Volume 12, Number 8—August 2006
Research
Antibody Response to Pneumocystis jirovecii
Cite This Article
Citation for Media
Abstract
We conducted a prospective pilot study of the serologic responses to overlapping recombinant fragments of the Pneumocystis jirovecii major surface glycoprotein (Msg) in HIV-infected patients with pneumonia due to P. jirovecii and other causes. Similar baseline geometric mean antibody levels to the fragments measured by an ELISA were found in both groups. Serum antibodies to MsgC in P. jirovecii patients rose to a peak level 3–4 weeks (p<0.001) after recovery from pneumocystosis; baseline CD4+ count >50 cells/μL and first episode of pneumocystosis were the principal host factors associated with this rise (both p<0.001). Thus, MsgC shows promise as a serologic reagent and should be tested further in clinical and epidemiologic studies.
Pneumocystis jirovecii, formerly known as Pneumocystis carinii special form hominis (1), is a leading cause of fatal pneumonia in HIV-positive persons and other immunosuppressed patients. Research on P. jirovecii has been hampered by the lack of a reliable in vitro culture system, so investigators have developed molecular techniques to characterize isolates. Studies by the Centers for Disease Control and Prevention, San Francisco General Hospital, and other medical centers in the United States that use these techniques have provided epidemiologic insights about P. jirovecii patients (2,3). Reports of P. jirovecii colonization detected by molecular probes in persons ranging from healthy young children to adults with chronic lung diseases raise the possibility that this organism may have medical and public health consequences beyond those on the immunocompromised host (4,5).
Serologic analysis may be useful in epidemiologic studies of P. jirovecii infection, especially in light of evidence that antibodies contribute to host defenses against the organism (6–8). Unfortunately, despite >40 years of investigation, a useful serologic test for P. jirovecii is not yet available (9,10). Antigens have mainly consisted of crude extracts from infected human or rodent lungs. These preparations have shown that antibodies to the organism are highly prevalent in the general population (4,11,12) but have been unable to distinguish present from past infection or colonization from active disease. Specific native P. jirovecii antigens have shown more promise as serologic reagents, but they are in short supply (11,13). This problem has been exacerbated by data about genetic diversity and host specificity of Pneumocystis, which have emphasized the importance of matching organisms used in studies with the host from which they have been derived (1).
More recent attention has turned toward the use of recombinant P. jirovecii antigens to study host immune responses (14–19). The major surface glycoprotein (Msg or gpA) is highly immunogenic and contains protective B- and T-cell epitopes, and the heavily glycosylated portion of the antigen plays a central role in the interaction of the organism with the host (6–10,20). Msg represents a family of proteins encoded by multiple genes and is thus capable of antigenic variation, which may serve as a mechanism to evade host immune responses. Our strategy has been to use a single Msg isoform that would enable us to begin to understand the host immune response to this complex glycoprotein. We developed 3 overlapping recombinant fragments (MsgA, MsgB, and MsgC), which span the entire length of the P. jirovecii Msg, and analyzed their reactivity with serum antibodies in different populations by Western blot (WB) and ELISA (17,18). A key finding in both studies was that asymptomatic, HIV-positive patients in Cincinnati with a past episode of Pneumocystis pneumonia (PCP) had a significantly higher degree of antibody reactivity to MsgC, the carboxyl terminus and most conserved part of the antigen, than patients who had never had the disease.
In this pilot study, we sought to determine whether serum antibody levels to MsgA, MsgB, and MsgC differed in HIV-positive patients with acute pneumonia due to P. jirovecii compared to those with pneumonia due to other causes. Further, we asked whether serum antibody levels would rise in these patients during treatment and recovery from pneumocystosis, which Msg fragment could best detect this increase, and whether specific host factors were associated with the antibody rise.
Patients and Study Design
As standard clinical practice, HIV-positive patients who came to San Francisco General Hospital with respiratory signs and symptoms compatible with pneumocystosis were evaluated by a uniform protocol that has been described previously (21). This protocol included obtaining specimens by induced sputum and, if necessary, bronchoscopy with bronchoalveolar lavage. Microscopic examination and cultures were used to establish a specific etiologic diagnosis. Consecutive patients undergoing sputum induction or bronchoscopy to diagnose PCP were enrolled in this study and provided written, informed consent to allow their medical records to be abstracted with a standardized data form. Study investigators classified patients as either PCP positive or PCP negative (controls), according to predetermined definitions that were blinded to serologic results. Pneumocystis patients were those patients with a microscopically confirmed diagnosis of P. jirovecii; these patients were treated with standard anti-Pneumocystis drugs as part of their regular medical care. Control patients were those whose microscopic examinations were negative for P. jirovecii, had Pneumocystis treatment discontinued, and recovered from acute pneumonia.
The study was conducted during a 4.5-year period (May 2000 through September 2004). During the first half of the study (2000–2002), an acute-phase serum specimen was drawn at the time of hospital admission for pneumonia, and a single convalescent-phase specimen was drawn at different intervals 5–12 weeks later. Preliminary analysis suggested that the Pneumocystis patients experienced a rise in mean serum antibody levels, whereas controls did not. Thus, during the later part of the study (2003–2004), additional serial convalescent-phase serum specimens were drawn every 1–2 weeks for 6 weeks from patients with pneumocystosis to measure early changes in antibody levels. Serum specimens were stored at -70°C and shipped to the University of Cincinnati for analysis. University of California San Francisco and University of Cincinnati institutional review boards approved the protocol.
Analysis of Serum Antibodies
Serum antibody levels to MsgA, MsgB, and MsgC were measured in a blinded manner by an ELISA as previously described (14,17,18). All serum specimens and the standard reference serum were diluted 1:100 and tested in duplicate wells of a 96-well plate against the following reagents: recombinant Msg fragments, Escherichia coli extract expressing the pET vector without insert (vector control), tetanus toxoid (TT) (positive control), and phosphate-buffered saline (PBS) without antigen (negative control). As an additional negative control, PBS was substituted for the serum specimen. Plates were washed, horseradish peroxidase (HRP)–labeled goat anti-human immunoglobulin G was added, plates were washed again, and tetramethylbenzidine substrate was added. The reaction was stopped by adding 0.18 mol/L H2SO4, and the plates were read at a wavelength of 450 nm. The reference serum specimen, which was obtained from a single person and had known reactivity to Msg, was run on each day as another control. HRP-labeled S-protein was used as a positive control and to correct for antigen loading. During the early part of the study, patient and reference serum specimens were tested at 1:100, 1:500 and 1:2,500 dilutions. The best results were obtained with the 1:100 dilution, so this dilution was used for the remainder of the study. The reactivity of each serum specimen to Msg was expressed as the ratio of reactivity to the pET vector: (mean optical density [OD] Msgtest serum – mean OD PBStest serum) / (mean OD pETtest serum __ mean OD PBStest serum).
Statistical Analysis
Sex and racial distributions of Pneumocystis patients and controls were compared by using χ2 tests of equality of proportions. Means and standard deviations were calculated to compare the center and spread of age and continuous parameters measured at enrollment (baseline). Mean values for Pneumocystis patients and controls were compared and tested for equality by using unpaired t tests with adjustment for unequal variances, either on the original or logarithmic scale. Geometric means and geometric standard deviations were calculated when data were approximately lognormally distributed, as judged by Shapiro-Wilks tests and visual assessment. Quantile-quantile plots of serum antibody levels showed that their distributions were approximately lognormal, except for larger numbers of values at the lower quantiles. Pearson product moment correlations among clinical parameters and Msg fragments were obtained. Analyses of trends in mean values of each outcome were performed for all patients enrolled at baseline during either study period and followed up at least once at weeks 1–2, 3–4, or 5–6. Sparse data at later follow-up weeks precluded their inclusion in the regression analyses because of instability of parameter estimates. In addition, the numbers of patients observed at baseline and each follow-up time were too small to provide meaningful inferences from analysis.
Two stratifications of Pneumocystis patients were investigated with respect to trends and mean levels of Msg fragments: patients with CD4+ counts above and below the median value (≈50 cells/μL) and patients with and without a history of pneumocystosis. The analysis provided comparisons between means for patient categories at each follow-up week, as well as differences in patterns of change over time between patient groups. We obtained p values that compared mean values between periods and levels of CD4+ count or history of Pneumocystis pneumonia by using t tests with Tukey's adjustment for multiple comparisons. Patient-specific deviations from group means were included in the analysis as a random effect, which allowed the correlation between repeated measurements on the same patient over time to be included in between- and within-patient variance estimates. A p value <0.05 (2-tailed) was considered significant, unless stated otherwise. Analyses were performed by using the SAS procedure PROC MIXED (SAS for Windows version 9.2, SAS Institute Inc., Cary, NC, USA)
Demographic and Clinical Characteristics
No significant differences were seen between the 80 Pneumocystis patients and the 41 control patients with other causes of pneumonia in terms of sex, race, or age (Table 1). Baseline serum albumin levels, arterial blood gas measurements, and the proportion of patients who required mechanical ventilation were also similar, which indicates that the overall general health of patients and the severity of pneumonia in these 2 groups were also comparable. In addition, the proportion of patients with prior pneumocystosis was similar in both groups. However, the Pneumocystis patients had more advanced or more poorly controlled HIV infection than did the controls. The P. jirovecii patients had a significantly lower geometric mean CD4+ count and a significantly higher mean plasma HIV RNA level than did control patients. Patients with pneumocystosis also exhibited a significantly higher mean serum lactate dehydrogenase (LDH) level than did controls; this finding is consistent with the observation that elevated serum LDH is a nonspecific indicator of PCP.
Baseline and Sequential Serum Antibody Levels to MsgC
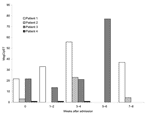
At the time of hospital admission for pneumonia, geometric mean serum antibody levels to MsgC in Pneumocystis patients and controls were similar (Table 1). Forty-one of 80 patients with PCP had >1 convalescent-phase serum specimen drawn in the first 6 weeks after hospital admission. The total number of patient visits was 62. Patients observed at weeks 3–4 (n = 19) had a higher mean serum antibody level to MsgC than the average level of all patients at baseline (n = 80) and patients observed at weeks 1–2 (n = 25) and weeks 5–6 (n = 18). Differences were significant, as determined by t statistics comparing group means, adjusted for paired comparisons (p<0.01 to p<0.001) (Table 2). Mean antibody levels at subsequent time points were 3.7 at 5–6 weeks, 4.0 at 7–8 weeks, and 3.0 at 9–12 weeks (data not shown).
Analysis of serum antibody levels in individual patients showed different patterns of reactivity (Figure). Eleven (58%) of the 19 patients studied at 3–4 weeks had an increase in their antibody levels, ranging from 1.4- to 22-fold above baseline levels. To determine if the rise in serum antibodies in the Pneumocystis patients at 3–4 weeks was specific for P. jirovecii or part of a broader increase in antibody reactivity, we examined the changes in antibody levels to TT in these 19 patients. The geometric mean antibody levels of 80 U at baseline and 117 U at 3–4 weeks were not significantly different.
Predictors of Serum Antibody Response to MsgC among Pneumocystis Patients
We examined clinical characteristics associated with an increase in antibody levels. No relationship was found between antibody level and sex, race, age, HIV viral load, serum albumin level, oxygenation, LDH level, or use of mechanical ventilation. CD4+ cell count (analyzed as CD4+ count <50 cells/μL or >50 cells/μL) was significantly associated with the ability to generate an increase in antibody levels (Table 2). Patients with CD4+ counts <50 cells/μL exhibited no significant changes in antibody levels to MsgC at any time point. In contrast, patients with CD4+ counts >50 cells/μL had a rise in mean antibody level, which peaked at 30.4 at 3–4 weeks and which was significantly higher than antibody levels at baseline (p<0.001) and at 5–6 weeks (p<0.001). The mean antibody level at 3–4 weeks in these patients was also significantly higher than the corresponding level in patients with <50 CD4+ cells (30.4 vs. 6.3, p<0.03).
The lack of a history of pneumocystosis also seemed to influence antibody response but not baseline antibody level. The mean antibody level to MsgC in the 59 patients with first episode of PCP was 4.1; this value was not significantly different from the mean value of 4.2 in the 19 patients with recurrent pneumocystosis (Table 3). Sequential antibody responses were compared in PCP patients whose episode of pneumonia was their first experience with the disease versus those whose pneumonia was a recurrent bout of the disease. Antibody levels in naive patients increased and reached a peak mean value of 17.9 at 3–4 weeks compared to 4.1 at baseline (p<0.001), whereas patients with a history of P. jirovecii pneumonia experienced no increase. Because the number of patients was low, we could not analyze the combined effects of PCP history and CD4+ cell count.
Baseline and Sequential Serum Antibody Levels to MsgA and MsgB
The baseline geometric mean serum antibody level to MsgA of 5.6 in PCP patients was not significantly different from the mean level of 3.7 in the controls (Table 1). Patients with pneumocystosis exhibited a different pattern of antibody response to MsgA than to MsgC in that the mean peak antibody levels to MsgA at 1–2 weeks and 3–4 weeks were similar (Table 4). However, none of the differences in antibody levels at different time points reached significance. In addition, no significant differences were found in the antibody levels at different time points in patients with and without a history of pneumocystosis or in patients with CD4+ cell counts <50 cells/μL or patients with CD4+ counts >50 cells/μL (data not shown).
No significant difference in baseline geometric mean serum antibody levels to MsgB was seen in patients with PCP and controls (Table 1). No significant differences were seen in antibody levels related to different time points, CD4+ counts, or history of pneumocystosis (data not shown).
Recombinant antigens derived from P. jirovecii have begun to attract attention as possible reagents for analyzing antibodies to Pneumocystis in humans (14–19). We have previously reported that HIV-positive, PCP-positive patients in Cincinnati had a significantly higher frequency and level of serum antibodies to MsgC than did HIV-positive, PCP-negative patients (17,18); this difference was not found with MsgA or MsgB. These patients were selected on the basis of a history of pneumocystosis and were clinically healthy. The present pilot study has extended these observations to HIV-positive patients hospitalized with acute pneumonia due to P. jirovecii and other causes (controls) in San Francisco. The goal of the first part of this study was to determine if baseline antibody levels to the 3 Msg fragments in PCP patients differed from those in the controls. Data showed that the geometric mean antibody levels to MsgC, MsgA, and MsgB were similar in both groups.
In the second part of this study, we analyzed the sequential changes in antibody levels to the 3 Msg fragments in P. jirovecii patients after treatment and recovery from pneumocystosis. The results showed a significant rise in mean antibody levels to MsgC that reached a peak at 3–4 weeks. In contrast to MsgC results, no significant changes in antibody levels to MsgA or MsgB occurred at any time point. The pattern of antibody reactivity to these Msg fragments also differed to some degree from the pattern of reactivity to MsgC. Taken together, these data suggest that MsgC is the best Msg fragment to use to analyze antibody responses in this population of HIV-positive patients with active Pneumocystis pneumonia.
An antibody rise by 3–4 weeks occurred in 58% of the Pneumocystis patients we studied. Of the potential host factors that could affect antibody responses, we were most interested in CD4+ cells, HIV RNA level, and previous history of pneumocystosis. Patients with CD4+ cell counts <50 cells/μL did not mount an antibody response, whereas patients with CD4+ counts >50 cells/μL that peaked 3–4 weeks after diagnosis showed a vigorous antibody response. In contrast to CD4+ count, mean viral load in P. jirovecii patients was not associated with an increase in antibody levels. Previous reports have shown that CD4+ cells and HIV itself affect antibody responses in HIV-positive patients, which can be reversed by highly active antiretroviral therapy (HAART) (22–24). In HIV-positive patients who are severely immunocompromised and have experienced opportunistic infections such as pneumocystosis, however, this immune reconstitution may be incomplete (25). Perhaps some who did not respond to treatment fall into this category, but we did not have information about HAART use and immune reconstitution in this cohort.
Our data showed that pneumocystosis patients with or without a previous episode of the disease had similar baseline antibody levels to MsgC; however, patients who experienced their first bout of PCP had better antibody responses after recovery from the disease. Those who were experiencing a recurrent bout of pneumocystosis may have been unable to mount an antibody response to previous episodes and remained at risk. CD4+ cell count or differences in the treatment of pneumocystosis might also play a role in the ability of patients without a previous history of pneumocystosis to mount an antibody response, but our analysis was not powered to analyze multiple factors simultaneously.
Comparison of our results with previous work is complicated by the fact that these earlier studies were performed in the pre-HAART era and involved crude or native antigens. Analysis of whether a detectable antibody response to Pneumocystis antigens could develop in HIV-positive patients who recovered from pneumocystosis produced conflicting results (9–11,26–30). One report showed a rise in antibodies to native Msg in 43% of HIV-positive patients; host factors such as CD4+ count or pO2 could not distinguish responders from nonresponders (13). A more recent study that used different recombinant Msg constructs than we used found that HIV-negative, immunocompromised patients who recovered from pneumocystosis had increased antibody levels, but HIV-positive patients who recovered had lower levels and poor antibody responses (16). One factor that may contribute to these disparate results is antigenic variation, which involves differences in the Msg constructs themselves. We have developed several variants of our current Msg construct and found that they differ in their ability to distinguish among HIV-positive patients who have and not have had pneumocystosis (unpub. data). PCP patients exhibit greater reactivity with multiple MsgC clones than do patients without PCP or blood donors, but whether the antigens that are recognized are cross-reactive or clone specific is unclear. Further studies to identify broadly reactive MsgC antigens associated with recovery from PCP, as well as proteins (e.g., Kex1) encoded by single-copy genes, would be of interest (19).
The sequential serologic results reported here, which were obtained from a limited number of patients, provide the basis for a large, prospective, multisite study of sequential antibody responses to MsgC in HIV-positive patients who have pneumonia caused by P. jirovecii and other organisms. Serologic surveys need to be performed in different areas to determine which Msg fragment is the predominant fragment recognized by HIV-positive patients and healthy persons. Standardizing Msg antigen preparations, ELISA conditions, and data analysis would be helpful so that serologic results could be reproduced in different laboratories.
The development of a successful serologic test for Pneumocystis infection will have clinical and epidemiologic applications. Serologic tests with MsgC might be used in the diagnosis of pneumonia in situations (e.g., in developing countries) in which a specific cause cannot be established; in cohort studies to investigate the relationship of serum antibody levels and the risk for, and recovery from, Pneumocystis pneumonia; in seroepidemiologic surveys and outbreaks of pneumocystosis; and in investigating the pathogenic role for P. jirovecii in chronic lung diseases in which colonization of the organism has been detected (5).
Dr Daly is a senior research associate at the University of Cincinnati. He joined the university in 1998 and has been studying immune responses to Pneumocystis since then. His research interests include viral and tumor immunology.
Acknowledgment
Support for this study was provided by Department of Veterans Affairs (K.D., P.W.) and the National Institutes of Health R03 AI570636 (K.D.), K23 HL072117 (L.H.), K23 HL072837 (A.M.), NIH/NCRR K12 RR01175584-01 (K.C.), and AACTG.21.Pc, U01 AI25467, R01 AI AI06492, F33 AI06527 (P.W.).
References
- Redhead SA, Cushion MT, Frenkel JK, Stringer JR. Pneumocystis and Trypanosoma cruzi: nomenclature and typifications. J Eukaryot Microbiol. 2006;53:2–11. DOIPubMedGoogle Scholar
- Beard CB, Carter JL, Keely SP, Huang L, Pieniazek NJ, Moura IN, Genetic variation in Pneumocystis carinii isolates from different geographic regions: implications for transmission. Emerg Infect Dis. 2000;6:265–72. DOIPubMedGoogle Scholar
- Crothers K, Beard CB, Turner J, Groner G, Fox M, Morris A, Severity and outcome of HIV-associated Pneumocystis pneumonia containing Pneumocystis jirovecii dihydropteroate synthase gene mutations. AIDS. 2005;19:801–5. DOIPubMedGoogle Scholar
- Vargas SL, Hughes WT, Santolaya ME. Search for primary infection by Pneumocystis carinii in a cohort of normal, healthy infants. Clin Infect Dis. 2001;32:855–61. DOIPubMedGoogle Scholar
- Morris A, Sciurba FC, Githaiga A, Lebedeva I, Elliott WM, Hogg JC, Association of chronic obstructive pulmonary disease severity and Pneumocystis colonization. [Medline]. Am J Respir Crit Care Med. 2004;170:408–13. DOIPubMedGoogle Scholar
- Gigliotti F, Hughes WT. Passive immunoprophylaxis with specific monoclonal antibody confers partial protection against Pneumocystis carinii pneumonitis in animal models. J Clin Invest. 1988;81:1666–8. DOIPubMedGoogle Scholar
- Garvy BA, Wiley JA, Gigliotti F, Harmsen AG. Protection against Pneumocystis carinii pneumonia by antibodies generated from either T helper 1 or T helper 2 responses. Infect Immun. 1997;65:5052–6.PubMedGoogle Scholar
- Zheng M, Shellito JE, Marrero L, Zhong Q, Julian S, Ye P, CD4+ T cell–independent vaccination against Pneumocystis carinii in mice. J Clin Invest. 2001;108:1469–74.PubMedGoogle Scholar
- Smulian AG, Walzer PD. Serological studies of Pneumocystis carinii infection. In: Walzer PD, editor. Pneumocystis carinii pneumonia. New York: Marcel Dekker; 1994. p. 141–51.
- Walzer PD. Immunological features of Pneumocystis infection. In: Cushion MT, Walzer PD. Pneumocystis pneumonia. New York: Marcel Dekker; 2005. p. 451–78.
- Peglow SL, Smulian GA, Linke MJ, Crisler J, Phair JWM, Gold J, Serologic responses to specific Pneumocystis carinii antigens in health and disease. J Infect Dis. 1990;161:296–306. DOIPubMedGoogle Scholar
- Smulian AG, Sullivan D, Linke MJ, Halsey N, Quinn T, MacPhail AP, Geographic variation in the humoral response to Pneumocystis carinii. J Infect Dis. 1993;167:1243–7. DOIPubMedGoogle Scholar
- Lundgren B, Lundgren JD, Nielsen T, Mathiesen L, Nielsen JO, Kovacs JA. Antibody responses to a major Pneumocystis carinii antigen in human immunodeficiency virus–infected patients with and without P. carinii pneumonia. J Infect Dis. 1992;165:1151–5. DOIPubMedGoogle Scholar
- Garbe TR, Stringer JR. Molecular characterization of clustered variants of genes encoding major surface antigens of human Pneumocystis carinii. Infect Immun. 1994;62:3092–101.PubMedGoogle Scholar
- Mei Q, Turner RE, Sorial V, Klivington D, Angus CW, Kovacs JA. Characterization of major surface glycoprotein genes of human Pneumocystis carinii and high-level expression of a conserved region. Infect Immun. 1998;66:4268–73.PubMedGoogle Scholar
- Bishop LR, Kovacs JA. Quantitation of anti-Pneumocystis jiroveci antibodies in healthy persons and immunocompromised patient. J Infect Dis. 2003;187:1844–8. DOIPubMedGoogle Scholar
- Daly KR, Fichtenbaum C, Tanaka R, Linke MJ, Obert R, Thullen T, Serologic responses to epitopes of the major surface glycoprotein of Pneumocystis jiroveci differ in human immunodeficiency virus–infected and uninfected persons. J Infect Dis. 2002;186:644–51. DOIPubMedGoogle Scholar
- Daly KR, Koch J, Levin L, Walzer PD. Enzyme-linked immunosorbent assay and serologic responses to Pneumocystis jiroveci. Emerg Infect Dis. 2004;10:848–54.PubMedGoogle Scholar
- Kutty G, Kovacs JA. A single-copy gene encodes Kex1, a serine endoprotease of Pneumocystis jiroveci. Infect Immun. 2003;71:571–4. DOIPubMedGoogle Scholar
- Theus SA, Andrews RP, Stelle P, Walzer PD. Adoptive transfer of lymphocytes sensitized to the major surface glycoprotein of Pneumocystis carinii confers protection in the rat. J Clin Invest. 1995;95:2587–93. DOIPubMedGoogle Scholar
- Huang L, Stansell JD. AIDS and the lung. Med Clin North Am. 1996;80:775–801. DOIPubMedGoogle Scholar
- Malaspina A, Moir S, Orsega SM, Vasquez J, Miller NJ, Donoghue ET, Compromised B cell responses to influenza vaccination in HIV-infected individuals. J Infect Dis. 2005;191:1442–50. DOIPubMedGoogle Scholar
- De Milito A. B lymphocyte dysfunctions in HIV infection. Curr HIV Res. 2004;2:11–21. DOIPubMedGoogle Scholar
- Jacobson MA, Khyam-Bashi H, Martin JN, Black D, Ng V. Effect of long-term highly active antiretroviral therapy in restoring HIV-induced abnormal B-lymphocyte function. J Acquir Immune Defic Syndr. 2002;31:472–7. DOIPubMedGoogle Scholar
- D'Amico R, Yang Y, Mildvan D, Evans SR, Schnizlein-Bick CT, Hafner R, Lower CD4+ T lymphocyte nadirs may indicate limited immune reconstitution in HIV-1 infected individuals on potent antiretroviral therapy: analysis of immunophenotypic marker results of AACTG 5067. [Medline]. J Clin Immunol. 2005;25:106–15. DOIPubMedGoogle Scholar
- Burns SM, Read JA, Yap PL, Brettle RP. Reduced concentrations of IgG antibodies to Pneumocystis carinii in HIV-infected patients during active Pneumocystis carinii infection and the possibility of passive immunization. J Infect. 1990;20:33–9. DOIPubMedGoogle Scholar
- Hofmann B, Odum N, Cerstoft J, Platz P, Ryder LP, Svjgaard A, Humoral response to Pneumocystis carinii in patients with acquired immunodeficiency syndrome and in immunocompromised homosexual men. J Infect Dis. 1985;152:838–40. DOIPubMedGoogle Scholar
- Hofmann B, Nielsen PB, Odum N, Gerstoft J, Platz P, Ryder LP, Humoral and cellular responses to Pneumocystis carinii, CMV, and herpes simplex in patients with AIDS and in controls. Scand J Infect Dis. 1988;20:389–94. DOIPubMedGoogle Scholar
- Elvin K, Bjorkman A, Heurlin N, Eriksson BM, Barkholt L, Linder E. Seroreactivity to Pneumocystis carinii in patients with AIDS verus other immunosuppressed patients. Scand J Infect Dis. 1994;26:33–40. DOIPubMedGoogle Scholar
- Laursen AL, Andersen PL. Low levels of IgG antibodies against Pneumocystis carinii among HIV-infected patients. Scand J Infect Dis. 1998;30:495–9. DOIPubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 12, Number 8—August 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Peter D. Walzer, Research Service (151), VA Medical Center, 3200 Vine St, Cincinnati, OH 45220, USA
Top