were separated by electrophoresis on 1% agarose gels and then purified by using a QIAquick PCR purification kit (Qiagen, Hilden, Germany), as described by the manufacturer. PCR products were sequenced by using the d-rhodamine terminator-cycle sequencing kit (PE Applied BioSystems, Courtabeuf, France), as described by the manufacturer. The sequences obtained were compared with those available in the GenBank DNA database by using the program Basic Local Alignment Search Tool (BLAST, version 2.0, National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov/BLAST/).
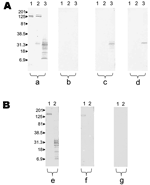
A rickettsial infection was diagnosed by using MIF for 31 of the 44 patients: 5 patients with the final diagnosis of R. felis infection, 13 with the diagnosis of R. typhi infection, and 13 with the diagnosis of indeterminate rickettsioses (Table). The diagnosis of R. felis infection was based on Western blot results on adsorbed sera for all patients. All the antibodies of these patients were removed when the R. felis–adsorbed sera were analyzed with R. typhi and R. felis antigens, whereas antibodies to R. felis remained when the R. typhi–adsorbed sera were analyzed.
Western blots performed with unadsorbed and adsorbed sera are represented in the Figure. Features of patients are indicated in the Appendix. Some differences were found between groups. The interval between the beginning of clinical signs and symptoms and evaluation was significantly more prolonged in the R. felis group than others. In the R. typhi group, odynophagia, cough, and rash were more frequent. When we compared biologic data, no difference was observed between R. typhi and R. felis groups, except for milder hypertransaminasemia in the latter group. Finally, 2 PCR products were obtained and sequenced from 2 fleas. Both sequences were 100% similar to R. felis citrate synthase gene in GenBank accession no. AF210692. No fleas were positive for R. typhi. Amplification was unsuccessful in all negative controls.
In the past 10 years, application of molecular tools has resulted in discovery of several new species of pathogenic rickettsiae, including R. felis. Since then, this bacterium was cultivated, and its genome was sequenced (1,13). Its pathogenic role was recently demonstrated in patients with serologic evidence of infection in Brazil, France, and Germany (1). R. felis DNA has also been detected in sera in Texas, Mexico, Brazil, and Germany (1,4,8,14). Autochthonous human rickettsioses that occur in the Canary Islands include murine typhus; SFG infections have never been reported (9). We diagnosed 5 cases of acute R. felis infection (15). The clinical picture is globally similar to murine typhus (4). However, the R. felis infection in our study seemed to be milder, and no skin rash was observed. The incidence of R. felis infection in the Canary Islands is probably underestimated; therefore, serologic tests for R. felis should be performed in patients with prolonged fever or suspected rickettsioses.
Cross-reactions in serologic testing for R. felis are unpredictable (3). In our study, patients with R. felis infection more frequently had high antibody titers (IgM >1:32 and IgG >1:64) to R. conorii and R. typhi (2 of 5 patients) than did patients with R. typhi infection (0 of 13). On the basis of R. felis data, we conclude that patients with R. felis infection may have no cross-reactivity with other rickettsiae, cross-reactivity with SFG rickettsiae, or cross-reactivity with both SFG rickettsiae and R. typhi. Genetic support for cross-reactivity with R. conorii is plausible because most membrane proteins of SFG and R. felis are extremely close (surface cell antigen [Sca] family). Genome analysis showed that several genes were present in R. felis and R. typhi and absent for other SFG, which could explain the cross-reactivity between R. felis and R. typhi (13). Finally, if <2-fold differences in IgG/IgM titers between R. felis and other SFG and typhus group rickettsiae are observed, only Western blot and cross-adsorptions will allow a specific diagnosis once reactivity has disappeared after adsorption with R. felis antigen. By contrast, a band of ≈31 kDa for the R. felis antigen persists after adsorption with R. conorii and R. typhi.
Dr Pérez-Arellano is chief of the Infectious Diseases and Tropical Medicine Service and professor in the Department of Medical and Surgical Sciences, Health Sciences Faculty, University of Las Palmas de Gran Canaria. His main research interests are imported and emerging infectious diseases.
Acknowledgments
We thank Kelly Johnston and Silpak Biswas for reviewing the manuscript.
This research was supported in part by a grant from the Canary Foundation of Investigation and Health.
References
- Raoult D, La Scola B, Enea M, Fournier PE, Roux V, Fenollar F, A flea-associated Rickettsia pathogenic for humans. Emerg Infect Dis. 2001;7:73–81. DOIPubMedGoogle Scholar
- La Scola B, Meconi S, Fenollar F, Rolain JM, Roux V, Raoult D. Emended description of Rickettsia felis (Bouyer et al. 2001), a temperature-dependent cultured bacterium. Int J Syst Evol Microbiol. 2002;52:2035–41. DOIPubMedGoogle Scholar
- Rolain JM, Franc M, Davoust B, Raoult D. Molecular detection of Bartonella quintana, B. koehlerae, B. henselae, B. clarridgeiae, Rickettsia felis, and Wolbachia pipientis in cat fleas, France. Emerg Infect Dis. 2003;9:338–42.PubMedGoogle Scholar
- Schriefer ME, Sacci JB Jr, Dumler JS, Bullen MG, Azad AF. Identification of a novel rickettsial infection in a patient diagnosed with murine typhus. J Clin Microbiol. 1994;32:949–54.PubMedGoogle Scholar
- Parola P, Sanogo OY, Lerdthusnee K, Zeaiter Z, Chauvancy G, Gonzalez JP, Identification of Rickettsia spp. and Bartonella spp. in from the Thai-Myanmar border. Ann N Y Acad Sci. 2003;990:173–81. DOIPubMedGoogle Scholar
- Kelly PJ, Meads N, Theobald A, Fournier PE, Raoult D. Rickettsia felis, Bartonella henselae, and B. clarridgeiae, New Zealand. Emerg Infect Dis. 2004;10:967–8.PubMedGoogle Scholar
- Zavala-Velazquez J, Ruiz-Sosa J, Sanchez-Elias R, Becerra-Carmona G, Walker D. Rickettsia felis in Yucatan. Lancet. 2000;356:1079–80. DOIPubMedGoogle Scholar
- Richter J, Fournier P, Petridou J, Häussinger D, Raoult D. Rickettsia felis infection acquired in Europe and documented by polymerase chain reaction. Emerg Infect Dis. 2002;8:207–8. DOIPubMedGoogle Scholar
- Hernandez CM, Angel-Moreno A, Santana E, Bolanos M, Frances A, Martin-Sanchez MS, Murine typhus with renal involvement in Canary Islands, Spain. Emerg Infect Dis. 2004;10:740–3.PubMedGoogle Scholar
- Teysseire N, Raoult D. Comparison of Western blot immunoblotting and microimmunofluorescence for diagnosis of Mediterranean spotted fever. J Clin Microbiol. 1992;30:455–60.PubMedGoogle Scholar
- Raoult D, Fournier P, Fenollar F, Jensenius M, Prioe T, De Pina J, Rickettsia africae, a tick-borne pathogen in travelers to Sub-Saharan Africa. N Engl J Med. 2001;344:1504–10. DOIPubMedGoogle Scholar
- La Scola B, Rydkina L, Ndihokubwayo JB, Raoult D. Serological differentiation of murine typhus and epidemic typhus using cross-adsorption and western blotting. Clin Diagn Lab Immunol. 2000;7:612–6.PubMedGoogle Scholar
- Ogata H, Renesto P, Audic S, Robert C, Blanc G, Fournier PE, The genome sequence of Rickettsia felis identifies the first putative conjugative plasmid in an obligate intracellular parasite. PLoS Biol. 2005;3:248. DOIPubMedGoogle Scholar
- Zavala-Velazquez JE, Zavala-Castro JE, Vado-Solis I, Ruiz-Sosa JA, Moron CG, Bouyer DH, Identification of Ctenocephalides felis fleas as a host of Rickettsia felis, the agent of a spotted fever rickettsiosis in Yucatan, Mexico. Vector Borne Zoonotic Dis. 2002;2:69–75. DOIPubMedGoogle Scholar
- La Scola B, Raoult D. Laboratory diagnosis of rickettsioses: current approaches to diagnosis of old and new rickettsial diseases. J Clin Microbiol. 1997;35:2715–27.PubMedGoogle Scholar
Figure
Table
Cite This Article1These authors contributed equally to this article.
Table of Contents – Volume 11, Number 12—December 2005
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
D. Raoult, Unité des Rickettsies, CNRS UMR 6020, Faculté de Médecine, Université de la Méditerranée, 27 Bd Jean Moulin, 13385 Marseille CEDEX 05, France; fax: 33-4-91-38-77-72
Top