Volume 14, Number 4—April 2008
Peer Reviewed Report Available Online Only
Conference Report on Public Health and Clinical Guidelines for Anthrax
Figure
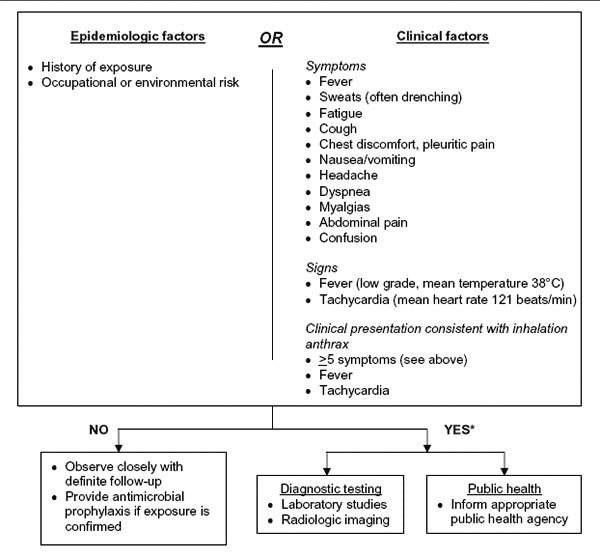
Figure. Revisions to the Centers for Disease Control and Prevention (CDC) interim inhalation anthrax screening guidelines proposed by Mayer et al (29), and reviewed by participants in CDC meeting on public health and clinical guidelines for anthrax. For further detail on subsequent algorithm steps see (29). Adapted from (29).
References
- Bell DM, Kozarsky PE, Stephens DS. Conference Summary: Clinical issues in the prophylaxis, diagnosis, and treatment of anthrax. Emerg Infect Dis. 2002;8:222–5.PubMedGoogle Scholar
- Walsh JJ, Pesik N, Quinn CP, Urdaneta V, Dykewicz CA, Boyer AE, A case of naturally acquired inhalation anthrax: clinical care and analyses of anti-protective antigen immunoglobulin G and lethal factor. Clin Infect Dis. 2007;44:968–71. DOIPubMedGoogle Scholar
- Jernigan DB, Raghunathan PL, Bell BP, Brechner R, Resnitz EA, Butler JC, Investigation of bioterrorism-related anthrax, United States, 2001: epidemiologic findings. Emerg Infect Dis. 2002;8:1019–28.PubMedGoogle Scholar
- Shepard CW, Soriano-Gabarro M, Zell ER, Hayslett J, Lukacs S, Goldstein S, Antimicrobial postexposure prophylaxis for anthrax: adverse events and adherence. Emerg Infect Dis. 2002;8:1124–32.PubMedGoogle Scholar
- CDC. Use of anthrax vaccine in response to terrorism: supplemental recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2002;51:1024–6.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Responding to detection of aerosolized Bacillus anthracis by autonomous detection systems in the workplace. MMWR Morb Mortal Wkly Rep. 2004;53(RR07):1–12.
- Doganay M, Aydin N. Antimicrobial susceptibility of Bacillus anthracis. Scand J Infect Dis. 1991;23:333–5. DOIPubMedGoogle Scholar
- Lightfoot N, Scott R, Turnbull B. Antimicrobial susceptibility of Bacillus anthracis. Salisbury Med Bull. 1990;68(Suppl):95–8.
- Turnbull PC, Sirianni NM, LeBron CI, Samaan MN, Sutton FN, Reyes AE, MICs of selected antibiotics for Bacillus anthracis, Bacillus cereus, Bacillus thuringiensis, and Bacillus mycoides from a range of clinical and environmental sources as determined by the Etest. J Clin Microbiol. 2004;42:3626–34. DOIPubMedGoogle Scholar
- Chen Y, Succi J, Tenover FC, Koehler TM. ß-lactamase genes of the penicillin-susceptible Bacillus anthracis Sterne strain. J Bacteriol. 2003;185:823–30. DOIPubMedGoogle Scholar
- Materon IC, Queenan AM, Koehler TM, Bush K, Palzkill T. Biochemical characterization of beta-lactamases Bla1 and Bla2 from Bacillus anthracis. Antimicrob Agents Chemother. 2003;47:2040–2. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: Investigation of bioterrorism-related anthrax and interim guidelines for exposure management and antimicrobial therapy, October 2001. MMWR Morb Mortal Wkly Rep. 2001;50:909–19.PubMedGoogle Scholar
- Athamna A, Athamna M, Abu-Rashed N, Medlej B, Bast DJ, Rubinstein E. Selection of Bacillus anthracis isolates resistant to antibiotics. J Antimicrob Chemother. 2004;54:424–8. DOIPubMedGoogle Scholar
- Price LB, Vogler A, Pearson T, Busch JD, Schupp JM, Keim P. In vitro selection and characterization of Bacillus anthracis mutants with high-level resistance to ciprofloxacin. Antimicrob Agents Chemother. 2003;47:2362–5. DOIPubMedGoogle Scholar
- Choe CH, Bouhaouala SS, Brook I, Elliot TB, Knudson GB. In vitro development of resistance to ofloxacin and doxycycline in Bacillus anthracis Sterne. Antimicrob Agents Chemother. 2000;44:1766. DOIPubMedGoogle Scholar
- Brook I, Elliott TB, Pryor HI, Sautter TE, Gnade BT, Thaker JH, In vitro resistance of Bacillus anthracis Sterne to doxycycline, macrolides and quinolones. Int J Antimicrob Agents. 2001;18:559–62. DOIPubMedGoogle Scholar
- Friedlander AM, Welkos SL, Pitt ML, Ezzell JW, Worsham PL, Rose KJ, Postexposure prophylaxis against experimental inhalation anthrax. [s]. J Infect Dis. 1993;167:1239–43.PubMedGoogle Scholar
- Department of Health and Human Services. Food and Drug Administration. Prescription Drug Products: Doxycycline and penicillin G procaine administration for inhalational anthrax (post-exposure). Fed Regist. 2001;66:55679–82.
- Davis H, McGoodwin E, Reed TG. Anaphylactoid reactions reported after treatment wth ciprofloxacin. Ann Intern Med. 1989;111:1041–3.PubMedGoogle Scholar
- Shapiro LE, Knowles SR, Shear NH. Comparative safety of tetracycline, minocycline and doxycycline. Arch Dermatol. 1997;133:1224–30. DOIPubMedGoogle Scholar
- Tierney BC, Martin SW, Franzke LH, Marano N, Reissman DB, Louchart RD, Serious adverse events among participants in the Centers for Disease Control and Prevention's anthrax vaccine and antimicrobial availability program for persons at risk for bioterrorism-related inhalational anthrax. Clin Infect Dis. 2003;37:905–11. DOIPubMedGoogle Scholar
- Ortho-McNeil Pharmaceutical. Product insert, Levaquin (Levofloxacin) tablets, 2004.
- Weingartner L, Sitka U, Patsch R, Thiemann HH, Bromme W, Richter I. Amoxicillin in paediatrics, with special reference to its excretion into bronchial secretion. Int J Clin Pharmacol Ther Toxicol. 1980;18:185–9.PubMedGoogle Scholar
- Cook PJ, Andrews JM, Woodcock J, Wise R, Honeybourne D. Concentration of amoxycillin and clavulanate in lung compartments in adults without pulmonary infection. Thorax. 1994;49:1134–8.PubMedGoogle Scholar
- Meselson M, Guillemin J, Hugh-Jones M, Langmuir A, Popova I, Shelokov A, The Sverdlovsk anthrax outbreak of 1979. Science. 1994;266:1202–8. DOIPubMedGoogle Scholar
- Vietri NJ, Purcell BK, Lawler JV, Leffel EK, Rico P, Gamble CS, Short-course postexposure antibiotic prophylaxis combined with vaccination protects against experimental inhalational anthrax. Proc Natl Acad Sci U S A. 2006;103:7813–6. DOIPubMedGoogle Scholar
- Pittman PR, Norris SL, Barrera Oro JG, Bedwell D, Cannon TL, McKee KT Jr. Patterns of antibody response in humans to the anthrax vaccine adsorbed (AVA) primary (six-dose) series. Vaccine. 2006;24:3654–60. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: Investigation of bioterrorism-related anthrax and interim guidelines for clinical evaluation of persons with possible anthrax. MMWR Morb Mortal Wkly Rep. 2001;50:941–8.PubMedGoogle Scholar
- Mayer TA, Morrison A, Bersoff-Matcha S, Druckenbrod G, Murphy C, Howell J, Inhalational anthrax due to bioterrorism: would current Centers for Disease Control and Prevention guidelines have identified the 11 patients with inhalational anthrax from October through November 2001? Clin Infect Dis. 2003;36:1275–83. DOIPubMedGoogle Scholar
- Hupert N, Bearman G, Mushlin A, Callahan M. Accuracy of screening for inhalational anthrax after a bioterrorist attack. Ann Intern Med. 2003;139:337–45.PubMedGoogle Scholar
- Sejvar JJ, Tenover FC, Stephens DS. Management of anthrax meningitis. Lancet Infect Dis. 2005;5:287–95. DOIPubMedGoogle Scholar
- Fritz DL, Jaax NK, Lawrence WB, Davis KJ, Pitt ML, Ezzell JW, Pathology of experimental inhalation anthrax in the rhesus monkey. Lab Invest. 1995;73:691–702.PubMedGoogle Scholar
- Jernigan JA, Stephens DS, Ashford DA, Omenaca C, Topiel MS, Galbraith M, Bioterrorism-related inhalational anthrax: the first 10 cases reported in the United States. Emerg Infect Dis. 2001;7:933–44.PubMedGoogle Scholar
- Holty JE, Bravata DM, Liu HRAO. K.M. M, Owens DK. Systematic review: a century of inhalational anthrax cases from 1900 to 2005. Ann Intern Med. 2006;144:270–80.PubMedGoogle Scholar
- Abramova FA, Grinberg LM, Yampolskaya OV, Walker DH. Pathology of inhalational anthrax in 42 cases from the Sverdlovsk outbreak of 1979. Proc Natl Acad Sci U S A. 1993;90:2291–4. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: Investigation of bioterrorism-related anthrax and interim guidelines for exposure management and antimicrobial therapy, October 2001. MMWR Morb Mortal Wkly Rep. 2001;50:909–19.PubMedGoogle Scholar
- Brachman P, Gold H, Plotkin S, Fekety F, Werrin M, Ingraham N. Field evaluation of a human anthrax vaccine. Am J Public Health. 1962;52:632–45.
- Knudson GB. Treatment of anthrax in man: history and current concepts. Mil Med. 1986;151:71–7.PubMedGoogle Scholar
- Committee on Obstetric Practice. ACOG Committee Opinion number 268, February 2002. Management of asymptomatic pregnant or lactating women exposed to anthrax. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2002;99:366–8. DOIPubMedGoogle Scholar
- Wiesen AR, Littell CT. Relationship between prepregnancy anthrax vaccination and pregnancy and birth outcomes among US Army women. JAMA. 2002;287:1556–60. DOIPubMedGoogle Scholar
Page created: July 16, 2010
Page updated: July 16, 2010
Page reviewed: July 16, 2010
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.