Volume 14, Number 4—April 2008
Letter
Rat-to-Elephant-to-Human Transmission of Cowpox Virus
Figure
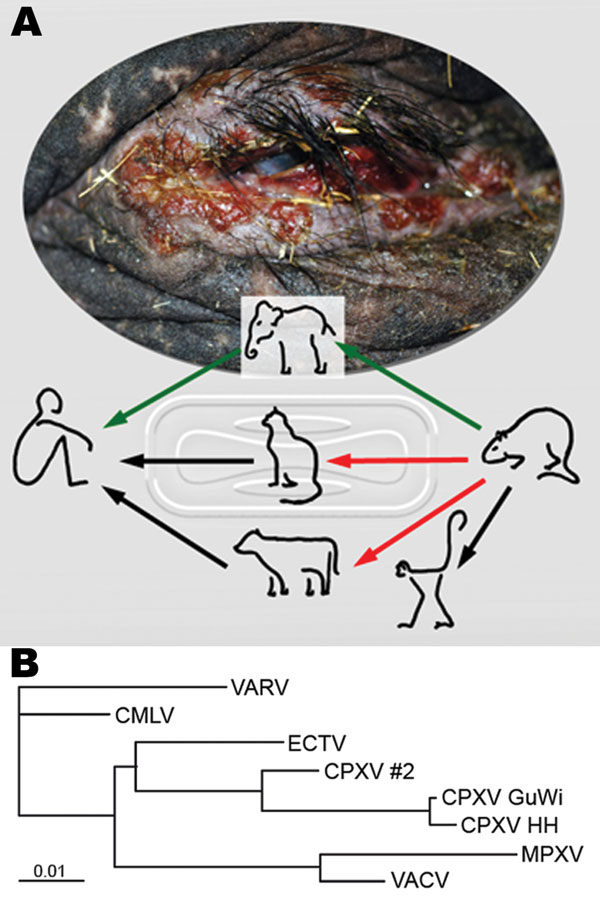
Figure. Route of cowpox virus (CPXV) transmission and phylogenetic analysis of orthopoxviruses. A) Disseminated ulcerative lesions of the skin around the eye of the circus elephant. Although transmission of CPXV has been confirmed from cats and cows to humans (black arrows) (1,2), transmission from rodents, commonly mice, to cats and cows is suspected but still unproven (red arrows) (3). Rats have been confirmed as vectors for CPXV transmission to monkeys and humans (4,7). A complete chain of CPXV infection is verified from rat to elephant and from elephant to human (green arrows). B) Phylogenetic tree of nucleotide sequences of the complete hemagglutinin open reading frame (921 bp) from CPXV isolates from the elephant and rat (CPXV GuWi), and additional poxviruses available in GenBank: VARV (variola major virus, strain Bangladesh-1975; L22579), CMLV (camelpox virus M-96, Kazakhstan; AF438165.1), ECTV (ectromelia virus, strain Moscow; AF012825.2), CPXV HH (cowpox virus cowHA68, Hamburg; AY902298.2), MPXV (monkeypox virus, strain Zaire-96-I-16; AF380138.1), and VACV, (vaccinia virus WR; AY243312). In addition, the complete sequence of the hemagglutinin gene obtained from a different human CPXV case (CPXV #2) found in that area is shown. Nucleotide sequences were aligned and analyzed by using the BioEdit software package (www.mbio.ncsu.edu/BioEdit/bioedit.htm). A multiple alignment was analyzed with the neighbor-joining method. The branch length is proportional to evolutionary distance (scale bar).
References
- Kurth A, Nitsche A. Fast and reliable diagnostic methods for the detection of human poxvirus infections. Future Virology. 2007;2:467–79. DOIGoogle Scholar
- Baxby D, Bennett M, Getty B. Human cowpox 1969–93: a review based on 54 cases. Br J Dermatol. 1994;131:598–607. DOIPubMedGoogle Scholar
- Chantrey J, Meyer H, Baxby D, Begon M, Bown KJ, Hazel SM, Cowpox: reservoir hosts and geographic range. Epidemiol Infect. 1999;122:455–60. DOIPubMedGoogle Scholar
- Wolfs TF, Wagenaar JA, Niesters HG, Osterhaus AD. Rat-to-human transmission of cowpox infection. Emerg Infect Dis. 2002;8:1495–6.PubMedGoogle Scholar
- Eis-Hubinger AM, Gerritzen A, Schneweis KE, Pfeiff B, Pullmann H, Mayr A, Fatal cowpox-like virus infection transmitted by cat. Lancet. 1990;336:880. DOIPubMedGoogle Scholar
- Baxby D, Bennett M. Cowpox: a re-evaluation of the risks of human cowpox based on new epidemiological information. Arch Virol Suppl. 1997;13:1–12.PubMedGoogle Scholar
- Martina BE, van Dornum DG, Dorrstein GM, Niesters HG, Stittelaar KJ, Wolters MA, Cowpox virus transmission from rats to monkeys, the Netherlands. Emerg Infect Dis. 2006;12:1005–7.PubMedGoogle Scholar
- Marennikova SS, Ladnyj ID, Ogorodinikova ZI, Shelukhina EM, Maltseva NN. Identification and study of a poxvirus isolated from wild rodents in Turkmenia. Arch Virol. 1978;56:7–14. DOIPubMedGoogle Scholar
- Essbauer S, Meyer H. Genus orthopoxvirus: cowpox virus. In: Mercer AA, Schmidt A, Weber O, editors. Poxviruses. Basel (Switzerland): Birkhäuser, 2007. p.75–88.
- Begon S, Hazel SM, Baxby D, Bown K, Cavanagh R, Chantrey J, Transmission dynamics of a zoonotic pathogen within and between wildlife host species. Proc Biol Sci. 1999;266:1939–45. DOIPubMedGoogle Scholar