Volume 12, Number 1—January 2006
Research
Economics of Neuraminidase Inhibitor Stockpiling for Pandemic Influenza, Singapore
Cite This Article
Citation for Media
Abstract
We compared strategies for stockpiling neuraminidase inhibitors to treat and prevent influenza in Singapore. Cost-benefit and cost-effectiveness analyses, with Monte Carlo simulations, were used to determine economic outcomes. A pandemic in a population of 4.2 million would result in an estimated 525–1,775 deaths, 10,700–38,600 hospitalization days, and economic costs of $0.7 to $2.2 billion Singapore dollars. The treatment-only strategy had optimal economic benefits: stockpiles of antiviral agents for 40% of the population would save an estimated 418 lives and $414 million, at a cost of $52.6 million per shelf-life cycle of the stockpile. Prophylaxis was economically beneficial in high-risk subpopulations, which account for 78% of deaths, and in pandemics in which the death rate was >0.6%. Prophylaxis for pandemics with a 5% case-fatality rate would save 50,000 lives and $81 billion. These models can help policymakers weigh the options for pandemic planning.
Ten percent of the world's population and 20% of the population of tropical Singapore are infected with influenza virus annually (1,2). Amid growing concern about influenza pandemics, national preparedness plans have become essential. In a pandemic hastened by globalization, vaccination is not a viable initial solution because vaccine production requires an estimated 6 months (1,3). Instead, neuraminidase inhibitors are influenza-specific antiviral agents that figure strongly in preparedness plans. Many nations are acquiring stockpiles of these drugs because of their effectiveness in influenza treatment and prophylaxis (4).
Studies have compared the cost-effectiveness of vaccination versus treatment with antiviral agents (5–7), but only l study has examined the cost-effectiveness of prophylaxis (8). We provide further comparison of the economic outcomes of prophylaxis or treatment with antiviral agents to provide national planners with optimal strategies.
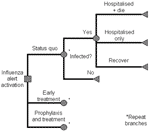
This study used a decision-based model (Figure 1) to perform cost-benefit and cost-effectiveness analyses for stockpiling antiviral agents in Singapore. Oseltamivir was the drug of choice because of its safety profile (9,10) and available data on influenza prophylaxis and treatment (11,12). The model compared 3 strategies: supportive management (no action), early treatment of clinical influenza with oseltamivir (treatment only), and prophylaxis in addition to early treatment (prophylaxis). Costs were assigned to each outcome, and probabilities at each node were aggregated as population rates for calculating overall costs for each outcome. Decision branches were similar for each strategy, but probabilities at individual nodes differed.
Cost-benefit analyses were used to compare treatment-only and prophylaxis strategies to taking no action. These analyses included direct and indirect economic costs, such as the cost of death. However, quantifying the societal cost of death is difficult, and cost-effectiveness analyses based on cost per life saved by treatment only and prophylaxis, compared to no action, were included. The model was run by using Excel spreadsheets (Microsoft Corp, Redmond, WA, USA); details are shown in the Appendix and on Tan Tock Seng Hospital's website (http://www.ttsh.com.sg/doc/Pandemic%20influenza%20in%20Singapore%20-%20economic%20analysis%20of%20treatment%20and%20prophylaxis%20stockpiling%20strategies.pdf). Costs are represented in 2004 Singapore dollars (2004 exchange rate, USD$1 = SGD$1.6908).
Pandemic influenza is unpredictable: uncertainties surround its occurrence and outcomes (13). Excess deaths in annual epidemics occur mostly in the elderly (14), but the 1918–1919 Spanish flu pandemic had higher death rates among adults (15). To account for such uncertainties, the input variables were modeled as triangular distributions centered on base values, with ranges corresponding to minimum and maximum values (Table 1). Sensitivity analyses, including 1-way analysis, were conducted to identify variables of highest impact and the outcome's sensitivity to treatment and prophylaxis stockpiles. Monte Carlo simulation analyses were performed to determine outcomes under different scenarios.
Treatment stockpiles, based on proportions of the population, are used on all influenzalike-illness cases, from pandemic plan activation until the pandemic ceases or the stockpile is depleted, whichever comes first. Analysis was conducted to determine the proportion of untreated influenza patients and simulation iterations with complete coverage, by stockpile levels. Further analysis was then performed for prophylaxis stockpiles where prophylaxis, by weeks, is given to the population over and above treatment requirements.
Input Variables
Input variables are shown in Table 1. Conservative values favoring no action were used to justify alternative strategies. The study was conducted on Singapore's 2004 midyear population of 4,240,300 (16), divided into 3 age groups, each consisting of 2 risk groups (low and high risk, according to underlying medical conditions predisposing the patient to influenza complications), for a total of 6 groups that represented differing infection outcomes and drug responses (13).
The clinical attack rates during the 1918 and 1957 pandemics were 29.4% and 24%, respectively (23), and attack rates in Singapore during the 1967 pandemic were 12.8%–36.4% (22). This study assumed a base clinical attack rate of 30% (range 10%–50%), corresponding to rates in other studies (4,13,24).
Case-fatality rates were derived from Singapore's excess deaths from interpandemic influenza; hospitalization and death were assumed to occur only in clinical influenza. To reflect hospitalization rates in relation to case-fatality rates, both rates were correlated. For outpatient visits, clinical influenza patients were assumed to seek medical care and take medical leave. However, some patients may not be treated effectively within 48 hours of infection, and they were assumed not to benefit from treatment.
For pandemic duration, influenza activity in tropical climates commonly rises above the baseline for >12 weeks (31,33), compared to 6 weeks in temperate climates (34). This study assumed a 12-week pandemic duration base value with a range from 6 weeks (average temperate duration) to 24 weeks (assumed vaccine development).
Individual economic value was calculated from the net present value of future earnings for average-aged persons in the respective age groups, adjusted for age. Other costs included were hospitalizations and work days lost; all costs were standardized to 2004 Singapore dollars.
Oseltamivir
This study relied on international studies on oseltamivir. Oseltamivir has a good safety profile with insignificant rates of severe adverse events and drug withdrawal (9). Costs from side effects were thus assumed to be insignificant compared to costs for pandemic illness and deaths. The known safe administration duration of 8 weeks represents only studied durations (35). Extension is assumed possible, and the model included up to 24 weeks' prophylaxis. Oseltamivir trials have lacked the power to detect mortality reductions because influenza deaths in trials are rare (14), and wide ranges were used to account for uncertainty. Oseltamivir is also less effective in the elderly (24). Immunity after prophylaxis among those without clinical infection was assumed to be 35%, as shown during an influenza study in which 38% of study participants on prophylaxis had serologic infection but no clinical infection (12). Oseltamivir's pharmacologic action is selective and is assumed to be inactive against noninfluenza illnesses.
Stockpile use depends on the probability of an influenza pandemic occurring. Antigenic shifts and reappearances of past variants were estimated to have pandemic potential every 8–10 years (31,32). Using oseltamivir's shelf-life of 4 years and patent expiration in 2016, the model assumed a conservative base value of 2.25 stockpile cycles before use (range 1–3.5 cycles) to account for significantly reduced costs after patent expiration. The model assumed that all unused stockpiles are lost.
If no action were taken during a pandemic, the mean number of simulated deaths in Singapore would be 1,105 (5th and 95th percentiles of 525 and 1,775), with mean hospital days of 23,098 (10,736, 38,638). The mean economic cost would exceed SGD$1.43 billion (0.73, 2.19), and 78% of all deaths would occur in groups at high risk. From the sensitivity analyses, the outcome was most sensitive to changes in attack rate and case-fatality rate reduction with treatment and was sensitive to the variables of treatment and prophylaxis stockpiles.
Table 2 shows the cost and outcomes of various treatment stockpiles; each shelf-like cycle of the stockpile (which is 4 years, after which the drug has to be repurchased) costs SGD$13.1 million for 10% of the population. Stockpiles of <20% did not provide complete coverage in any simulated iterations, while stockpiles of >60% always provided complete coverage. The maximal mean economic benefit of SGD$414 million occurred at a 40% stockpile with 418 lives saved.
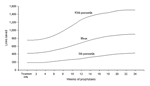
The population cost-benefit and cost-effectiveness outcomes from the Monte Carlo simulation analyses are shown in Table 3. The treatment-only strategy provided the best overall economic benefit, and the no-action strategy was dominated by the treatment-only strategy in cost per life saved.1 Each additional week of prophylaxis costs SGD$92 million but reduced the overall economic benefit. Figure 2 shows that increasing the duration of prophylaxis increased lives saved. Lives saved from prophylaxis compared to treatment increased significantly only after prophylaxis of >4 weeks and increased steadily until 20 weeks; costs per life saved also increased.
Table 4 shows that treatment-only provided the greatest economic benefit across all groups. As prophylaxis duration increased, economic benefit decreased. However, for the 3 groups at high risk (Table 1), the mean overall economic benefit of up to 24 weeks' prophylaxis remained positive compared to that seen if no action was taken.
The simulated proportion of decisions with treatment only or 24 weeks' prophylaxis as the optimal outcome is shown in Figure 3. At case-fatality rates of 0.05% (similar to interpandemic epidemics), the decision always favored treatment-only. With increasing case-fatality rates, the decision increasingly favored prophylaxis and intersects between rates of 0.4% and 0.6%. Prophylaxis was always optimal in case-fatality rates of >1.5%. If no action was taken with a 5% case-fatality rate (the 1918 pandemic average) (23), 63,000 deaths, 1.5 million hospital days, and economic costs of SGD$112 billion would occur. Treatment-only saved 30,000 lives, benefited the economy by SGD$28–$84 billion, and required 780,000 hospital days. Twenty-four weeks of prophylaxis saved 50,000 lives, benefited the economy by SGD$46–$132 billion, and required 240,000 hospital days.
The analyses suggest that treatment is always beneficial compared to no action and that the optimal treatment stockpile is 40%–60%: 40% maximizes economic benefits, while 60% maximizes treatment benefits. Compared to other strategies, treatment-only was the optimal economic strategy, while no action was always the least desirable option. Although treatment-only saved fewer lives than prophylaxis, stockpiling costs for treatment were lower. Prophylaxis was only economically beneficial compared with no action in subpopulations at high risk.
Substantial outcomes with prophylaxis occurred with durations of >4 weeks because shorter durations prolonged the pandemic, were insufficient for immunity, and did not cover the pandemic's peak. Increasing duration improved outcomes because it covered the pandemic's peak, but the improved outcomes tapered off after 20 weeks, resulting in a sigmoid curve (Figure 2).
In low-risk groups with low death and hospitalization rates, increasing prophylaxis duration decreased economic benefit and increased cost per life saved. In contrast, groups at high risk, who had higher death and hospitalization rates, were affected substantially by prophylaxis, resulting in overall benefits compared to taking no action. Elderly groups had the smallest populations but the highest risk levels and most deaths. However, their lower average future earnings compared to those of younger age groups resulted in lower overall benefits.
This study of pandemic outcomes in a tropical climate is similar to an Israeli study that compared treatment and prophylaxis strategies (8). Our study used local health outcome rates but did not include a ring prophylaxis strategy. Both studies found that oseltamivir treatment is economically beneficial, but in addition, our study showed that long-duration prophylaxis is beneficial for high-risk groups and high case-fatality pandemics.
Limitations of this study include the disregard for intangible costs, such as societal value of health; cost-utility analyses could address these costs. Also, indirect effects on national economy and world trade were not considered. For comparability, neither treatment nor prophylaxis was assumed to alter the pandemic's transmission dynamics. This assumption may be true if therapy is limited to small subpopulations, but it understates the benefits if infection is delayed until the pandemic is resolved or vaccine becomes available; it overestimates the benefits if the pandemic continues (4,24). Correlation between attack rates and pandemic duration was not accounted for, and all possible combinations were included.
Policy Implications
Stockpiling is insurance in planning for pandemics with high case-fatality rates, in which more severe outcomes and higher risks demand higher premiums. Policymakers should consider lives saved even if economic costs outweigh incremental benefits. Prophylaxis of high-risk groups balances saving lives with economic benefits. Prophylaxis also reduces hospitalizations, which may otherwise overwhelm the healthcare system. Analysis of peak pandemic healthcare use is required to determine the effects of prophylaxis. Other options to reduce a pandemic's impact, including reducing influenza attack rates by quarantine or closing borders, should be considered as alternative strategies.
The current avian influenza (H5N1) outbreak in Asia, which has a high case-fatality rate, indicates the need for decisive action. Oseltamivir is effective against H5N1 and is used as treatment in Vietnam (36,37). Although resistance has been detected, resistant strains have poor infectivity (37). Prophylaxis with oseltamivir will reduce illness, deaths, and economic costs and may reduce spread. If avian influenza develops species crossover with case fatalities exceeding those of the 1918 Spanish influenza pandemic, then stockpiling for treatment and prophylaxis accrues substantial benefits.
The decision to stockpile requires predetermined objectives; noneconomic, moral, and ethical implications should be considered. Treatment-only maximizes economic benefits, while prophylaxis saves most lives. Policymakers have to act decisively, and determine the subpopulations to be given priority, to enable preparedness plans to succeed.
Details of the Equations Used in the Analysis
Antiviral stockpiles will be used on clinical influenza cases according to the pandemic distribution curve, assumed to be normally distributed (14). Baseline influenzalike illness rates are assumed to be constant.
Proportion Untreated
The population proportion with clinical influenza left untreated because of treatment stockpile deficiencies is calculated as follows:
No. of doses required = (influenzalike illness per week × pandemic duration) + no. of clinical influenza cases
Shortfall of doses for treatment = no. of doses required – no. of doses available
The proportion untreated is the shortfall of treatment doses matched to the number of case-patients who require treatment, according to the pandemic distribution curve.
Cost of Treatment and Prophylaxis
The cost of treatment was calculated as follows:
Total cost of treatment agerisk group = cost of treatment per course × stockpile percentage × populationage, risk group
The cost of prophylaxis for 1 stockpile cycle was calculated as follows:
Total cost of prophylaxisage, risk group = cost of prophylaxis per week × no. weeks of prophylaxis × populationage, risk group
Cost of Outpatient Clinical Influenza
The medical cost of outpatient clinical influenza was calculated as follows:
Outpatient medical costsage, risk group = populationage, risk group × attack rate × consultation and treatment cost
The cost of outpatient lost days was calculated by using work days lost for the adult population and unspecified days lost for the young and elderly populations, as follows:
Economic cost of outpatient lost daysage, risk group = populationage, risk group × attack rate × outpatient days lost × value of a day lostage, risk group
Cost of Hospitalizations
The hospitalization cost for influenza-related complications was calculated by summing direct hospitalization cost with cost of additional days lost after hospitalization.
The direct hospitalization cost was calculated as follows:
Economic cost of hospitalizationage, risk group = populationage, risk group × attack rate × hospitalization rate age, risk group × length of stay age, risk group × (hospitalization cost + value of a day lost age, risk group)
The cost from additional days lost was calculated as follows:
Economic cost of additional days lost after hospitalization = population age, risk group × attack rate × hospitalization rateage, risk group × additional days lostage, risk group × value of a day lostage, risk group
Cost from Influenza Deaths
The cost from influenza deaths is calculated as follows:
Economic cost from influenza deaths = populationage, risk group × attack rate × case-fatality rateage, risk group × net present value of future earningsage, risk group
Economic Calculations
For cost-benefit comparisons, the following equation is used:
Overall benefit = overall costtreatment only or prophylaxis – overall costno action
For the cost-effectiveness comparisons, the following equation is used:
Cost per-life-saved compared to no action = (cost excluding cost per lifetreatment-only or prophylaxis – cost excluding cost per lifeno action) / (deathsno action – deathstreatment-only or prophylaxis)
The individual costs that constitute the total costs are calculated for the strategies of no action, treatment-only, and prophylaxis as follows:
Overall costno action, treatment-only, prophylaxis = Σ (populationage, risk group × probability of outcomeclinical influenza, hospitalization, death × cost of outcomeclinical influenza, hospitalization, death × effectivenesstreatment-only, prophylaxis) + cost of strategytreatment-only, prophylaxis
Dr Lee is a preventive medicine physician with the Singapore Ministry of Defence, currently working at the Communicable Disease Centre, Tan Tock Seng Hospital, Singapore. His research interests include clinical cost-effectiveness, emerging infectious diseases management, and clinical process improvement.
Acknowledgment
We thank K. Satku, Director of Medical Services, the staff at the Ministry of Health, and A. Earnest for their kind assistance.
References
- Gerdil C. The annual production cycle for influenza vaccine. Vaccine. 2003;21:1776–9. DOIPubMedGoogle Scholar
- Ng TP, Pwee TH, Niti M, Goh LG. Influenza in Singapore: assessing the burden of illness in the community. Ann Acad Med Singapore. 2002;31:182–8.PubMedGoogle Scholar
- Gust ID, Hampson AW, Lavanchy D. Planning for the next pandemic of influenza. Rev Med Virol. 2001;11:59–70. DOIPubMedGoogle Scholar
- van Genugten MLL, Heijnen MA, Jager JC. Pandemic influenza and healthcare demand in the Netherlands: scenario analysis. Emerg Infect Dis. 2003;9:531–8. DOIPubMedGoogle Scholar
- Muenning PA, Khan K. Cost-effectiveness of vaccination versus treatment of influenza in healthy adolescents and adults. Clin Infect Dis. 2001;33:1879–85. DOIPubMedGoogle Scholar
- Bridges CB, Thompson WW, Meltzer MI, Reeve GR, Talamonti WJ, Cox NJ, Effectiveness and cost-benefit of influenza vaccination of healthy working adults: a randomized controlled trial. JAMA. 2000;284:1655–63. DOIPubMedGoogle Scholar
- Lee PY, Matchar DB, Clements DA, Huber J, Hamilton JD, Peterson ED. Economic analysis of influenza vaccination and antiviral treatment for healthy working adults. Ann Intern Med. 2002;137:225–31.PubMedGoogle Scholar
- Balicer RD, Huerta M, Davidovitch N, Grotto I. Cost-benefit of stockpiling drugs for influenza pandemic. Emerg Infect Dis. 2005;11:1280–2.PubMedGoogle Scholar
- Nicholson KG, Aoki FY, Osterhaus AD, Trottier S, Carewicz O, Mercier CH, Efficacy and safety of oseltamivir in treatment of acute influenza: a randomized controlled trial. Lancet. 2000;355:1845–50. DOIPubMedGoogle Scholar
- Massarella JW, He GZ, Dorr A, Nieforth K, Ward P, Brown A. The pharmacokinetics and tolerability of the oral neuraminidase inhibitor oseltamivir (Ro 64-0796/GS4104) in healthy adult and elderly volunteers. J Clin Pharmacol. 2000;40:836–43. DOIPubMedGoogle Scholar
- Kaiser L, Wat C, Mills T, Mahoney P, Ward P, Hayden F. Impact of oseltamivir treatment on influenza-related lower respiratory tract complications and hospitalizations. Arch Intern Med. 2003;163:1667–72. DOIPubMedGoogle Scholar
- Hayden FG, Atmar RL, Schilling M, Johnson C, Poretz D, Paar D, Use of the selective oral neuraminidase inhibitor oseltamivir to prevent influenza. N Engl J Med. 1999;341:1336–43. DOIPubMedGoogle Scholar
- Meltzer MI, Cox NJ, Fukuda K. The economic impact of pandemic influenza in the United States: priorities for intervention. Emerg Infect Dis. 1999;5:659–71. DOIPubMedGoogle Scholar
- Lui KJ, Kendal AP. Impact of influenza epidemics on mortality in the United States from October 1972 to May 1985. Am J Public Health. 1987;77:712–6. DOIPubMedGoogle Scholar
- Ammon CE. Spanish flu epidemic in 1918 in Geneva, Switzerland. Euro Surveill. 2002;7:190–2.PubMedGoogle Scholar
- Key statistics. Singapore Department of Statistics. [cited 2005 May 3]. Available from http://www.singstat.gov.sg/
- Tan CH, Emmanuel SC, Tan BY, Jacob E. Prevalence of diabetes and ethnic differences in cardiovascular risk factors. Diabetes Care. 1999;22:241–7. DOIPubMedGoogle Scholar
- Ng TP. Adult asthma prevalence, morbidity and mortality and their relationships with environmental and medical care factors in Singapore. Asian Pac J Allergy Immunol. 1999;17:127–35.PubMedGoogle Scholar
- Heng DM, Lee J, Chew SK, Tan BY, Hughes K, Chia KS. Incidence of ischaemic heart disease and stroke in Chinese, Malays, and Indians in Singapore: Singapore Cardiovascular Cohort Study. Ann Acad Med Singapore. 2000;29:231–6.PubMedGoogle Scholar
- Wang XS, Tan TN, Shek LP, Chng SY, Hia CP, Ong NB, The prevalence of asthma and allergies in Singapore; data from two ISAAC surveys seven years apart. Arch Dis Child. 2004;89:423–6. DOIPubMedGoogle Scholar
- Emmanuel SC, Phua HP, Cheong PY. 2001 survey on primary medical care in Singapore. Singapore Med J. 2004;45:199–213.
- Kadri ZN. An outbreak of "Hong Kong flu" in Singapore. Singapore Med J. 1970;11:30–2.
- Glezen WP. Emerging infections: pandemic influenza. Epidemiol Rev. 1996;18:64–76.PubMedGoogle Scholar
- Turner D, Wailoo A, Nicholson K, Cooper N, Sutton A, Abrams K. Systematic review and economic decision modelling for the prevention and treatment of influenza A and B. Health Technol Assess. 2003;7:1–182.PubMedGoogle Scholar
- Haddix AC, Teutsh SM, Corso PS. Prevention effectiveness—a guide to decision analysis and economic evaluation. 2nd ed. New York: Oxford University Press; 2003.
- Lee KH, Chin NK, Tan WC, Lim TK. Hospitalised low risk community-acquired pneumonia: outcome and potential for cost-savings. Ann Acad Med Singapore. 1999;28:389–91.PubMedGoogle Scholar
- Treanor JJ, Hayden FG, Vrooman PS, Barbarash R, Bettis R, Riff D, Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza: a randomized controlled trial. US Oral Neuraminidase Study Group. JAMA. 2000;283:1016–24. DOIPubMedGoogle Scholar
- Woodall J, Rowson KEK, McDonald JC. Age and Asian influenza. BMJ. 1958;4:1316–8. DOIPubMedGoogle Scholar
- Bowles SK, Lee W, Simor AE, Vearncombe M, Loeb M, Tamblyn S, ; Oseltamivir Compassionate Use Program Group. Use of oseltamivir during influenza outbreaks in Ontario nursing homes, 1999–2000. J Am Geriatr Soc. 2002;50:608–16. DOIPubMedGoogle Scholar
- Welliver R, Monto AS, Carewicz O, Schatteman E, Hassman M, Hedrick J, ; Oseltamivir Post Exposure Prophylaxis Investigator Group. Effectiveness of oseltamivir in preventing influenza in household contacts: a randomized controlled trial. JAMA. 2001;285:748–54. DOIPubMedGoogle Scholar
- Doraisingham S, Goh KT, Ling AE, Yu M. Influenza surveillance in Singapore: 1972–86. Bull World Health Organ. 1988;66:57–63.PubMedGoogle Scholar
- Scholtissek C. Source for influenza pandemics. Eur J Epidemiol. 1994;10:455–8. DOIPubMedGoogle Scholar
- Chew FT, Doraisingham S, Ling AE, Kumarasinghe G, Lee BW. Seasonal trends of viral respiratory tract infections in the tropics. Epidemiol Infect. 1998;121:121–8. DOIPubMedGoogle Scholar
- Nguyen-Van-Tam J. Epidemiology of influenza. In: Nicholson KG, Webster RG, Hay AJ, editors. Textbook of influenza. Oxford: Blackwell Science Ltd; 1998. p. 181–206.
- Chik KW, Li CK, Chan PKS, Shing MMK, Lee V, Tam JSL, Oseltamivir prophylaxis during the influenza season in a paediatric cancer centre: prospective observational study. Hong Kong Med J. 2004;10:103–6.PubMedGoogle Scholar
- Govorkova EA, Leneva IA, Goloubeva OG, Bush K, Webster RG. Comparison of efficacies of RWJ-270201, zanamivir, and oseltamivir against H5N1, H9N2, and other avian influenza viruses. Antimicrob Agents Chemother. 2001;45:2723–32. DOIPubMedGoogle Scholar
- McKimm-Breschkin JL. Management of influenza virus infections with neuraminidase inhibitors: detection, incidence, and implications of drug resistance. Treat Respir Med. 2005;4:107–16. DOIPubMedGoogle Scholar
Figures
Tables
Cite This Article1"Dominate" is a term used in cost-effectiveness analyses and refers to a strategy that is both more efficacious and less costly than another strategy.
Table of Contents – Volume 12, Number 1—January 2006
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Vernon J Lee, Block 802, Communicable Disease Centre, Moulmein Rd, Singapore 308433; fax: 65-63577465
Top