Volume 13, Number 10—October 2007
Dispatch
Exposure to Wild Primates among HIV-infected Persons
Cite This Article
Citation for Media
Abstract
HIV-1 is an immunosuppressive pathogen. Our behavioral data for 191 HIV-1–infected rural Cameroonians show frequent exposure to nonhuman primates through activities such as hunting and butchering. Immunosuppression among persons exposed to body fluids of wild nonhuman primates could favor the process of adaptation and subsequent emergence of zoonotic pathogens.
Worldwide, ≈1% of the population is immunodeficient. Although immunodeficiency has numerous causes, such as malnutrition or iatrogenic medical therapies for cancer and organ transplantation, the most significant factor globally is HIV-1 infection (1). In 2006, ≈40 million persons were infected with HIV-1, and >50% were in sub-Saharan Africa, where AIDS caused 2.1 million deaths (2).
Immunodeficiency resulting from HIV-1 infection renders the host susceptible to infections usually controlled by cellular immunity through unrelenting loss of CD4+ T-helper lymphocytes. This susceptibility predisposes affected persons to common disease-causing pathogens such as Mycobacterium tuberculosis, Salmonella spp., Coccidioides spp., and Histoplasma spp. Other pathogens that are rarely pathogenic for immunocompetent persons, such as Cytomegalovirus, human herpesvirus-8, Pneumocystis spp., Cryptococcus spp., and M. avium complex, also become common causes of disease.
HIV-1–induced immunosuppression has also been proposed as a factor affecting the global emergence and reemergence of diseases (1,3). Among emerging infectious diseases in humans, ≈75% are caused by zoonotic pathogens (4), highlighting the potentially important risk for zoonotic exposures for HIV-1–infected populations. Central African forests, where hunting and butchering nonhuman primates are common practices, provide a ripe environment for zoonotic transmission (5). These areas have fostered human acquisition of Ebola (6,7), monkeypox (8), simian immunodeficiency viruses (9), simian foamy viruses (10), and primate T-lymphotropic viruses (11). Because HIV-1 infection is epidemic in Africa, persons involved in hunting and butchering of wild animals (including nonhuman primates) are possibly HIV-1–infected and thus at risk for successful infection with novel zoonotic viral infections. Additionally, HIV-1–induced immunosuppression in the wider community poses an additional risk for secondary transmission that could facilitate early viral adaptation to humans (12).
As part of a community-based HIV-1 prevention campaign, February 2001–January 2003, we collected oral questionnaire data about basic demographics and behavior associated with exposure to the blood or body fluids of wild animals. In addition, blood samples were collected and transported to a central laboratory for HIV testing. We present behavioral data pertaining to animal exposures of HIV-1–infected persons in 17 rural villages in Cameroon (5). These are key sites for the emergence of nonhuman primate retroviruses because of the high levels of human contact with wild nonhuman primates (5) and cross-species transmission of simian foamy virus (10) and primate T-lymphotropic viruses (11).
Study participation was voluntary and performed under a protocol approved by the Johns Hopkins Committee for Human Research, the Cameroon National Ethical Review Board and the HIV Tri-Services Secondary Review Board. A single project assurance was obtained from the Cameroonian Ministry of Health and accepted by the National Institutes of Health Office for Protection from Research Risks.
HIV testing was performed by using an ELISA/Western blot algorithm. The ORTHO HIV1/2 (ORTHO Clinical Diagnostics Gmbh, Neckargemünd, Germany) ELISA was used as the screening test, and the HIV Blot 2.2 (Genelabs Diagnostics, Singapore) Western blot assay was used for confirmation.
Complete questionnaire data and plasma samples were collected from 3,955 persons, of whom 46.3% were female and 53.7% were male. Age range was 16–97 years (42% 16–30, 27% 31–45, 21% 46–60, and 10% >60 years). Screening for HIV-1 infection found 191 seropositive persons (prevalence 4.8% overall, 1.9%–16.3% from the 17 sites), of whom 60.2% were female and 39.8% were male. No persons were HIV-2 seropositive.
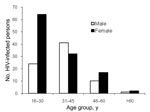
The HIV-1–infected persons were examined in greater detail. Within the younger age group (16–30 years), women were overrepresented; among persons >30 years of age, the number of infected men and women was similar (Figure 1). Of the HIV-1–infected persons, 89.0% reported having lived in a major city or another country, compared with 82.8% of the HIV-negative study population). Agricultural activities were reported as daily activities by 46.6% of the HIV-1–positive persons; household activities, by 22.0%; and fishing, by 13.6%. Hunting was reported as a daily activity by 12.6%, and contact with wild animals was reported by an even higher proportion. Among HIV-1–positive persons, 79.6% reported butchering wild animals (as many as 20× per month), 26.2% reported hunting wild animals (also as many as 20× per month), 12.6% reported having kept a wild animal as a pet, and 95.8% reported eating wild animals (Figure 2).
HIV-1–infected persons had significant contact with nonhuman primates; hunting of these species was reported by 11.0%. Monkeys were hunted up to 10× per month (median 3× per month); chimpanzees and gorillas were hunted less frequently (always <1× per month). In terms of butchering, 55.5% reported butchering nonhuman primates: monkeys <10× per month (median 1× per month), chimpanzees 2× per month (median <1× per month), and gorillas <1× per month. Furthermore, 8.4% of HIV-1–infected persons reported keeping nonhuman primates as pets, and 83.8% reported eating nonhuman primates.
Other direct animal exposures were reported by HIV-1–infected persons (Table); bites or scratches from wild animals were reported by 12.0% and from nonhuman primates by 2.6%. Although 4.7% of persons reported having received injuries during hunting and butchering, none reported having received injuries during hunting or butchering of nonhuman primates. However, 1.7% of the rural population in this area reports such injuries (5).
These data demonstrate an overlap of areas where HIV-1 is epidemic and areas where human-nonhuman primate contact is common. This overlap is cause for concern because humans and nonhuman primates share susceptibility to a range of pathogens, and the potential for successful cross-species transmission from nonhuman primates to humans is considered great (5). Access to treatment for HIV-1 infection is improving but is limited in remote central African communities; progressive disease and immunosuppression develop in most persons in these areas. Exposure of immunocompromised persons to nonhuman primates poses ongoing opportunities for zoonotic viruses to leap to humans, and the high concentration of other immunocompromised hosts offers an increased risk for secondary transmission and adaptation to humans. The emergence of HIV-1 is an example of such a process; data suggest several abortive nonhuman primate-to-human transmission events before eventual establishment of the HIV pandemic. This foothold gained by HIV-1 may now offer a boost for other pathogens to enter the human population. Moreover, the prevalence of HIV-1 in rural areas is lower than that in adjacent urban communities in Cameroon (13) and may increase. Such circumstances are not limited to central Africa; recent reports from Asia have demonstrated the risk for zoonotic infections with nonhuman primate viruses (14,15). And although nonhuman primates may present particular risks for disease emergence, HIV-associated immunosuppression likely increases the risk for acquisition, adaptation, and emergence of zoonoses infecting other animals that are hunted extensively in these communities (Figure 2), such as monkeypox and hantaviruses in rodents and Lyssavirus in bats.
The risk for emergence of novel zoonotic infections in rural hunting communities should be considered in healthcare policy. Community health education and HIV/AIDS counseling should account for the fact that many persons in these communities rely on wild animals for food and household income. Targeted interventions could include culturally appropriate suggestions for avoiding handling or butchering of wild animals, such as developing alternative food sources, or taking precautions if such activities are necessary. Reducing the prevalence of HIV-1–induced immunosuppression through prevention and treatment and minimizing zoonotic exposures will be crucial for preventing future outbreaks of novel viral pathogens in humans.
Mr LeBreton is a wildlife ecologist who works with the Johns Hopkins Cameroon Program. His research interests include the interactions among the fields of public health, ecology, and conservation.
Acknowledgments
We thank the staff of the Johns Hopkins Cameroon Program and the US Embassy in Yaoundé for assistance and the Government of Cameroon for permission to undertake this study.
This work was supported by the US Military HIV Research Program, the National Institutes of Health (NIH) Fogarty International Center (International Research Scientist Development Award Grant no. 5 K01 TW000003-05 and AIDS International Training and Research Program Grant no. 2 D 43 TW000010-17-AITRP), the NIH Director’s Pioneer Award Program (grant no. DP1-OD000370), the W.W. Smith Charitable Trust, National Geographic Society Committee for Research and Exploration, the Johns Hopkins Center for AIDS Research, and the Johns Hopkins Center for a Livable Future.
References
- Morens DM, Folkers GK, Fauci AS. The challenge of emerging and re-emerging infectious diseases. Nature. 2004;430:242–9. DOIPubMedGoogle Scholar
- Joint United Nations Program on HIV/AIDS and the World Health Organization. AIDS epidemic update: December 2006. Geneva: The Program and The Organization; 2006. p. 1–90. [cited 2007 July 12]. Available from http://www.unaids.org/en/hiv_data/epi2006
- Morris JG Jr, Potter M. Emergence of new pathogens as a function of changes in host susceptibility. Emerg Infect Dis. 1997;3:435–41. DOIPubMedGoogle Scholar
- Taylor LH, Latham SM, Woolhouse MEJ. Risk factors for human disease emergence. Philos Trans R Soc Lond B Biol Sci. 2001;356:983–9. DOIPubMedGoogle Scholar
- Wolfe ND, Prosser AT, Carr JK, Tamoufe U, Mpoudi-Ngole E, Ndongo Torimiro J, Exposure to nonhuman primates in rural Cameroon. Emerg Infect Dis. 2004;10:2094–9.PubMedGoogle Scholar
- Leroy EM, Rouquet P, Formenty P, Souquiere S, Kilbourne A, Froment JM, Multiple Ebola virus transmission events and rapid decline of central African wildlife. Science. 2004;303:387–90. DOIPubMedGoogle Scholar
- Rouquet P, Froment JM, Bermejo M, Yaba P, Délicat A, Rollin PE, Wild animal mortality monitoring and human Ebola outbreaks, Gabon and Republic of Congo, 2001–2003. Emerg Infect Dis. 2005;11:283–90.PubMedGoogle Scholar
- Jezek Z, Arita I, Mutombo M, Dunn C, Nakano JH, Szczeniowski M. Four generations of probable person-to-person transmission of human monkeypox. Am J Epidemiol. 1986;123:1004–12.PubMedGoogle Scholar
- Hahn BH, Shaw GM, De Cock KM, Sharp PM. AIDS as a zoonosis: scientific and public health implications. Science. 2000;287:607–14. DOIPubMedGoogle Scholar
- Wolfe ND, Switzer WM, Carr JK, Bhullar VB, Shanmugam V, Tamoufe U, Naturally acquired simian retrovirus infections in central African hunters. Lancet. 2004;363:932–7. DOIPubMedGoogle Scholar
- Wolfe ND, Heneine W, Carr JK, Garcia AD, Shanmugam V, Tamoufe U, Emergence of unique primate T-lymphotropic viruses among central African bushmeat hunters. Proc Natl Acad Sci U S A. 2005;102:7994–9. DOIPubMedGoogle Scholar
- Antia R, Regoes RR, Koella JC, Bergstrom CT. The role of evolution in the emergence of infectious diseases. Nature. 2003;426:658–61. DOIPubMedGoogle Scholar
- Anonymous . Health and demographic survey III. Report by Government of the Republic of Cameroon, 2004.
- Engel G, Hungerford LL, Jones-Engle L, Travis DA, Eberle R, Fuentes A, Risk assessment: a model for predicting cross-species transmission of simian foamy virus from macaques (M. fascicularis) to humans at a monkey temple in Bali, Indonesia. Am J Primatol. 2006;68:934–48. DOIPubMedGoogle Scholar
- Jones-Engel L, Engel G, Schillaci MA, Rompis ALT, Putra A, Suaryana K, Primate-to-human retroviral transmission in Asia. Emerg Infect Dis. 2005;11:1028–35.PubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 13, Number 10—October 2007
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Nathan D. Wolfe, Department of Epidemiology, UCLA School of Public Health, 650 Charles E. Young Dr S, CHS 71-279B, Box 177220, Los Angeles, CA 90095-1771, USA;
Top