Volume 13, Number 3—March 2007
Letter
Fulminant Supraglottitis from Neisseria meningitidis
Cite This Article
Citation for Media
To the Editor: A 68-year-old Caucasian woman with non–insulin-dependent diabetes mellitus, hypertension, and peripheral vascular disease sought treatment at an emergency department after experiencing 2 days of pharyngitis and 1 day of fatigue and dysphagia for solid food. The morning of admission she noted dysphagia for solid food and liquids, dysphonia, severe anterior neck pain, swelling, and erythema, dyspnea, and a temperature of 102.3°F (39°C). A computed tomographic (CT) scan demonstrated substantial neck soft tissue edema and narrowing of the oropharynx and hypopharynx. She received single doses of intravenous ampicillin/sulbactam, clindamycin, dexamethasone (10 mg), and methylprednisolone (125 mg) before being evacuated by air to our intensive care unit (ICU) at Walter Reed Army Medical Center. Intravenous ampicillin/sulbactam, 3 g every 6 hours, and clindamycin, 900 mg every 8 hours, were continued after the transfer. Two doses of intravenous vancomycin, 1 g every 12 hours, were given before vancomycin was discontinued. Results of laboratory studies were the following: leukocyte count 13.3/mm3 (71% polymorphonuclear leukocytes, 18% bands) and normal hematocrit, platelet count, blood urea nitrogen and creatinine concentrations, and liver-associated enzymes.
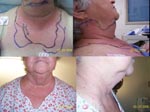
A marker pen was used to track the rapid advance of erythema overnight from her anterior, inferior chin to the top of her breasts (Figure). The infectious disease service was consulted the next morning. When she was examined, her condition had improved; she had normal vital signs, a slightly hoarse voice, and the ability to swallow some saliva. She had no headache or meningismus. The chest erythema was receding. Oral examination demonstrated erythema and an abrasion in the posterior pharynx. Her tongue was not elevated and her uvula was midline. Anterior firm edema without crepitus extended from her chin to the mid-neck. Results of her examination were otherwise unremarkable. The infectious disease consultant recommended restarting a course of vancomycin and discontinuing clindamycin.
A follow-up CT scan with contrast demonstrated anterior cervical soft tissue edema and patent airway with surrounding abnormal thickness and soft tissue density. No abscess or clot was seen. Endoscopic examination in the ICU showed diffuse erythema and generalized supraglottic edema affecting mostly the epiglottis and arytenoids. Dental examination demonstrated no acute pathologic features. Blood cultures at our hospital yielded no growth, and throat culture was negative for group A streptococci.
The patient recovered without requiring intubation (Figure). On the day of discharge, a blood culture from the referring hospital’s emergency department was reported to be positive for Neisseria meningitidis, serogroup Y. Immediate family members and the otolaryngologists who conducted the endoscopic examination were given postexposure prophylaxis. The patient also received terminal prophylaxis. Results of a screening CH50 for terminal complement deficiency were normal.
This patient’s condition is consistent with fulminant meningococcal supraglottitis. Supraglottitis implies involvement of the epiglottis and surrounding structures and is more commonly used to describe adult infection than is epiglottitis (1). Epiglottitis has become more common in adults than in children since the introduction of the Haemophilus influenzae type b vaccine. Other organisms responsible for epiglottitis in adults include H. influenzae, H. parainfluenzae, pneumococci, Staphylococcus aureus, and group A streptococci (2).
Despite its propensity to colonize the upper respiratory tract, N. meningitidis has rarely been identified as a cause of supraglottitis or other deep neck infections. Only 6 cases have been reported, the first in 1995 (3–8). Previously reported cases were equally apportioned by sex, and patients were 44 to 95 years of age (3–8). Including our patient, 3 of 7 were diabetic (6,7). None showed evidence of meningitis or fulminant meningococcemia, but all had fever, pharyngitis, and airway compromise. Five required airway intervention: 3 intubations and 2 urgent tracheostomies. Two received steroids (3,4), a 54-year-old man required urgent tracheostomy before receiving steroids, and a 60-year-old man’s condition “deteriorated rapidly,” but the report does not indicate the interval between receipt of steroids and intubation. Although steroids have been used, their benefit is unproven, and no controlled clinical trials have been conducted (9).
Blood cultures have been positive from all reported case-patients. Two isolates were typed as serogroup B, 4 as serogroup Y, and the serotype of 1 was unreported. Meningococcal strains causing supraglottitis appear to be more locally aggressive but cause less disseminated disease, possibly due to decreased tropism for endothelial cells (8).
To our knowledge, ours is the second case of meningococcal supraglottitis reported with severe neck edema and cellulitis; a 44-year-old woman in a prior review had features similar to our patient (8). An 81-year-old woman with diabetes was noted to have “reddish swelling” on the right side of the neck (7), but little was described beyond that. All 3 had serogroup Y infection. We wondered whether serogroup Y might have a propensity to cause cellulitis; however, a review of 10 cases of meningococcal cellulitis included patients with multiple serogroups: C (4 cases), B (2 cases), Y (2 cases), and unknown (2 cases) (10).
N. meningitidis may cause supraglottitis more frequently than is recognized (3). Timely drawing of blood cultures in relation to administration of antimicrobial drugs is most likely to identify this pathogen in this setting. Because of its public health implications and potential for rapid progression to airway compromise, N. meningitidis should be considered among the differential diagnoses of supraglottitis/epiglottitis.
Dr Kortepeter is a fellow in infectious diseases at Walter Reed Army Medical Center. He served previously as chief of the Medical Division at the US Army Medical Research Institute of Infectious Diseases, Fort Detrick, Maryland. His research interests focus on medical prevention of biological warfare and terrorism.
References
- Shapiro J, Eavey RD, Baker AS. Adult supraglottitis—a prospective analysis.JAMA. 1988;259:563–7. DOIPubMedGoogle Scholar
- Wong EYH, Berkowitz RG. Acute epiglottitis in adults: the Royal Melbourne Hospital experience.ANZ J Surg. 2001;71:740–3. DOIPubMedGoogle Scholar
- Crausman RS, Pluss WT, Jennings CA. Acute epiglottitis in the adult caused by Neisseria meningitidis.Scand J Infect Dis. 1995;27:77–8. DOIPubMedGoogle Scholar
- Nelson K, Watanakunakorn C, Watkins DA. Acute epiglottitis due to serogroup Y Neisseria meningitidis in an adult.Clin Infect Dis. 1996;23:1192–3.PubMedGoogle Scholar
- Donnelly TJ, Crausman RS. Acute supraglottitis: when a sore throat becomes severe.Geriatrics. 1997;52:65–7.PubMedGoogle Scholar
- Sivalingam P, Tully AM. Acute meningococcal epiglottitis and septicaemia in a 65-year-old man.Scand J Infect Dis. 1998;30:196–8. DOIPubMedGoogle Scholar
- Mattila PS, Carlson P. Pharyngolaryngitis caused by Neisseria meningitidis.Scand J Infect Dis. 1998;30:198–200. DOIPubMedGoogle Scholar
- Schwam E, Cox J. Fulminant meningococcal supraglottitis: an emerging infectious syndrome?Emerg Infect Dis. 1999;5:464–7.PubMedGoogle Scholar
- Frantz TD, Rasgon BM, Quesenberry CPJr. Acute epiglottitis in adults: analysis of 129 cases.JAMA. 1994;272:1358–60. DOIPubMedGoogle Scholar
- Porras MC, Martinez VC, Ruiz IM, Encinas PM, Fernandez MT, Garcia J, Acute cellulitis: an unusual manifestation of meningococcal disease.Scand J Infect Dis. 2001;33:56–9. DOIPubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 13, Number 3—March 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Mark G. Kortepeter, Infectious Disease Service, Walter Reed Army Medical Center, 6900 Georgia Ave NW, Washington, DC 20307-5001, USA;
Top