Volume 11, Number 6—June 2005
Dispatch
Erythromycin-nonsusceptible Streptococcus pneumoniae in Children, 1999–2001
Cite This Article
Citation for Media
Abstract
After increasing from 1995 to 1999, invasive erythromycin-nonsusceptible Streptococcus pneumoniae rates per 100,000 decreased 53.6% in children from Baltimore, Maryland (US), from 1999 to 2001, which was partially attributed to strains related to the mefE-carrying England14-9 clone. The decline in infection rates was likely due to the pneumococcal 7-valent conjugate vaccine.
From 1995 to 1999, the prevalence of macrolide resistance among invasive pneumococci doubled to 20% in the United States (1). The rise in the 1990s was primarily due to strains with an M phenotype, a surrogate marker for the mefE gene (1,2). In 1999, children <5 years of age and white persons had a higher proportion of M phenotype strains causing invasive disease than did older persons and black persons (1). Most macrolide-resistant strains in the United States were also penicillin-nonsusceptible (1); modeling suggested that strains resistant to both drug classes would increase without a vaccine or other intervention (3). Since the introduction of the 7-valent pneumococcal conjugate vaccine (PCV7) in 2000, the overall incidence of macrolide-resistant infections, including serotype 14 strains, has decreased in Atlanta, Georgia (4). It is unclear whether these changes are caused by shifts in a small number of clones. Most drug-resistant infections in the United States are related to a small number of international clones (5).
The Baltimore metropolitan area is one of the sites in the Active Bacterial Core system that conducts active, laboratory surveillance for invasive pneumococcal disease. Approximately 15 million people residing in Maryland, Georgia, California, Minnesota, Oregon, Tennessee, and Connecticut were included in the multicenter study (1). In this study, we calculated the rates of invasive erythromycin-nonsusceptible S. pneumoniae disease, mefE-associated disease, and disease due to mefE-carrying clones in the Baltimore metropolitan area in 1995, 1999, and 2001. These years were chosen to validate the earlier multicenter study in the Baltimore metropolitan area (1995 and 1999) and include 1 year after licensure of PCV7 (2001). PCV7 includes serotypes 4, 6B, 9V, 14, 18C, 19F, and 23F. We also assessed whether the mefE-carrying strains were equally distributed in all populations, in all locations, and during both respiratory and nonrespiratory seasons from 1995 to 2001. Cases of invasive pneumococcal disease with an erythromycin MIC ≥0.5 μg/mL isolated from January 1,1995, to December 31, 2001, were included. Strains with an erythromycin MIC ≥0.5 μg/mL and penicillin MIC ≥0.12 μg/mL were defined as erythromycin-nonsusceptible and penicillin-nonsusceptible S. pneumoniae, respectively (6). Pneumococcal serotypes were determined at the Centers for Disease Control and Prevention by the latex agglutination test and confirmed with Quellung reaction. The presence of mef and/or ermB was determined by using a duplex polymerase chain reaction (PCR) (7), and mefA was differentiated from mefE by PCR-restriction fragment length polymorphisms (8). Pulsed-field gel electrophoresis (PFGE) was performed on all strains (9). Dendrograms were created in Bionumerics (Applied Maths, Sint-Martens-Latem, Belgium) with a position tolerance of 1.5%. Strains were compared to the first 25 international clones (http://www.sph.emory.edu/PMEN/) (7). Multi-locus sequence typing was performed on 87 (24.5%) of the 349 strains (10,11), and included the spectrum of PFGE patterns. Based on PFGE patterns with ≥80% relatedness by dendrogram, and/or 5 identical alleles, strains were classified into sequence type (ST)-complexes. The following 4 ST-complexes were classified as the clones in this analysis: ST9-complex (related to England14-9 clone), ST81-complex (related to Spain23F-1 clone), ST156-complex (related to Spain9V-3 clone), and ST236-complex (related to the Taiwan19F-14 clone).
Rates were calculated by using population estimates from US Census Bureau data for the Baltimore metropolitan area for 1995, 1999, and 2001. Chi-square and Fisher exact tests were used to compare the proportion of the population with disease in 1995, 1999, and 2001 (SAS 8.2, SAS Institute, Cary, NC, USA). Cochran-Armitage trend test was used to compare the proportion of erythromycin-nonsusceptible S. pneumoniae strains carrying the mefE gene from 1995 to 2001. Age groups were defined as children <5 years of age and persons ≥5 years of age; races were defined as persons of white and black race (1); respiratory and nonrespiratory seasons were defined as November–April and May–October, respectively (12).
Most cases of invasive pneumococcal disease occurred in 3 geographic regions: 61.0% (2,976/4,885) in Baltimore City, 18.3% (895/4,885) in Baltimore County, and 9.8% (480/4,885) in Anne Arundel County. From January 1, 1995, to December 31, 2001, a total of 4,885 pneumococcal cases were detected in the Baltimore metropolitan area, of which 85.8% (4,192/4,885) were available for MIC testing. Ninety-seven percent (349/360) of the erythromycin-nonsusceptible S. pneumoniae isolates were available for further analysis. Of these isolates, 255 (73.1%) carried only the mefE gene, 61 (17.5%) carried only the ermB gene, 8 (2.3%) carried both the mefE and ermB genes, 6 (1.7%) carried the mefA gene, and 19 (5.4%) had unknown resistance mechanisms. All isolates carrying both the ermB and mefE genes were serogroup 19 strains that were related to the Taiwan19F-14 clone. The mefA-carrying strains were either serotype 6B or serotype 14. The serotype 6B strains, belonging to ST146, were detected in Baltimore City during a 3-month period in 1998; the serotype 14 strains were detected in Howard County in 3 different years.
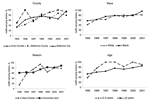
The incidence of invasive pneumococcal disease significantly increased from 1995 to 1999 before dramatically decreasing from 1999 to 2001. From 1995 to 1999, the overall rates of erythromycin-nonsusceptible S. pneumoniae and mefE-associated disease increased, and then stabilized from 1999 to 2001 (Table). While the overall rates of erythromycin-nonsusceptible S. pneumoniae were stable, the proportion of pneumococcal strains with reduced susceptibility to erythromycin increased from 5.1% (26/510) in 1995 to 13.6% (77/567) in 2001 (p<0.01). Moreover, the proportion of erythromycin-nonsusceptible S. pneumoniae strains carrying the mefE gene with an erythromycin MIC ≥16 μg/mL increased from 0% (0/12) in 1995 to 12.3% (8/65) in 2001 (χ2 for linear trend, p = 0.02). The proportion of erythromycin-nonsusceptible S. pneumoniae strains carrying the mefE gene increased from 48.0% (12/25) in 1995 to 85.5% (65/76) in 2001 (p<0.01). Of the erythromycin-nonsusceptible S. pneumoniae strains, the proportion of mefE-carrying strains that were penicillin-nonsusceptible S. pneumoniae rose from 20.0% (5/25) in 1995 to 72.4% (55/76) in 2001 (p<0.01). In 3 counties, in both age groups, in both races, and during both seasons, the proportion of mefE-carrying strains increased from 1995 to 2001 (Figure; p values for all analyses were ≤0.02). Sixty-nine percent (182/263) of the mefE-carrying strains were related to 4 international clones (percent serotype): 30.0% England14-9 clone (100% 14); 16.0% Spain23F-1 clone (83.3% 23F); 14.1% Spain9V-3 clone (97.3% 9V); and 9.1% Taiwan19F-14 clone (95.8% 19F).
Among children <5 years old, the incidence of erythromycin-nonsusceptible S. pneumoniae increased from 1995 to 1999 before declining by 53.6% (p = 0.03) between 1999 and 2001. Likewise, the incidence of mefE-associated disease rose in the 1990s before decreasing by 51.5% from 1999 to 2001 (p = 0.07). The rates of disease due to strains related to the England14-9 clone mirrored the age-specific trends from 1995 to 2001 (p≤0.05; Table). In contrast, in persons ≥5 years of age, the rate of erythromycin-nonsusceptible S. pneumoniae disease remained stable after increasing in the 1990s. From 1999 to 2001, the rate of mefE-associated resistance increased by 54.6% (p = 0.04; Table). Strains related to the England14-9 clone partially accounted for this increase.
The incidence of invasive disease in the Baltimore metropolitan area increased in the 1990s before declining from 1999 to 2001 (13). Likewise, the rate of invasive erythromycin-nonsusceptible S. pneumoniae disease increased in the 1990s (1). From 1999 to 2001, the overall incidence of erythromycin-nonsusceptible S. pneumoniae disease and mefE-associated disease stabilized. The proportion of erythromycin-nonsusceptible S. pneumoniae strains carrying the mefE gene dramatically increased from 1995 to 2001. The increase in the proportion of erythromycin-nonsusceptible S. pneumoniae strains carrying the mefE gene over time was detected in patients residing in all 3 geographic locations, from both races, from both age groups, and during both the respiratory and nonrespiratory seasons.
Both the incidence of invasive erythromycin-nonsusceptible S. pneumoniae disease and mefE-associated disease declined by >50% from 1999 to 2001 in children <5 years of age. This decrease was partially due to serotype 14 strains related to the England14-9 clone. Strains related to this clone may also account for the substantial decrease in macrolide-resistant serotype 14 infections noted in Atlanta (4). In contrast to the Atlanta study, the rates of mefE-associated disease increased among persons ≥5 years. The differences in rates detected in Baltimore compared to Atlanta may reflect regional variation and the inclusion of 2002 in the Atlanta analysis (4). In this study, the decrease in the incidence of erythromycin-nonsusceptible S. pneumoniae in children may have been due to variation in antimicrobial drug use or to introduction of PCV7.
In summary, after increasing in the 1990s, the rates of invasive erythromycin-nonsusceptible S. pneumoniae disease stabilized overall and decreased in children from 1999 to 2001. This remarkable decline was most likely due to PCV7, although differential antimicrobial drug may have been a contributor. Unfortunately, the lack of decline in the rate of invasive erythromycin-nonsusceptible S. pneumoniae infections among persons ≥5 years of age, coupled with the marked increase in dual resistance and the increase in the proportion with erythromycin MICs ≥16 μg/mL from 1995 to 2001 (14), is cause for concern. Public health initiatives that focus on judicious use of antimicrobial drugs and the PCV7 vaccine (13) may be beneficial in slowing these trends (15).
Dr. McEllistrem is an assistant professor in the Division of Infectious Diseases, Department of Medicine, at the University of Pittsburgh. Her current research interest focuses on molecular epidemiology and pathogenesis of pneumococcal infections in the 7-valent pneumococcal conjugate vaccine era.
Acknowledgments
We thank the participating hospital infection control practitioners and microbiology laboratory personnel in the Baltimore metropolitan area for identifying the pneumococcal cases and providing the microbial isolates; Yvonne Deane-Hibbert, Jackie Hunter, Carolyn Wright, and Tami Hilger Skoff for assistance in conducting surveillance; Kim Holmes for assistance with data collection; Althea Glenn Laboratories administration for processing the isolates; M. Leticia McElmeel, Sharon Crawford, Letitia Fulcher, and Christa Trippy for the antimicrobial susceptibility determinations; and Anne Schuchat for her cogent review of this manuscript.
These data were presented in part at the Fourth International Conference on Emerging Infectious Diseases in Atlanta, Georgia, in March 2004.
This work was supported in part by the State of Maryland, the Centers for Disease Control and Prevention's Emerging Infections Program, and career development awards from the National Institutes of Health to M.C.M. (K23 AI01788-03) and to L.H.H. (K24 AI52788).
References
- Hyde TB, Gay K, Stephens DS, Vugia DJ, Pass M, Johnson S, Macrolide resistance among invasive Streptococcus pneumoniae isolates. JAMA. 2001;286:1857–62. DOIPubMedGoogle Scholar
- Gay K, Baughman W, Miller Y, Jackson D, Whitney CG, Schuchat A, The emergence of Streptococcus pneumoniae resistant to macrolide antimicrobial agents: a 6-year population-based assessment. J Infect Dis. 2000;182:1417–24. DOIPubMedGoogle Scholar
- McCormick AW, Whitney CG, Farley MM, Lynfield R, Harrison LH, Bennett NM, Geographic diversity and temporal trends of antimicrobial resistance in Streptococcus pneumoniae in the United States. Nat Med. 2003;9:424–30. DOIPubMedGoogle Scholar
- Stephens DS, Zughaier SM, Whitney CG, Baughman WS, Barker L, Gay K, Incidence of macrolide resistance in Streptococcus pneumoniae after introduction of the pneumococcal conjugate vaccine: population-based assessment. Lancet. 2005;365:855–63. DOIPubMedGoogle Scholar
- Richter SS, Heilmann KP, Coffman SL, Huynh HK, Brueggemann AB, Pfaller MA, The molecular epidemiology of penicillin-resistant Streptococcus pneumoniae in the United States, 1994–2000. Clin Infect Dis. 2002;34:330–9. DOIPubMedGoogle Scholar
- National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial susceptibility testing. Document no. NCCLS M100-S14. Wayne (PA): National Committee for Clinical Laboratory Standards: 2004.
- McGee L, McDougal L, Zhou J, Spratt BG, Tenover FC, George R, Nomenclature of major antimicrobial-resistant clones of Streptococcus pneumoniae defined by the pneumococcal molecular epidemiology network. J Clin Microbiol. 2001;39:2565–71. DOIPubMedGoogle Scholar
- Del Grosso M, Iannelli F, Messina C, Santagati M, Petrosillo N, Stefani S, Macrolide efflux genes mef(A) and mef(E) are carried by different genetic elements in Streptococcus pneumoniae. J Clin Microbiol. 2002;40:774–8. DOIPubMedGoogle Scholar
- McEllistrem MC, Pass M, Elliott JA, Whitney CG, Harrison LH. Clonal groups of penicillin-resistant Streptococcus pneumoniae in Baltimore, Maryland: a population-based, molecular epidemiologic study. J Clin Microbiol. 2000;38:4367–72.PubMedGoogle Scholar
- McEllistrem MC, Adams J, Mason EO, Wald ER. Epidemiology of acute otitis media caused by Streptococcus pneumoniae before and after licensure of the 7-valent pneumococcal protein conjugate vaccine. J Infect Dis. 2003;188:1679–84. DOIPubMedGoogle Scholar
- Enright MC, Spratt BG. A multilocus sequence typing scheme for Streptococcus pneumoniae: identification of clones associated with serious invasive disease. Microbiology. 1998;144:3049–60. DOIPubMedGoogle Scholar
- Albanese BA, Reed ZH, Roche JC, Whitney CG, McEllistrem MC, Harrison LH. Geographic, demographic, and seasonal differences in penicillin-resistant Streptococcus pneumoniae in Baltimore. Clin Infect Dis. 2002;34:15–21. DOIPubMedGoogle Scholar
- Whitney CG, Farley MM, Hadler J, Harrison LH, Bennett NM, Lynfield R, Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. N Engl J Med. 2003;348:1737–46. DOIPubMedGoogle Scholar
- Fogarty C, Goldschmidt R, Bush K. Bacteremic pneumonia due to multidrug-resistant pneumococci in 3 patients treated unsuccessfully with azithromycin and successfully with levofloxacin. Clin Infect Dis. 2000;31:613–5. DOIPubMedGoogle Scholar
- Seppala H, Klaukka T, Vuopio-Varkila J, Muotiala A, Helenius H, Lager K, The effect of changes in the consumption of macrolide antibiotics on erythromycin resistance in group A streptococci in Finland. Finnish Study Group for Antimicrobial Resistance. N Engl J Med. 1997;337:441–6. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleTable of Contents – Volume 11, Number 6—June 2005
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
M. Catherine McEllistrem, Division of Infectious Diseases, Suite 3A, Falk Medical Bldg, 3601 Fifth Ave, University of Pittsburgh, Pittsburgh, PA 15213-2582, USA; fax: 412-648-6399
Top