Volume 14, Number 5—May 2008
Dispatch
Morbillivirus and Pilot Whale Deaths, Mediterranean Sea
Cite This Article
Citation for Media
Abstract
An outbreak of a lethal morbillivirus infection of long-finned pilot whales occurred in the Mediterranean Sea from the end of October 2006 through April 2007. Sequence analysis of a 426-bp conserved fragment of the morbillivirus phosphoprotein gene indicates that the virus is more closely related to dolphin morbillivirus than to pilot whale morbillivirus.
Morbilliviruses have emerged as serious pathogens of cetaceans and pinnipeds worldwide (1). The 2 cetacean morbilliviruses that have been identified are porpoise morbillivirus (PMV), isolated from harbor porpoises that died along the coast of Ireland, and dolphin morbillivirus (DMV), first identified in striped dolphins from the Mediterranean Sea (1,2). Although to our knowledge, morbillivirus outbreaks in pilot whales have not been previously reported, antibodies to morbilliviruses have been reported in 86% of 2 species of pilot whales (Globicephala melas and G. macrorrhynchus) in the western Atlantic (3). Barrett et al. found that 93% of stranded long-finned pilot whales (G. melas) were seropositive for morbillivirus, which provides more evidence that cetacean morbilliviruses are widespread (4). Molecular evidence from a pilot whale that was stranded on the coast of New Jersey, USA, and died from encephalitis, suggested that the long-finned pilot whale is host for a different, novel type of cetacean morbillivirus (pilot whale morbillivirus [PWMV]), which is distinct from PMV and DMV (5). We report an epizootic of lethal morbillivirus infection in long-finned pilot whales that occurred in the Mediterranean Sea.
During a 6-month period (end of October 2006 through April 2007), >27 long-finned pilot whales were found stranded, 6 alive and 21 dead, along the southern Spanish Mediterranean coast and Balearic Islands. According to information from the Andalucia regional stranding network, CIRCE, (Conservation, Information, Research, Cetaceans), nongovernment organizations, and scientists working on that coastal area, 10 of these pilot whales were stranded in the Strait of Gibraltar area from the end of October 2006 through early February 2007. From January through April 2007, 7 of these whales were found stranded on the Almería coast, 6 on the Murcia coast, 2 on the Valencia coast; another 2 were found beached on the Baleares Islands. The Table compares the times and locations of these strandings with those of historical strandings.
Of these stranded whales, 18 were found in an advanced autolytic condition, but 9 were fresh or only moderately autolytic, of which complete necropsies were performed on 7, partial necropsies on 2, and samples were collected from all 9. Histologic and immunohistochemical examination of formalin-fixed tissues (mainly lymph node, brain, esophagus, liver, and kidney) was performed for 9 whales, and a virologic examination was performed on frozen tissues (mainly lymph node, lung, and brain) from 6.
According to biological and morphometric parameters, all stranded pilot whales were adults or subadults, except 2 that were juveniles. One female whale stranded off Baleares Islands was 7 months pregnant. For most of the stranded whales, the main macroscopic findings detected during the necrospsy were moderate to severe cachexia, represented by marked loss of volume of epaxial musculature. Stomachs were empty. In 3 whales, subcutaneous tissues were yellowish (icteric) and edematous. All necropsied whales had enlarged edematous lymph nodes, which showed parenchymal multifocal necrosis (especially digestive tract lymph nodes). Erosive stomatitis and erosive-to-ulcerative necrotizing esophagitis was detected in 3 whales. For 2 whales, the urinary bladder was empty and had thickened walls containing yellowish dense mucus in the lumen.
Microscopically, the main lesions were found in lymph nodes, which had a multifocal necrotizing lymphoadenitis and multinuclear syncytial cells. A nonpurulent encephalitis with syncitial cells and intranuclear inclusion bodies, intracytoplasmic inclusion bodies, or both, were detected in 6 whales from which neurologic tissues were analyzed microscopically. Mild interstitial pneumonia was detected in 4 whales, but inflammatory lesions of the lung were absent in the others. One whale, stranded in Murcia, had a focal pyogranulomatous pneumonia caused by Aspergillus sp. Mild to severe, erosive to ulcerative necrotizing esophagitis was detected microscopically in all analyzed whales found to have gross lesions in this organ.
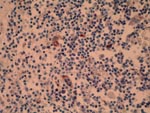
Immunohistochemical staining, with a polyclonal antibody (6), showed morbillivirus antigen in bronchiolar epithelium, syncytial cells, monocyte-like cells, and cell debris of affected lymph nodes and brain; these tissues often showed a positive intracytoplasmic globular or granular immunoreaction. Morbillivirus antigen was detected in all whales for which an immunohistologic study was performed, mainly in the brain (n = 6), lymph nodes (n = 9), and lungs (n = 4) (Figure 1).
Reverse transcription–PCR (RT-PCR) to detect cetacean morbillivirus (CetMV) was performed for available samples of brain, lung, spleen, lymph node, liver, and kidney from 6 of the pilot whales and 1 fetus. Molecular detection of CetMV was performed by a 1-step RT-PCR of a 426-bp conserved region of the phosphoprotein gene, described previously (7). We conducted a BLAST (www.ncbi.nlm.nih.gov/blast/Blast.cgi) search to compare sequenced products with sequences described in the GenBank for morbillivirus. All sequences alignments were obtained, and p-distances were calculated by using MEGA version 3.1 (8).
Of those whales analyzed for virus (6 pilot whales and 1 fetal whale), a morbillivirus was detected by RT-PCR in the brains of 5, lymph nodes of 6, and the lungs of 4. All samples from the fetus (brain, lung, lymph nodes, liver, and kidney) were RT-PCR positive for morbillivirus. Sequencing showed the same sequence in all positive samples from animals stranded in different areas of the southern coast of Spain (Figure 2). The novel sequence obtained was closely related to DMV (p-distance 0.01–0.03) and less closely related (more divergent) to PWMV (p-distance 0.11).
The morbillivirus epizootic reported here induced high mortality rates among long-finned pilot whales in the Mediterranean Sea (Table). The epizootic had a spatiotemporal sequence, involving the long coast from southern Spain, beginning October–November 2006 in the Strait of Gibraltar, spreading eastward to Almería and finally northeast to Murcia; the last cases were detected in Valencia and the Balearic Islands in April 2007. High mortality rates among striped dolphins (Stenella coeruleoalba) have been noted since July 2007 in those coastal areas (currently under investigation along the coasts of Almería, Murcia, Valencia, and Catalunian) (data not shown). In our laboratories, a DMV has been isolated from 3 of those stranded striped dolphins (1 stranded along Murcia and 2 along the Almería coasts). This virus is molecularly almost identical to that reported here as affecting pilot whales (F. Esperón, pers. comm.).
The first morbillivirus epizootic described in cetaceans involved striped dolphins in the Mediterranean Sea in the 1990s when a DMV was described (1,2). Because the viruses isolated from those striped dolphins and these pilot whales are closely related phylogenetically, interspecies transmission should be considered. This epidemiologic point is reinforced by a new die-off event of striped dolphins in Mediterranean waters associated temporally and spatially with the pilot whale deaths caused by a DMV reported here. In the pilot whales the central nervous and lymphatic systems were the most severely affected tissues. Although pilot whales worldwide may be enzootically infected with morbillivirus (9), the virus involved in the present epizootic differs from PWMV (5), which supports previous evidence that different strains of CetMV may be infecting dolphins and whales (10).
Possible explanations for how and why the disease starts are, among others, pollutants (11), the high intensive chronic anthropogenic effects in the Strait of Gibraltar area, a DMV entering a naive pilot whale population, or a progressive decreasing of humoral immunity against the virus in these populations (12). Further research is needed to investigate the role of morbilliviruses on the health and massive deaths of pilot whales and other cetaceans.
Professor Fernández is director of the Research Institute for Animal Health at the University of Las Palmas de Gran Canaria. His areas of expertise include veterinary pathology (including whales and dolphin pathology) and pathogenesis of animal infectious diseases, and his research interests include infectious diseases and anthropogenic effects (e.g., ship collision, sound, and fishing) on whales and dolphins.
Acknowledgments
We thank J.A. Raga for providing samples from a pilot whale stranded in Valencia. We also thank Centro Recuperacion Animales Marinos, CIRCE, PROMAR, and Universidad Murcia for data regarding historical stranding records in southern Spain and Fundación Parques Acuaticos-Nature-Islas Baleares and PROMAR for collaborating in all work done with stranded pilot whales and dolphins in the Baleares Islands and Almería.
This work was partially funded by National Research Project (MEC. AGL2005-07947) and has been done in coordination with the Spanish Ministerio Medio Ambiente and Ministerio Agricultura.
References
- Barrett T, Visser IKG, Mamaev L, Goatley L, van Bressem MF, Osterhaus ADME. Dolphin and porpoise morbilliviruses are genetically distinct from phocine distemper virus. Virology. 1993;193:1010–2. DOIPubMedGoogle Scholar
- Domingo M, Visa J, Pumarola M, Marco A, Ferrer L, Rabanal R, Pathologic and immunocytochemical studies of morbillivirus infection in striped dolphins (Stenella coeruleoalba). Vet Pathol. 1992;29:1–10.PubMedGoogle Scholar
- Duignan PJ, House C, Geraci JR, Duffy N, Rima BK, Walsh MT, Morbillivirus infection in cetaceans of the Western Atlantic. Vet Microbiol. 1995;44:241–9. DOIPubMedGoogle Scholar
- Barrett T, Blixenkrone-Moller M, Di Guardo G, Domingo M, Duignan P, Hall A, Morbilliviruses in aquatic mammals: report on round table discussion. Vet Microbiol. 1995;44:261–5. DOIPubMedGoogle Scholar
- Taubenberger JK, Tsai MM, Atkin TJ, Fanning TG, Krafft AE, Moeller RB, Molecular genetic evidence of a novel morbillivirus in a long-finned pilot whale (Globicephalus melas). Emerg Infect Dis. 2000;6:42–5.PubMedGoogle Scholar
- Yang WC, Pang VF, Jeng CR, Chou LS, Chueh LL. Morbilliviral in a pigmy sperm whale (Kogia breviceps) from Taiwanese waters. Vet Microbiol. 2006;116:69–76. DOIPubMedGoogle Scholar
- Reidarson TH, McBain J, House C, King DP, Stott JL, Krafft A, Morbillivirus infection in stranded common dolphins from the Pacific Ocean. J Wildl Dis. 1998;34:771–6.PubMedGoogle Scholar
- Kumar S, Tamura K, Nei M. MEGA3. Integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform. 2004;5:150–63. DOIPubMedGoogle Scholar
- Van Bressem M, Van Waerebeek K, Jepson PD, Raga JA, Duignan PJ, Nielsen O, An insight into the epidemiology of dolphin morbillivirus worldwide. Vet Microbiol. 2001;81:287–304. DOIPubMedGoogle Scholar
- Taubenberger JK, Tsai M, Krafft AE, Lichy JH, Reid AH, Schulman FY, Two morbilliviruses implicated in bottlenose dolphin epizootics. Emerg Infect Dis. 1996;2:213–6.PubMedGoogle Scholar
- Aguilar A, Borrell A. Abnormally high polychlorinated biphenyl levels in striped dolphins (Stenella coeruleoalba) affected by the 1990–1992 Mediterranean epizootic. Sci Total Environ. 1994;154:237–47. DOIPubMedGoogle Scholar
- Van Bressem MF, Visser IK, Van de Bildt MW, Teppema JS, Raga JA, Osterhaus AD. Morbillivirus infection in Mediterranean striped dolphins (Stenella coeruleoalba). Vet Rec. 1991;129:471–2.PubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 14, Number 5—May 2008
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Antonio Fernández, Institute for Animal Health, Veterinary School, University of Las Palmas de Gran Canaria, Canary Islands, Spain;
Top