Volume 14, Number 9—September 2008
Dispatch
Neurobrucellosis in Stranded Dolphins, Costa Rica
Cite This Article
Citation for Media
Abstract
Ten striped dolphins, Stenella coeruleoalba, stranded along the Costa Rican Pacific coast, had meningoencephalitis and antibodies against Brucella spp. Brucella ceti was isolated from cerebrospinal fluid of 6 dolphins and 1 fetus. S. coeruleoalba constitutes a highly susceptible host and a potential reservoir for B. ceti transmission.
Brucellosis is a zoonotic disease of terrestrial and marine mammals. During the past 3 decades, contacts between cetaceans and humans have increased worldwide (1), augmenting the risk for transmission of pathogenic Brucella spp. from these animals to people (2). Indeed, Brucella marine strains are capable of infecting humans and livestock (3,4).
From August 2004 through April 2007, 10 live striped dolphins, Stenella coeruleoalba (3 female adults, 2 female juveniles, 1 female calf, 4 juvenile males), were found stranded in populated areas at the Pacific shoreline of the Puntarenas Province of Costa Rica. All animals had swimming problems compatible with neurologic disorders and died within 48 hours of being found. Corpses were kept on ice and transported to the Pathology Unit, Veterinary School, National University, Costa Rica, for sampling; necropsy; and histopathologic, immunohistochemical, and serologic studies. With exception of 1 dwarf sperm whale, Kogia sima, these 10 dolphins were the only cetaceans we were able to examine during this 32-month period.
Because marine Brucella spp. have been reported to cause intracerebral infections (3), we decided to perform immunohistochemical and serologic tests. For these tests, rabbit immunoglobulin (Ig) G anti–B. abortus lipopolysaccharide (LPS) was produced and isolated as described elsewhere (5). Antibodies against dolphin Steno bredanensis IgG were produced in rabbits, purified according to described protocols (6). Both rabbit antibodies were linked to fluorescein isothiocyanate and peroxidase and were assayed by using immunofluorescent and Western blot techniques, respectively (5,7). Rose Bengal agglutination test, immunofluorescent assays, and competitive ELISA were designed and used as described (8,9).
Blood was collected from the live dolphins in situ, serum was obtained, and physical and chemical examinations were performed, followed after death by necropsies and gross pathologic and histopathologic studies. Tissues were fixed in formalin, embedded in paraffin wax, sectioned, and stained with hematoxylin and eosin (10). Organs and tissues of 5 adult females and 1 juvenile male were analyzed for bacteria (11); however, no samples for bacteriologic studies were available from the other dolphins that were stranded before July 2006. Identification of the bacterial isolates was performed according to standard protocols (11,12). Fresh tissue impressions or pellets from supernatants of macerated tissues were fixed with cold 3% paraformaldehyde for 15 min on ice and subjected to immunofluorescence for detection of Brucella spp. (9). Genotyping of Brucella isolates was performed by PCR, using 5′-GGCTGATCTCGCAAAGAT-3′ and 5′-CCAGGTCCTTGGCTTCCTTGAG-3′ primers (Invitrogen Corporation, Carlsbad, CA, USA) for the amplification of ribosomal protein L12 and PCR-StyI restriction fragment length polymorphism of the omp2b locus, as described (11,12).
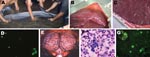
No penetrating wounds or mutilations were found in the dolphins. All animals displayed neurologic disorders characterized by the inability to maintain buoyancy and by opisthotonus, tremors, and seizures (Figure 1, panel A). In all animals, parasitosis was evident and included gastric, intestinal, and pulmonary nematodes as well as subblubber and retroperitoneal cestode larvae. Some dolphins had moderately to severely congested lungs, and 1 dolphin had splenomegaly. The pregnant female had milk in her mammary glands, a 66-cm fetus, and small placental abscesses (Figure 1, panels B, C). Hyperemic meninges, congested brain, and altered cerebrospinal fluid (augmented in volume and cellularity) were evident in all dolphins (Figure 1, panels E, F).
All dolphins had antibodies against Brucella LPS, as determined by immunofluorescence (>1:150 dilution), competitive ELISA (>60% positivity), and Western blot. With the exception of the female calf, all dolphins displayed positive Rose Bengal agglutination. In 6 dolphins, Brucella organisms were demonstrated by immunofluorescence of cerebrospinal fluids and thereafter in tissue impressions of the brain, spinal cord, lymph nodes, spleen, liver, and kidneys. Bacteria were isolated from the same organs and fluids. The pregnant animal had bacteria in the placenta (Figure 1, panel D), umbilical cord, milk, allantoic and amniotic fluids, and fetal tissues. A large number of phagocytes infiltrating the cerebrospinal fluid of these 6 dolphins contained intracellular Brucella organisms (Figure 1, panel G).
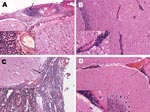
Histopathologic examination of the central nervous system was performed on only the 6 dolphins from which Brucella organisms were isolated; tissues examined were spinal cord, medulla oblongata, cerebellum, pons, thalamic area, and the occipital and frontal cortices of the cerebrum. The most common and relevant histopathologic findings demonstrated meningoencephalitis (Figure 2) and were similar to those previously described (13). The predominant feature was nonsuppurative meningitis, which was more severe in the spinal cord, medulla oblongata, and cerebellum and somewhat more moderate and mild in the cerebral cortices. The meninges were hyperemic and, in most dolphins, edematous. Mild encephalitis was evidenced by a perivascular mononuclear infiltrate in the white and gray matter of the cerebrum, cerebellum, and brainstem and by a periventricular encephalitis that was widespread surrounding the third ventricle with the same cellular infiltrate found in the meninges. A major loss of ependyma was evident; a moderate to severe mononuclear choroiditis was also present. The predominant cellular infiltrate was composed of plasma cells, small lymphocytes, and macrophages. Little or no involvement of the neural tissue was noted in areas other than the periventricular zone. Vasculitis was not found.
Organisms compatible with Brucella (nonmotile; urease-, catalase-, and oxidase positive; gram-negative bacteria not requiring CO2 for growth) were isolated in blood agar or selective media from different organs, brain, and fluids from the 6 dolphins and the fetus subjected to detailed analysis. Standard bacteriologic, immunochemical, biochemical, and molecular typing indicated that the causative agent in all cases was B. ceti type I dolphin strain (Table).
This report documents the presence of marine brucellosis along Latin American shorelines. The dolphins’ neurologic lesions were similar to those described previously in an infected S. coeruleoalba dolphin (13). This finding calls attention to possible increased susceptibility of this species to neurobrucellosis. Indeed, from a total of 46,826 individuals, corresponding to 28 cetacean species counted in the Costa Rican littorals, striped dolphins (inhabiting deep waters) represent only 13% of the animals sighted (14). From January 2004 through December 2006, the number of cetaceans reportedly stranded along the Pacific coast of Costa Rica was 31 (15). Of these, 14 were S. coeruleoalba; the other 16 comprised 11 species of odontocetes. During the 32-month period, we were able to collect samples: 10 (this study) from S. coeruleoalba and 1 from K. sima. This whale was negative for Brucella spp. infections. Endemic and migrating groups of Delphinus delphis and Stenella attenuate are found more frequently along the Costa Rican shorelines; however, few strandings of these species have been reported (14). The relatively high number of stranded S. coeruleoalba dolphins along the Costa Rican shorelines is in agreement with records of strandings along the European littorals (11). These results argue in favor of higher susceptibility of this species to neurobrucellosis.
The isolation of B. ceti from milk, fetal tissues, and secretions of a pregnant dolphin, and a similar discovery in European littorals, suggests that B. ceti is able to display tropism for placental and fetal tissues and that the bacteria may be shed, as it is with Brucella-infected livestock. This finding documents vertical transmission and the possibility of horizontal transmission to newborns. Moreover, the localization of the bacteria in particular organs suggests the possibility of transmission through sexual intercourse and may ensure the prevalence of both clinical and latent infections.
In terms of zoonotic transmission, we noticed that people handled and touched all these infected dolphins, mainly as an attempt to return them to the ocean. Other stranded dolphins have been transferred to privately owned swimming pools or to slaughter for use as a food source for humans and domestic animals. In this regard, susceptibility of S. coeruleoalba as reservoirs of Brucella spp. and modes of transmission must be taken into consideration.
Dr Hernández-Mora is a veterinarian and a researcher at the Tropical Disease Research Program of the National University. Her primary interests are the diagnosis and pathobiology of Brucella infections in cetaceans.
Acknowledgments
We thank José-María Gutiérrez for assistance with preliminary diagnosis of the histologic sections and Charles Manire for advice during the development of this work. We thank Fundación PROMAR and Fundación Keto for logistic support during these 2 years of investigations.
This work was funded by the following grants: Fondo Institucional de Desarrollo Académico-2006-Universidad Nacional, Fondo del Sistema-Consejo Nacional de Rectores, Florida Ice & Farm, Ministerio de Ciencia y Tecnología/Consejo Nacional de Investigaciones Científicas y Tecnológicas-Fondo de Riesgo para la Investigación-FV-001-07, NeTropica 8-N-2005, Costa Rica; and International Foundation for Science B/3456-1, B/3456-2, and B/3456-2F and Comisión Interministerial de Ciencia y Tecnología (grant AGL-2005-0740-CO3), Spain.
References
- Malakoff D. Marine mammals. Scientists use strandings to bring species to life. Science. 2001;293:1754–7. DOIPubMedGoogle Scholar
- Parsons ECM, Rose NA, Bass C, Perry C, Simmonds MP. It’s not just poor science—Japan’s “scientific” whaling may be a human health risk too. Mar Pollut Bull. 2006;52:1118–20. DOIPubMedGoogle Scholar
- Sohn AH, Probert WS, Glaser CA, Gupta N, Bollen AW, Wong JD, Human neurobrucellosis with intracerebral granuloma caused by a marine mammal Brucella spp. Emerg Infect Dis. 2003;9:485–8.PubMedGoogle Scholar
- Perrett LL, Brew SD, Stack JA, MacMillan AP, Bashiruddin JB. Experimental assessment of the pathogenicity of Brucella strains from marine mammals for pregnant sheep. Small Rumin Res. 2004;51:221–8. DOIGoogle Scholar
- Rojas N, Freer E, Weintraub A, Ramírez M, Lind S, Moreno E. Immunochemical identification of Brucella abortus lipopolysaccharide epitopes. Clin Diagn Lab Immunol. 1994;1:206–13.PubMedGoogle Scholar
- Hudson L, Hay FC. Practical immunology. Oxford (UK): Blackwell Scientific; 1976. p. 298.
- Gall D, Colling A, Mariño O, Moreno E, Nielsen K, Pérez B, Enzyme immunoassays for the serological diagnosis of bovine brucellosis: a trial in Latin America. Clin Diagn Lab Immunol. 1998;5:654–61.PubMedGoogle Scholar
- Chaves-Olarte E, Guzmán-Verri C, Méresse S, Desjardins M, Pizarro-Cerdá J, Badilla J, Activation of Rho and Rab GTPases dissociate Brucella abortus internalization from intracellular trafficking. Cell Microbiol. 2002;4:663–76. DOIPubMedGoogle Scholar
- Kiernan JA. Histological and histochemical methods: theory and practice. New York: Oxford University Press; 2003. p. 477.
- Muñoz PM, García-Castrillo G, López-García P, González-Cueli JC, De Miguel MJ, Marín CM, Isolation of Brucella species from a live-stranded striped dolphin (Stenella coeruleoalba) in Spain. Vet Rec. 2006;158:450–1.PubMedGoogle Scholar
- Foster G, Osterman BS, Godfroid J, Jacques I, Cloeckaert A. Brucella ceti sp. nov. and Brucella pinnipedialis sp. nov. for Brucella strains with cetaceans and seals as their preferred hosts. Int J Syst Evol Microbiol. 2007;57:2688–93. DOIPubMedGoogle Scholar
- González L, Patterson IA, Reid RJ, Foster G, Barberán M, Blasco JM, Chronic meningoencephalitis associated with Brucella sp. infection in live-stranded striped dolphins (Stenella coeruleoalba). J Comp Pathol. 2002;126:147–52. DOIPubMedGoogle Scholar
- May-Collado L, Gerrodette T, Calambokidis J, Rasmussen K, Sereg I. Patterns of cetacean sighting distribution in the Pacific exclusive economic zone of Costa Rica based on data collected from 1979–2001. Rev Biol Trop. 2005;53:249–63.PubMedGoogle Scholar
- PROMAR Foundation. Cetacean strandings in Costa Rica. The Costa Rican Network for the Rescue of Marine Mammals 1999–2004 [in Spanish] [cited 2008 Apr 12]. San José (Costa Rica). Available from http://fundacionpromar.org/docs/index.html
Figures
Table
Cite This ArticleTable of Contents – Volume 14, Number 9—September 2008
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Edgardo Moreno, Programa de Investigación en Enfermedades Tropicales, Escuela de Medicina Veterinaria, Universidad Nacional, Heredia, Costa Rica;
Top