Volume 13, Number 11—November 2007
Synopsis
Literature Review and Case Histories of Histoplasma capsulatum var. duboisii Infections in HIV-infected Patients
Cite This Article
Citation for Media
Abstract
African histoplasmosis caused by Histoplasma capsulatum var. duboisii is an invasive fungal infection endemic in central and west Africa. Most of its ecology and pathogenesis remain unknown. H. capsulatum var. capsulatum is an AIDS-defining opportunistic infection in HIV-infected patients who are living in or have traveled to histoplasmosis-endemic areas. In contrast, reports concerning African histoplasmosis during HIV infection are rare, although both pathogens coexist in those regions. We report 3 cases of imported African histoplasmosis diagnosed in France in HIV-infected patients and a literature review on similar cases.
Human histoplasmosis is caused by 2 varieties of Histoplasma. The most common variety worldwide is H. capsulatum var. capsulatum, which has been reported from many disease-endemic areas where HIV infection is prevalent. Histoplasmosis is more frequent in the United States (Ohio and Mississippi River valleys), but it is not unusual in other parts of the world, such as Africa (1,2). In the western and central regions of sub-Saharian Africa, H. capsulatum var. capulatum coexists with another variety, H. capsulatum var. duboisii, whose ecology and pathogenesis remain almost unknown. Cases due to H. capsulatum var. duboisii are scarce in Europe, and all are imported (3).
Before the era of highly-active antiretroviral therapy (HAART), the prevalence of H. capsulatum var. capsulatum infections reached up to 30% of HIV-infected patients in hyperendemic areas of the southeastern part of the United States (4). The infection occurs more often in patients with a CD4 count <50/mm3 and is usually disseminated. For unknown reasons, although HIV infection and H. capsulatum var. duboisii coexist in Africa, this coinfection remains rare (5). We report 3 imported cases of the potentially emerging histoplasmosis due to H. capsulatum var. duboisii diagnosed in France during the course of HIV infection and a literature review on similar cases.
A 37-year-old man from the Democratic Republic of Congo, who had lived in France since 1980, was infected by HIV-1 since 1987. He was admitted to the hospital in 1992 because of a fever of unknown origin. His physical examination showed a left axillary tumefaction 2 inches in diameter. This mass had already been explored 5 months before. At that time, histopathologic examination disclosed a necrotizing lymphadenitis with epithelioid cells but without caseum. No microorganism was seen after Ziehl, periodic acid–Schiff, and Grocott stainings, but culture was not performed. When the patient was hospitalized in 1992, laboratory tests showed an erythrocyte sedimentation rate of 104 mm, fibrin 4.5 g/L, C-reactive protein 74 mg/L, and a CD4 count of 100/mm3 (9%). The adenopathy was surgically removed. Histopathologic examination showed necrosis and large yeasts, and culture grew Histoplasma sp. on day 12. Anti-Histoplasma antibody detection was negative. No other lymph node, bone, skin, or bone marrow involvement was found.
Itraconazole treatment was started (400 mg/d), but it was switched to amphotericin B (1 mg/kg/d) after 3 weeks because local symptoms persisted. The total dose of amphotericin B was 1,200 mg. Itraconazole (400 mg/d) was then restarted for 1 year. The clinical course was satisfactory, so itraconazole was lowered to 200 mg/d for 3 years. The patient died in 1995 of HIV-related encephalitis despite antiretroviral therapy, including nucleoside reverse transcriptase inhibitors, without recurrence of histoplasmosis.
A 41-year-old man from the Democratic Republic of Congo, who had lived in France since 1981, was infected by HIV-1 in 1994. He received AZT (3′-azido-3′-deoxythymidine) and diethyldithiocarbamate (ddC) when Pneumocystis jiroveci pneumonia was diagnosed in December 1995. Despite the introduction of HAART in 1996 (AZT, lamivudine [3TC], and ritonavir), his CD4 count remained <50/mm3. In mid-1996, nodular cutaneous lesions, a right cervical adenopathy, and a right Bell’s palsy developed. Direct examination of the lymph node showed numerous yeasts with a typical lemon shape and a narrow budding, suggestive of H. capsulatum var. duboisii. The culture grew Histoplasma sp.
The patient did not respond to itraconazole (400 mg/d). After 1 month, he was given conventional amphotericin B (1 mg/kg/d); severe renal insufficiency developed within 8 days. Treatment was switched to liposomal amphotericin B (3 mg/kg/d) with a dramatic improvement of the symptoms and partial regression of the renal insufficiency. Immune reconstitution inflammatory syndrome also developed; its characteristics were reported previously (6). After 1 month, treatment was switched to itraconazole, 400 mg/d, for long-term therapy. Because of a persistent low CD4 count despite undetectable viral load, the patient benefited from several courses of interleukin-2 (IL-2) therapy, which allowed a marked and sustained increase of the CD4 count. Itraconazole was stopped in 1996. His condition remains stable 11 years later, and no recurrence of histoplasmosis has been observed.
A 2-year-old girl from the Democratic Republic of Congo was referred to the hospital in June 2001 for a fever of unknown origin. (She arrived in France in 2001 at the age of 18 months.) Investigations showed Escherichia coli pyelonephritis. HIV-1 serologic test results were positive, and her CD4 count was 45/mm3. HAART was started quickly, combining AZT, 3TC, and nelfinavir. This treatment resulted in a decrease in, but not elimination of, the viral load and a CD4 count <200/mm3 despite appropriate nelfinavir serum concentrations.
In August 2001, a frontal swelling appeared, associated with fever and generalized weakness. Direct examination of a skin biopsy specimen showed large, lemon-shaped yeasts suggestive of H. capsulatum var. duboisii. Culture of this specimen grew Histoplasma sp. Diffuse bone involvement (several lytic lesions of the right humerus, left ulna, both tibias, right fibula) was found on radiographs. Culture of the buffy coat was concomitantly positive for Fusarium verticillioides. No involvement of the lungs or lymph nodes was found.
Treatment with liposomal amphotericin B was started but switched to itraconazole after 1 month. Fever relapsed shortly thereafter, as well as the facial tumefaction. Radiographic examination showed several lesions of the skull, and a bone biopsy demonstrated large yeasts on direct examination. Amphotericin B was restarted for 4 months. The patient’s status improved dramatically, and the treatment was switched to fluconazole until September 2003. She did not experience any relapse, and antifungal prophylaxis was discontinued because of the improvement of her immunologic status (CD4 count >200/mm3 and undetectable viral load). In July 2007, she is doing well with a CD4 count of 700/mm3 and a still-undetectable viral load.
H. capsulatum var. duboisii is also known as African histoplasmosis because it has only been described on that continent, mostly in central and western Africa. The prevalence of histoplasmosis due to variety duboisii has not been established in countries in these regions in HIV-negative patients. Fewer than 300 cases are reported in the literature (7). The reason it remains rare, despite the major HIV pandemic in Africa, is unknown. Potential explanations are that patients die from other causes before histoplasmosis develops (8) or that variety capsulatum is more virulent than variety duboisii. This situation is reminiscent of Cryptococcus gattii and C. neoformans. C. gattii is rarely identified in HIV-infected patients, in contrast with C. neoformans, whereas both are present in the environment in countries where the prevalence of HIV infection is high (9). However, variety capsulatum is frequent in Africa. No data on the relative frequency of those 2 varieties has been published. Skin reaction to histoplasmin in histoplasmosis-endemic areas showed a 3% prevalence (10), but variety capsulatum and variety duboisii were not able to be differentiated. Higher prevalence (≈35%) was found in rural populations, especially among farmers, traders, and cave guides (11). Histoplasmosis due to variety duboisii may be misdiagnosed in those areas because of physicians’ lack of awareness.
The pathogenesis of African histoplasmosis remains unclear. The main route of acquisition could be airborne contamination from the soil, rarely direct inoculation. Variety duboisii is classically associated with cutaneous lesions (nodules, ulcers) and osteolytic bone lesions, especially affecting the skull, ribs, and vertebrae (Table 1) (12,13). Histopathologic examination shows granuloma with necrosis and suppuration. Disseminated disease is not uncommon and can involve every organ; however, the heart and central nervous system are unusual locations. A total of 17 cases have been reported thus far among HIV-infected patients, including the 3 cases described here (14–19). An additional case has been reported, but without detailed description, in a Ugandan patient diagnosed in Japan (20). Among the well-described cases (Table 1), most involved patients with poor immunologic status (mean CD4 count 55/mm3), which also occurs with histoplasmosis due to variety capsulatum (21). Most patients had disseminated infections, and only 4 patients died. The prognosis of disseminated infection in this context is close to the 20% mortality rate reported for disseminated histoplasmosis due to variety capsulatum among AIDS patients (21), but the few number of cases does not allow us to extrapolate the mortality rate related to variety duboisii. Epidemiologic information, clinical manifestations, and outcomes of immunocompetent versus HIV-infected patients infected with variety duboisii are compared in Table 2 (13). These data confirm the tropism of variety duboisii for lymph nodes, skin, and bones. It is noteworthy that the disease is often located in the lungs in HIV-negative patients, whereas HIV-infected patients have substantially more disseminated disease. The latter finding may be explained by immunodepression, poor access to the healthcare system for HIV-infected persons in Africa, and late diagnoses of histoplasmosis.
Despite its rarity, African histoplasmosis should be kept in mind as a diagnosis in Africa-born patients or travelers to sub-Saharan West and central Africa who have compatible signs or symptoms, even if they are HIV-infected, because the saprophytic phase of this dimorphic fungus should be manipulated in a Biosafety Level 3 cabinet. The laboratory diagnosis is performed by direct examination and culture. Cultures of tissue samples or body fluids are made onto Sabouraud dextrose agar, incubated at 25°C; incubation could be prolonged for up to 6 weeks. The success rate depends on the extent of infection, the source of the sample, and the prompt processing of the sample.
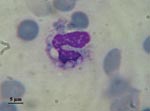
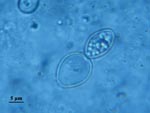
In addition to differences in clinical manifestations and epidemiology, the 2 varieties can be easily distinguished on observation of the yeast phases present in infected fresh or fixed tissues, whereas the saprophytic phase is identical. Variety capsulatum presents as small (3-μm) oval yeasts free or inside histocytes or macrophages (Figure 1), whereas yeasts of variety duboisii are large (7–15 μm), globose to ovoid, thick-walled, and typically lemon-shaped with a narrow budding (Figure 2). They are often seen in the cytoplasma of giant cells (1).
Diagnoses such as cryptococcosis and blastomycosis can be easily ruled out by direct examination or histopathology, but blastomycosis is unlikely in central and western African patients (22). The differential diagnosis is rarely difficult with cryptococcosis because of the shape and size of yeasts, presence of capsule, and lack of inflammation in the surrounding tissue. In any event, cryptococcal antigen testing and culture will easily ascertain the diagnosis.
Antigen detection in serum and urine is a sensitive test but has been developed for the variety capsulatum. It is validated in HIV-infected patients with disseminated diseases (8,23,24). H. duboisii is a cause of false-positive test results for antigen detection in urine. Antibody detection is useful for the retrospective diagnosis of histoplasmosis caused by variety capsulatum. Since variety duboisii antigens may cross-react with those of the variety capsulatum, serologic tests are potentially useful for diagnosis of African histoplamosis.
Although some PCR assays have been developed, they are not yet routinely used (25). Real-time and semi-nested PCR seem promising for the diagnosis of histoplasmosis due to variety capsulatum in blood and tissue samples (26–28). No PCR has yet been developed for variety duboisii, but a specific PCR assay could be helpful for this underdiagnosed disease.
Treatment of African histoplasmosis can be extrapolated from the guidelines of the Infectious Diseases Society of America established for histoplasmosis due to variety caspulatum (29). No clinical trial or efficacy studies have been performed for histoplasmosis due to variety duboisii, but as mortality rates are similar for the 2 species with the same management, the guidelines can be extrapolated to African histoplasmosis. In patients with AIDS, recommended therapy includes an intensive phase of 3 months with amphotericin B replaced by itraconazole (400 mg/d) for the severe forms, or itraconazole alone (600 mg/d for 3 days, then 400 mg/d) for mild forms. Fluconazole (800 mg/d) can be an alternative, but it has lower efficacy and a higher recurrence rate with isolates harboring higher MICs (30). Moreover, new azoles such as voriconazole require careful biologic and clinical monitoring when used for treating histoplasmosis in HIV-infected patients because of increased risk for in vitro resistance, especially in patients who had fluconazole (31). Nothing is known, however, about development of resistance for variety duboisii. Managing AIDS by HAART is an essential part of the treatment. The availability of HAART in Africa is increasing, but it may be absent in areas where histoplasmosis is endemic. This is a real concern for optimal management of such patients.
Maintenance therapy with itraconazole (200 mg or 400 mg/d) is recommended. Fluconazole (400 mg/d) should be avoided because of its reduced capacity to prevent relapses. However, as for many other opportunistic infections, maintenance therapy can be discontinued if the immunologic status of the patient improves, as described for case-patient 3. This patient’s prophylaxis was stopped 3 years ago, and she experienced no relapse and her CD4 count has always been >200/mm3. The stability of immune improvement has to be confirmed for several months before prophylaxis is stopped (32). Recent data suggest that the risk for relapse is rare after 12 months of treatment with a sustained immunologic improvement (CD4 >150/mm3) (33). However, in our experience based on the management of 20 cases of histoplasmosis due to variety duboisii in patients considered immunocompetent (13), relapses may be observed several years after the first episode. Thus, prolonged follow-up is mandatory for every patient with histoplasmosis due to variety duboisii.
Since HAART was introduced, the clinical and immunologic conditions of HIV-infected patients have dramatically improved, but physicians should now be aware of immune reconstitution inflammatory syndrome (IRIS) (34). As for many pathogens, both varieties of H. capsulatum can induce IRIS in HIV-infected patients, as recently reported by our group (6). The importance of the inflammatory reaction during IRIS contrasts with the mild one observed in the initial phase of the disease in severely immunocompromised patients and may require specific treatment.
Thus, histoplasmosis due to variety duboisii in HIV-infected patient remains a rare clinical entity but diagnosis should not be discounted because of the HIV status of the patient. Physicians working in Africa should be aware of H. capsulatum var. duboisii as a potentially emerging infection in HIV-infected patients.
Dr Loulergue is an infectious diseases fellow attending the Infectious Diseases Department of Necker-Enfants Malades University Hospital, Paris, France. His research interests include infectious diseases in immunocompromised hosts and development of vaccines.
References
- Wheat LJ, Kauffman CA. Histoplasmosis. [vii.]. Infect Dis Clin North Am. 2003;17:1–19. DOIPubMedGoogle Scholar
- Wheat LJ. Histoplasmosis: a review for clinicians from non-endemic areas. Mycoses. 2006;49:274–82. DOIPubMedGoogle Scholar
- Manfredi R, Mazzoni A, Nanetti A, Chiodo F. Histoplasmosis capsulati and duboisii in Europe: the impact of the HIV pandemic, travel and immigration. Eur J Epidemiol. 1994;10:675–81. DOIPubMedGoogle Scholar
- McKinsey DS, Spiegel RA, Hutwagner L, Stanford J, Driks MR, Brewer J, Prospective study of histoplasmosis in patients infected with human immunodeficiency virus: incidence, risk factors, and pathophysiology. Clin Infect Dis. 1997;24:1195–203. DOIPubMedGoogle Scholar
- Carme B, Ngolet A, Ebikili B, Ngaporo AI. Is African histoplasmosis an opportunistic fungal infection in AIDS? Trans R Soc Trop Med Hyg. 1990;84:293. DOIPubMedGoogle Scholar
- Breton G, Adle-Biassette H, Therby A, Ramanoelina J, Choudat L, Bissuel F, Immune reconstitution inflammatory syndrome in HIV-infected patients with disseminated histoplasmosis. AIDS. 2006;20:119–21. DOIPubMedGoogle Scholar
- Gugnani HC, Muotoe-Okafor F. African histoplasmosis: a review. Rev Iberoam Micol. 1997;14:155–9.PubMedGoogle Scholar
- Wheat LJ, Connolly-Stringfield PA, Baker RL, Curfman MF, Eads ME, Israel KS, Disseminated histoplasmosis in the acquired immune deficiency syndrome: clinical findings, diagnosis and treatment, and review of the literature. Medicine (Baltimore). 1990;69:361–74.PubMedGoogle Scholar
- Dromer F, Mathoulin-Pelissier S, Launay O, Lortholary O. Determinants of disease presentation and outcome during Cryptococcosis: the CryptoA/D study. PLoS Med. 2007;4:e21. DOIPubMedGoogle Scholar
- Gugnani HC, Egere JU, Larsh H. Skin sensitivity to capsulatum and duboisii histoplasmins in Nigeria. J Trop Med Hyg. 1991;94:24–6.PubMedGoogle Scholar
- Muotoe-Okafor FA, Gugnani HC, Gugnani A. Skin and serum reactivity among humans to histoplasmin in the vicinity of a natural focus of Histoplasma capsulatum var. duboisii. Mycopathologia. 1996;134:71–4. DOIPubMedGoogle Scholar
- Thompson GR III, LaValle CE III, Everett ED. Unusual manifestations of histoplasmosis. Diagn Microbiol Infect Dis. 2004;50:33–41. DOIPubMedGoogle Scholar
- Dupont B, Lortholary O, Datry A, Gentilini M, Vinchon I, Guillevin L. Imported histoplasmosis due to H. duboisii in France (1968–1994). 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; New Orleans, LA; 1996 Sept 15–18. Abstract K58.
- Minta DK, Dembele M, Lorre G, Diallo DA, Traore HA, Chabasse D. African histoplasmosis (Histoplasma capsulatum var. duboisii): a case report from Mali [in French]. Sante. 2005;15:195–9.PubMedGoogle Scholar
- Geffray L, Veyssier P, Cevallos R, Beaud B, Mayolle J, Nogier C, African histoplasmosis: clinical and therapeutic aspects, relation to AIDS. Apropos of 4 cases, including a case with HIV-1-HTLV-1 co-infection [in French]. Ann Med Interne (Paris). 1994;145:424–8.PubMedGoogle Scholar
- Murata M, Furusyo N, Otaguro S, Nabeshima S, Ariyama I, Hayashi J. HIV infection with concomitant cerebral toxoplasmosis and disseminated histoplasmosis in a 45-year-old man. J Infect Chemother. 2007;13:51–5. DOIPubMedGoogle Scholar
- Carme B, Ngaporo AI, Ngolet A, Ibara JR, Ebikili B. Disseminated African histoplasmosis in a Congolese patient with AIDS. J Med Vet Mycol. 1992;30:245–8. DOIPubMedGoogle Scholar
- Chandenier J, Goma D, Moyen G, Samba-Lefebvre MC, Nzingoula S, Obengui, et al. African histoplasmosis due to Histoplasma capsulatum var. duboisii: relationship with AIDS in recent Congolese cases [in French]. Sante. 1995;5:227–34.PubMedGoogle Scholar
- Antinori S, Magni C, Nebuloni M, Parravicini C, Corbellino M, Sollima S, Histoplasmosis among human immunodeficiency virus-infected people in Europe: report of 4 cases and review of the literature. Medicine (Baltimore). 2006;85:22–36. DOIPubMedGoogle Scholar
- Sharmin S, Ohori A, Sano A, Kamei K, Yamaguchi M, Takeo K, Histoplasma capsulatum variety duboisii isolated in Japan from an HIV-infected Ugandan patient. Nippon Ishinkin Gakkai Zasshi. 2003;44:299–306.PubMedGoogle Scholar
- Tobon AM, Agudelo CA, Rosero DS, Ochoa JE, De Bedout C, Zuluaga A, Disseminated histoplasmosis: a comparative study between patients with acquired immunodeficiency syndrome and non-human immunodeficiency virus-infected individuals. Am J Trop Med Hyg. 2005;73:576–82.PubMedGoogle Scholar
- Klassen-Fischer M, McEvoy P, Neafie RC, Nelson AM. Accurate diagnosis of infection with Histoplasma capsulatum var. duboisii. Clin Infect Dis. 2004;38:595, author reply 596. DOIPubMedGoogle Scholar
- Wheat LJ, Connolly-Stringfield P, Blair R, Connolly K, Garringer T, Katz BP, Effect of successful treatment with amphotericin B on Histoplasma capsulatum variety capsulatum polysaccharide antigen levels in patients with AIDS and histoplasmosis. Am J Med. 1992;92:153–60. DOIPubMedGoogle Scholar
- Wheat LJ, Connolly-Stringfield P, Blair R, Connolly K, Garringer T, Katz BP. Histoplasmosis relapse in patients with AIDS: detection using Histoplasma capsulatum variety capsulatum antigen levels. Ann Intern Med. 1991;115:936–41.PubMedGoogle Scholar
- Kauffman CA. Histoplasmosis: a clinical and laboratory update. Clin Microbiol Rev. 2007;20:115–32. DOIPubMedGoogle Scholar
- Maubon D, Simon S, Aznar C. Histoplasmosis diagnosis using a polymerase chain reaction method. Application on human samples in French Guiana, South America. Diagn Microbiol Infect Dis. 2007;58:441–4. Epub 2007 May 16. DOIPubMedGoogle Scholar
- Rickerts V, Bialek R, Tintelnot K, Jacobi V, Just-Nubling G. Rapid PCR-based diagnosis of disseminated histoplasmosis in an AIDS patient. Eur J Clin Microbiol Infect Dis. 2002;21:821–3. DOIPubMedGoogle Scholar
- Bracca A, Tosello ME, Girardini JE, Amigot SL, Gomez C, Serra E. Molecular detection of Histoplasma capsulatum var. capsulatum in human clinical samples. J Clin Microbiol. 2003;41:1753–5. DOIPubMedGoogle Scholar
- Wheat J, Sarosi G, McKinsey D, Hamill R, Bradsher R, Johnson P, Practice guidelines for the management of patients with histoplasmosis. Infectious Diseases Society of America. Clin Infect Dis. 2000;30:688–95. DOIPubMedGoogle Scholar
- Wheat LJ, Connolly P, Smedema M, Brizendine E, Hafner R. Emergence of resistance to fluconazole as a cause of failure during treatment of histoplasmosis in patients with acquired immunodeficiency disease syndrome. Clin Infect Dis. 2001;33:1910–3. DOIPubMedGoogle Scholar
- Wheat LJ, Connolly P, Smedema M, Durkin M, Brizendine E, Mann P, Activity of newer triazoles against Histoplasma capsulatum from patients with AIDS who failed fluconazole. J Antimicrob Chemother. 2006;57:1235–9. DOIPubMedGoogle Scholar
- Lortholary O, Denning DW, Dupont B. Endemic mycoses: a treatment update. J Antimicrob Chemother. 1999;43:321–31. DOIPubMedGoogle Scholar
- Goldman M, Zackin R, Fichtenbaum CJ, Skiest DJ, Koletar SL, Hafner R, Safety of discontinuation of maintenance therapy for disseminated histoplasmosis after immunologic response to antiretroviral therapy. Clin Infect Dis. 2004;38:1485–9. DOIPubMedGoogle Scholar
- Shelburne SA III, Hamill RJ. The immune reconstitution inflammatory syndrome. AIDS Rev. 2003;5:67–79.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 13, Number 11—November 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Olivier Lortholary, Centre National de Référence Mycologie et Antifongiques, Centre National de la Recherche Scientifique, Unité de Recherche Associée 3012, Institut Pasteur, 25, rue du Dr Roux, 75015 Paris, France;
Top