Volume 13, Number 3—March 2007
Dispatch
Cryptosporidium hominis Infection of the Human Respiratory Tract
Cite This Article
Citation for Media
Abstract
Cryptosporidium oocysts, observed in a natural sputum sample of a patient with HIV, were further studied by using DNA markers to determine the species of the parasite. C. hominis was identified as the species infecting the patient’s respiratory tract, a finding that strengthens evidence regarding this pathogen’s role in human disease.
Intestinal cryptosporidiosis is a common parasitic disease that causes self-limiting diarrhea in immunocompetent persons (1). Higher frequencies of Cryptosporidium infection are observed in immunocompromised humans, and the main clinical pattern of the infection in these persons is a chronic, life-threatening secretory diarrhea (2).
At least 8 species of Cryptosporidium are described as infecting humans. C. hominis and C. parvum are the most frequently observed in intestinal infections in humans (3). C. meleagridis is also detected both in immunocompetent and immunodeficient patients, although at a lower rate than C. parvum (4).
Respiratory tract infection by Cryptosporidium spp. has been described for immunodeficient persons, most all of whom were coinfected with HIV. However, pulmonary cryptosporidiosis was also described in patients without HIV infection (5,6). In all cases, no systematic identification of the species of Cryptosporidium was pursued except by Meamar et al. (7), in which the parasite was identified as C. parvum.
We describe the detection and identification of C. hominis in the respiratory secretions of a patient with HIV (sample Chile01). We used an oligonucleotide species-specific method and sequencing of parts of the 18S rRNA gene to determine the species of Cryptosporidium. Both analyses showed that the species of Cryptosporidium present in the pulmonary secretion of this patient was C. hominis.
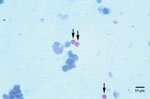
In September 2004, a 58-year-old man, who received an HIV diagnosis in 1996, was hospitalized with respiratory symptoms characterized by persistent cough. Cryptosporidium oocysts were detected in a sputum sample from the patient by using Ziehl-Neelsen stain (Figure 1). An aliquot of ≈10 mL of respiratory secretion was obtained. DNA was extracted as follows: 200 μL fluid was centrifuged and the pelleted material digested overnight at 65ºC with proteinase K in the presence of 10% sodium dodecyl sulfate. We then sequentially extracted the digested material with phenol, phenol-chloroform-isoamyl alcohol, and chloroform-isoamyl alcohol. The DNA were then precipitated with sodium acetate and ethanol, and after centrifugation, the pelleted DNA was dissolved in 50 µL of water.
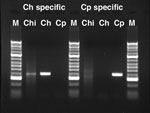
For molecular typing, we first used the species-specific oligonucleotide PCR assay Lib13, as described by Tanriverdi et al. (8), with a new sense oligonucleotide primer based on the genome sequences of C. hominis (9) and C. parvum (10). The new primer, Lib13SF02 (5′-TTTTTTCATTAGCTCGCTTC-3′), a fragment of ≈400 bp, was amplified specifically from C. hominis DNA with the anti-sense primers Lib13SRT-1 (5′-ATTTATTAATTTATCTCTTACTT-3′) and from C. parvum DNA with Lib13SRT-2 (5′-ATTTATTAATTTATCTCTTCG-3′) (Figure 2). Amplifications were carried out in a PCR mixture of 10 μL containing 0.25 mmol/L of each dNTP, 300 pmol/L of each olignucleotide, and 1 unit of Taq DNA polymerase (HotMaster, Eppendorf, New York, NY, USA). Temperature cycling was performed on a GeneAmp PCR System (ABI, Foster City, CA, USA) with initial denaturation performed at 95ºC for 5 min, then 40 cycles at 95ºC for 30 s, 52ºC for 30 s, and 68ºC for 30 s. The mixture was then cooled to 4ºC.
The region from bases 7 to 1036 (numbering according to C. hominis sequence GenBank no. L16996) of the 18S rRNA gene was sequenced from DNA fragments amplified using the primers 18SF (5′-GTTGATCCTGCCAGTAGTC-3′) and 18SR (5′-TAAGGTGCTGAAGGAGTAAGG-3′) and cloned into the TOPO TA vector (Invitrogen, Brandford, CT, USA) by using standard techniques. Automated sequencing was performed directly on the amplified fragments or on cloned fragments at the Nucleic Acid Research Facilities of Virginia Commonwealth University. All DNA sequences were analyzed by using Sequencher (Gene Codes Co., Ann Arbor, MI, USA).
Figure 1 shows fuchsia (Ziehl-Neelsen)–stained sputum from the patient. Three Cryptosporidium oocysts (arrows) with typical sizes ≈5 μm in diameter are visible. Measurements were performed in a calibrated microscope as described by Mercado and Santander (11).
A Lib13 PCR assay (8) was performed on the DNA purified from the respiratory secretion material, and the results are shown in Figure 2. With the C. hominis specific-primer pair (LIBF02/Lib13SRT1), a fragment of the expected size (≈400 bp) was amplified with the Chile01 isolate DNA (Figure 2, lane 2). With this primer pair, we also obtained amplification with the C. hominis isolate TU502 (12) DNA (Figure 2, lane 3) but not with the C. parvum isolate Moredun (13) DNA (Figure 2, lane 4). Conversely, the C. parvum–specific primer pair (LIBF02/Lib13SRT2) amplified a fragment only with the C. parvum DNA (Figure 2, lane 8). No amplification was observed with the Chile01 (Figure 2, lane 6) or the C. hominis DNA (Figure 2, lane 7). DNA sequencing of the amplified fragments confirmed the polymorphism to be that of C. hominis (results not shown).
We also analyzed the 18S small subunit rRNA gene by amplifying and sequencing an 18S rRNA fragment from the sputum DNA. Amplification and sequencing was concomitantly performed with the C. hominis and C. parvum DNA. A polymorphic site exists in C. hominis as a stretch of 10 to 12 thymines (T10–12), while in C. parvum, the sequence is TA:::TATATTTT (146). The 18S rRNA polymorphic sequence found in the sputum sample DNA (GenBank accession no. DQ286403) is that of C. hominis, with a stretch of 11 Ts (Table). Few other nucleotide polymorphisms were found between the sequences (results not shown), which reflect intraisolate variations (14). The results with Lib13 assay and the partial 18S rRNA sequence analysis, therefore, identify the species of Cryptosporidium infecting the respiratory tract of this patient as C. hominis.
Human cryptosporidiosis is better known as an intestinal disease both in immunocompetent and immunocompromised persons. Little information exists, however, on human pulmonary disease caused by Cryptosporidium spp., which reflects either the low prevalence of pulmonary cryptosporidiosis or the lack of testing in immunocompetent hosts. Further, if performed, current diagnostic tests may not be sensitive enough to detect the parasite.
In immunocompetent children with intestinal cryptosporidiosis, respiratory symptoms have been noted more frequently than expected (5). Studies are needed in immunocompetent persons, especially children <2 years of age, who belong to a group at high risk for intestinal, and by extension pulmonary, cryptosporidiosis.
More information about which species of this pathogen infect humans and the pathogenic patterns each species produces is needed. As we determined here, C. hominis have the capacity to adapt to different physiologic environments, such as intestinal and respiratory tract tissues. Our findings provide additional evidence supporting the role of this species of Cryptosporidium as a human pathogen and the need to evaluate the importance of pulmonary cryptosporidiosis as a disease in the immunocompromised host.
Dr Mercado is assistant professor at the Unidad Docente de Parasitologia, Facultad de Medicina, Universidad de Chile, Santiago, Chile. His research interests focus on human parasites and the epidemiology of infectious diseases.
Acknowledgments
We thank Giovanni Widmer and Sultan Tanriverdi for the C. parvum DNA and helpful discussions and Carmen Chambel, Claudio Rojas, and Sergio Guillermo Tula for technical assistance.
This work was supported by the Cryptosporidium Project 2004 for the Unidad Docente de Parasitologia, Faculty of Medicine, Universidad de Chile, and by funds from a National Institutes of Health (grant no. U01 AI46418).
References
- Ramírez NE, Ward LA, Sreevatsan S. A review of the biology and epidemiology of cryptosporidiosis in humans and animals. Microbes Infect. 2004;6:773–85. DOIPubMedGoogle Scholar
- Mercado R, Garcia M. Annual frequency of Cryptosporidium parvum infections in children and adult outpatients, and adults infected by HIV [in Spanish]. Rev Med Chil. 1995;123:479–84.PubMedGoogle Scholar
- Xiao L, Ryan UM. Cryptosporidiosis: an update in molecular epidemiology. Curr Opin Infect Dis. 2004;17:483–90. DOIPubMedGoogle Scholar
- Cama VA, Bern C, Sulaiman IM, Gilman RH, Ticona E, Vivar A, Cryptosporidium species and genotypes in HIV-positive patients in Lima, Perú. J Eukaryot Microbiol. 2003;50(Suppl):531–3. DOIPubMedGoogle Scholar
- Weitz JC, Tassara R, Muñoz P, Mercado R, Atías A. Cryptosporidiosis of the respiratory tract [in Spanish]. Rev Med Chil. 1986;114:691–2.PubMedGoogle Scholar
- Campayo Ibanez A, Lacruz Rodrigo J, Valls Ferrer JM, Bonora Tamarit V. Pulmonary cryptosporidiosis in an immunocompetent female patient [in Spanish]. Med Clin (Barc). 1994;103:237.PubMedGoogle Scholar
- Meamar AR, Rezaian M, Rezaie S, Mohraz M, Kia EB, Houpt ER, Cryptosporidium parvum bovine genotype oocysts in the respiratory samples of an AIDS patient: efficacy of treatment with a combination of azithromycin and paromomycin. Parasitol Res. 2006;98:593–5. DOIPubMedGoogle Scholar
- Tanriverdi S, Ozkan Arslan M, Akiyoshi DE, Tzipori S, Widmer G. Identification of genotypically mixed Cryptosporidium parvum populations in humans and calves. Mol Biochem Parasitol. 2003;130:13–22. DOIPubMedGoogle Scholar
- Xu P, Widmer G, Wang Y, Ozaki LS, Alves JM, Serrano MG, The genome of Cryptosporidium hominis. Nature. 2004;431:1107–12. DOIPubMedGoogle Scholar
- Abrahamsen MS, Templeton TJ, Enomoto S, Abrahante JE, Zhu G, Lancto CA, Complete genome sequence of the apicomplexan, Cryptosporidium parvum. Science. 2004;304:441–5. DOIPubMedGoogle Scholar
- Mercado R, Santander F. Size of Cryptosporidium oocysts excreted by symptomatic children of Santiago, Chile. Rev Inst Med Trop Sao Paulo. 1995;37:473–4. DOIPubMedGoogle Scholar
- Widmer G, Akiyoshi D, Buckholt MA, Fenga X, Richa SM, Deary KM, Animal propagation and genomic survey of a genotype 1 isolate of Cryptosporidium parvum. Mol Biochem Parasitol. 2000;108:187–97. DOIPubMedGoogle Scholar
- Okhuysen PC, Rich SM, Chappell CL, Grimes KA, Widmer G, Feng X, Infectivity of a Cryptosporidium parvum isolate of cervine origin for healthy adults and interferon-γ knockout mice. J Infect Dis. 2002;185:1320–5. DOIPubMedGoogle Scholar
- Gibbons-Matthews C, Prescott AM. Intra-isolate variation of Cryptosporidium parvum small subunit ribosomal RNA genes from human hosts in England. Parasitol Res. 2003;90:439–44. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 13, Number 3—March 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Rubén Mercado, Unidad Docente de Parasitologia, Facultad de Medicina, Universidad de Chile, PO Box 33052, Correo 33, Santiago, Chile
Top