Volume 13, Number 4—April 2007
Letter
Multidrug-resistant Acinetobacter baumannii, Russia
Cite This Article
Citation for Media
To the Editor: During the past decade, nosocomial infections due to multidrug-resistant Acinetobacter baumannii have been described with increasing frequency, mostly in intensive care units (ICUs), resulting in therapeutic difficulties (1). The main mechanism for resistance to extended-spectrum cephalosporins in A. baumannii is attributed to the overexpression of chromosome-encoded cephalosporinases or to plasmid-encoded Ambler class A, B, and D β-lactamases (2). A. baumannii that produce PER-1 extended-spectrum β-lactamase (ESBL) are rarely isolated outside Turkey and remain susceptible to carbapenems (3). Here we describe what we believe is the first ESBL-producing A. baumannii isolate resistant to carbapenems and the first characterization of a PER-1 A. baumannii isolate from Russia, further supporting the emergence and dissemination of PER-1 A. baumannii strains in eastern Europe and outside Turkey (3,4).
On April 17, 2005, a 79-year-old-man was hospitalized in the cardiology ward of a private hospital in Moscow, Russia, with cardiac arrhythmia and a pulmonary infarction subsequent to a pulmonary embolism. After 1 week, he was transferred to the ICU for multiple organ failure related to a nosocomial infection caused by an A. baumannii strain susceptible to several antimicrobial drugs, including imipenem (with positive lung aspiration and blood cultures). He received imipenem and amikacin at high doses. On May 5, 2005, he was transferred to the internal medicine ward of the American Hospital of Paris, Neuilly-sur-Seine, France. On the day of admission, bacterial cultures taken from sputum showed a multidrug-resistant A. baumannii MOS-1 strain, susceptible only to colistin and rifampin. The patient received intravenous and aerosolized colistin 3 times a day plus rifampin at 1,200 mg/d so that he could return to Russia. Rapid identification of A. baumannii MOS-1, increased awareness as a result of a French national alert signaling the emergence of ESBL VEB-1–producing A. baumannii (5), and implementation of strict barrier precautions prevented dissemination of this strain. No other multidrug-resistant A. baumannii isolate with a similar resistance profile has been isolated in the hospital before, during, or after this period.
A. baumannii MOS-1 was susceptible to colistin and rifampin only, and no synergy image could be observed between clavulanic acid and cefepime or ceftazidime discs on a routine antibiogram performed by the disc diffusion method. Only the use of cloxacillin-containing Mueller-Hinton agar plates (200 µg/mL) to inhibit the activity of the naturally occurring cephalosporinase (AmpC) allowed detection of a synergy image, signature of the presence of an ESBL (5). MICs for imipenem, determined by the agar dilution method (6), were >64 µg/mL. Clavulanic acid addition (2 µg/mL) decreased ticarcillin MIC from >512 to 256 µg/mL and ceftazidime MIC from 512 to 128 µg/mL but did not affect MIC of imipenem.
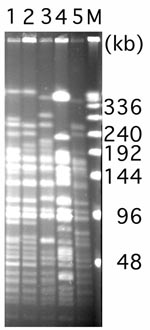
Genes coding for ESBLs and for class B and D carbapenemases were sought by PCR as previously described (4,5). Primers used for detection of TEM and PER β-lactamases gave 894-bp and 825-bp PCR products, respectively (4). Sequence analysis showed identity with blaTEM-1 and blaPER-1 genes (4). Results of isoelectric focusing showed 3 isoelectric point values (5.3 for PER-1, 5.4 for TEM-1, and >8.5 for AmpC) in A. baumannii MOS-1 (4). A crude β-lactamase extract of that isolate had no significant imipenem hydrolysis activity, which suggests that the carbapenem resistance may have emerged through a nonenzymatic mechanism such as mutations in porins (7). BlaPER-1 gene in A. baumannii MOS-1 was located on a composite transposon, Tn1213, identical to that characterized by Poirel et al. (8). Attempts to demonstrate plasmids or transfer genes encoding TEM-1 or PER-1 failed (data not shown), which suggests that the genes were chromosomally encoded. A. baumannii MOS-1 was not clonally related to well-characterized PER-1 A. baumannii strains from Turkey, France, and Belgium (4,9) (Figure), further supporting genetic heterogeneity of PER-1 A. baumannii isolates, even though the immediate genetic environment of blaPER-1 gene was similar (8).
The emergence and spread of ESBL-producing A. baumannii strains are of concern because they will increase carbapenem use, thus raising the risk for emergence of carbapenem-resistant isolates. A. baumannii MOS-1 is, we believe, the first description of an ESBL-producing A. baumannii isolate also being resistant to carbapenems. This resistance was likely acquired in vivo under imipenem treatment, but the susceptible strain was not available for strain comparison. This is also the first description of a PER-1 A. baumannii isolate from Russia, a country from which little epidemiologic data on antimicrobial drug resistance are available, except for the emergence of ESBLs of CTX-M type in Enterobactericeae (10).
This study highlights the importance of international patient transfer in the spread of antimicrobial drug resistance, thus emphasizing the need for hospitals to isolate and screen for multidrug-resistant pathogens in all patients admitted to hospitals from foreign countries. This is particularly critical when the foreign country is known for a high prevalence of multidrug-resistant bacteria or when no antimicrobial drug resistance data are available.
Dr Naas is associate professor in microbiology at the University of Paris, South Medical School. His primary research interest is the genetic basis of antimicrobial drug resistance gene acquisition.
Acknowledgment
This work was funded by a grant from the Ministère de l’Education Nationale et de la Recherche (UPRES-EA3539), Université Paris XI, and by the European Community (6th PCRD, LSHMCT- 2003-503-335).
References
- Allen DM, Hartman BJ. Acinetobacter species. In: Mandell GL, Bennett JE, Dolin R, editors. Principles and practice of infectious diseases. 5th ed. Philadelphia: Churchill Livingstone; 2000. p. 2339–44.
- Nordmann P, Poirel L. Emerging carbapenemases in gram-negative aerobes. Clin Microbiol Infect. 2002;8:321–31. DOIPubMedGoogle Scholar
- Kolayli F, Gacar G, Karadenizli A, Sanic A, Vahaboglu H; The Study Group. PER-1 is still widespread in Turkish hospitals among Pseudomonas aeruginosa and Acinetobacter spp. FEMS Microbiol Lett. 2005;249:241–5. DOIPubMedGoogle Scholar
- Naas T, Bogaerts P, Bauraing C, Degheldre Y, Glupczynski Y, Nordmann P. Emergence of PER and VEB extended-spectrum beta-lactamases in Acinetobacter baumannii in Belgium. J Antimicrob Chemother. 2006;58:178–82. DOIPubMedGoogle Scholar
- Naas T, Coignard B, Carbonne A, Blanckaert K, Bajolet O, Bernet C, VEB-1 extended-spectrum β-lactamase–producing Acinetobacter baumannii, France. Emerg Infect Dis. 2006;12:1214–22.PubMedGoogle Scholar
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: 15th informational supplement. M100-S15. Wayne (PA): Clinical and Laboratory Standards Institute; 2005.
- Fernández-Cuenca F, Martínez-Martínez L, Conejo MC, Ayala JA, Perea EJ, Pascual A. Relationship between β-lactamase production, outer membrane protein and penicillin-binding protein profiles on the activity of carbapenems against clinical isolates of Acinetobacter baumannii. J Antimicrob Chemother. 2003;51:565–74. DOIPubMedGoogle Scholar
- Poirel L, Cabanne L, Vahaboglu H, Nordmann P. Genetic environment and expression of the extended-spectrum beta-lactamase blaPER-1 gene in gram-negative bacteria. Antimicrob Agents Chemother. 2005;49:1708–13. DOIPubMedGoogle Scholar
- Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol. 1995;33:2233–9.PubMedGoogle Scholar
- Edelstein M, Pimkin M, Palagin I, Edelstein I, Stratchounski L. Prevalence and molecular epidemiology of CTX-M extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae in Russian hospitals. Antimicrob Agents Chemother. 2003;47:3724–32. DOIPubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 13, Number 4—April 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Thierry Naas, Service de Bactériologie-Virologie, Hôpital de Bicêtre, 78 rue du Général Leclerc, 94275 K.-Bicêtre Cedex, France;
Top