Volume 15, Number 10—October 2009
Dispatch
Molecular Epidemiology of Clade 1 Influenza A Viruses (H5N1), Southern Indochina Peninsula, 2004–2007
Cite This Article
Citation for Media
Abstract
To determine the origin of influenza A virus (H5N1) epizootics in Cambodia, we used maximum-likelihood and Bayesian methods to analyze the genetic sequences of subtype H5N1 strains from Cambodia and neighboring areas. Poultry movements, rather than repeated reintroduction of subtype H5N1 viruses by wild birds, appear to explain virus circulation and perpetuation.
From 2004 through 2007, a total of 26 outbreaks of influenza A virus (H5N1) infection have occurred in poultry in Cambodia, and 7 human cases have been reported. Subtype H5N1 infections were observed primarily during the dry season, from January through April. The origin of these epizootics in Cambodia remains unclear. Here we have used maximum likelihood and Bayesian methods to analyze the Cambodian virus genetic sequences, together with those obtained from other H5N1 viruses isolated in neighboring countries since 2004, to understand that patterns of virus transmission.
We analyzed the sequences of all 8 genomic segments of 33 subtype H5N1 viruses sampled in Cambodia from 2004 through 2007, together with publicly available sequences from 116 isolates from Southeast Asia obtained from GenBank. We included all subtype H5N1 viruses sequences from southern Vietnam. In Cambodia, we sequenced systematically at least 2 strains from each infected flock and obtained a total of 137 sequences. When sequences obtained from viruses isolated during the same outbreak site were identical, we used only one for the analyses. After alignment, phylogenetic analyses were conducted by using maximum-likelihood and Bayesian methods (1,2).
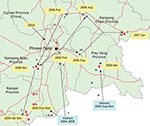
All H5N1 viruses that were detected in Cambodia from 2004 through 2007 belong exclusively to clade 1, genotype Z (Figure 1, panel A) (3,4). The hemagluttinin (HA) phylogenetic tree shows that viruses isolated from Yunnan in 2002 and 2003 are at the origin of all clade 1 viruses from Thailand, Cambodia, and Vietnam (Figure 1, panel A). Sequences obtained from viruses first isolated in Cambodia in January 2004 cluster with a large number of sequences of H5N1 viruses isolated in 2004 and 2005 in Thailand (Figure 1). Subsequent Cambodian subtype H5N1 isolates are grouped into closely related phylogenetic sublineages and are denoted by sublineage number (arbitrarily numbered I–VII for purposes of molecular epidemiology), and these sublineages are also denoted in the map (Figure 2) to show their geographic locations and relationships to likely virus introduction routes. All of these viruses are within the World Health Organization (WHO) clade 1, and these sublineage numbers are not to be confused with the WHO clade/subclade nomenclature.
In 2005, all 4 reported human cases were identified in a limited area of Kampot Province, near the Vietnamese border (Figure 2). Sequences of A/Cambodia/JP52a/2005 detected in January 2005 cluster with strains isolated in 2004 in Southeast Vietnam. The HA gene sequences fall within a cluster of viruses, closely related phylogenetically within clade 1, which we arbitrarily named sublineage I (Figure 1, panel A; Figure 2). Similarly, the strains A/chicken/Cambodia/013LC2b/2005, A/Cambodia/P0322095/2005, and A/Cambodia/408008/2005 detected during March–April 2005 in Kampot and Kandal provinces form separates clusters with sequences of South Vietnamese strains isolated in 2004–2005, namedand named sublineages II, III, and IV, respectively (Figures 1 and 2).
When all the major outbreak sites were plotted on a map of Cambodia, especially sites where viruses from sublineage V (Figure 1, panel A; Figure 2) were isolated in February and early and late March 2006, respectively, it appears that subtype H5N1 virus spread north to south following a main road (Figure 2). Virus A/Cambodia/Q0405047/2006 detected in March 2006 (sublineage VI) is phylogenetically closely related to viruses detected in 2 Vietnamese provinces between August and December 2005 (Figure 1, panel A). We observed 2 virus strains, A/duck/Cambodia/D2KC/2006 and A/duck/Cambodia/D1KC/2006, which were isolated in poultry at the same time and location (February 2006 in Kampong Cham Province), but belong to different sublineages, V and VI, respectively. The HA sequences of the 2 viruses differ by 2 nonsynonymous substitutions that lead to amino acid changes at positions 184 and 195, and by 23 synonymous substitutions. The neuraminidase (NA) gene sequences of both viruses are phylogenetically closely related to the same NA sublineage V, which suggests that a reassortment event occurred (Figure 1, panel B).
Sublineage VII contains 5 separate branches (Figure 1, panel A), which correspond viruses isolated from different geographic locations at different times: Kampong Speu in March 2006 (LC/2006), Kampong Cham in August 2006 (e.g., D1KC167/2006), Prey Veng in August 2006 (e.g., D1PV/2006, which also clusters with a 2007 Vietnamese virus), Kampong Cham in April 2007 (e.g., R0405050/2007) and Vietnam 2006/2007 (Figure 1, panel A). A/Chicken/Vietnam/29/2007 is closely related phylogenetically to Cambodian viruses detected in August 2006 and not closely related to the group of Vietnamese strains from 2006/2007. This suggests that the 2007 Vietnamese strains also have various parental origins, probably related to geographically distinct circulation areas.
For all gene segments, the percentage of homology varied from 98% (for the HA segment) to 99.8% (for the M1 coding gene) between sequences of the first and the latest virus for which complete genome sequences were obtained. Mutations S123P and S129A, R to G substitution in the HA cleavage site, and S155N and K189N mutations at antigenic site B (which could explain the antigenic drift measured) are characteristics that seem to have become established in the latest Cambodian isolates (Appendix Table).
Cambodia is essentially a poultry-importing country. The first poultry deaths were observed in semi-industrial chicken farms that imported broiler and layer parental stocks from a sister company in Thailand, where outbreaks occurred contemporaneously. Whether the viruses were introduced from Vietnam into Cambodia or vice versa cannot be ascertained from the phylogenetic evidence. However, given the known direction of poultry imports from Vietnam to Cambodia, multiple introductions of subtype H5N1 viruses most likely occurred through illegal trading in poultry from Vietnam; this hypothesis would explain how several sublineages emerged in Cambodia in 2005 and 2006. The national veterinary surveillance system in Cambodia is probably not sufficiently sensitive to detect outbreaks resulting from a low prevalence of circulating subtype H5N1. The silent circulation of subtype H5N1 virus in vaccinated poultry (e.g., in ducks) in Vietnam in October 2005 could explain why Vietnam appeared free of subtype H5N1 infections from December 2005 through October 2006, while viruses were still able to cross the border and to infect nonimmune (nonvaccinated) Cambodian poultry (5). Similar to the situation in Vietnam (6), Cambodian farmers tend to release ducks in the paddy fields, particularly after harvesting periods in which duck flocks could feed on the harvest’s leftovers. Seasonality could then be explained by higher duck density (7,8). Viruses in Cambodia very likely represent multiple external introductions rather than becoming established within Cambodia on a continuous basis. This may be related to the fact that the poultry density in Cambodia is lower (average 94 heads/km2) (9) compared with that in neighboring countries such as Thailand and Vietnam (average 640 heads/km2) (10), and one may speculate that a minimum density of poultry is required for maintaining an endemic virus.
Lake Tonle Sap (16,000 km2 surface) and other lakes and wetlands host numerous wild birds, mostly resident or regional migratory birds (M. Gilbert, pers.comm.), which could be involved in the spread of subtype H5N1 virus. Should this mode of transmission play a role, it would probably be limited to local or regional transmission, especially since viruses from other clades (i.e., clade 2.2) that are imported to neighboring countries by migratory birds from the People’s Republic of China, have never been detected in Cambodia.
Sublineage VII–specific mutations (i.e., R325G, S123P) in strains from Cambodia and Vietnam suggest that this new sublineage became endemic in the southern Cambodia–Mekong Delta region during 2006. The phenomenon of virus spread through poultry trading, particularly along the roads, accounts for maintenance and extension of virus within a given region and sustains the risks of transmission to humans.
Dr Buchy is head of the virology unit at the Institut Pasteur in Cambodia. His research interests include H5N1 and other emerging viruses, respiratory viruses, and arboviruses.
Acknowledgments
We are grateful to Richard Webby, Ian Barr, and Alexander Klimov for providing the antisera and the reference viruses used for HI tests and to John Wood for providing NIBRG23 recombinant virus. We also thank the technical staff of the NIC Northern-France for performing some of the HI tests as well as the laboratory technologists from the NIC-Virology Unit at the Institut Pasteur in Cambodia for their precious technical support.
This study was supported by the French Ministry of Health, the French Agency for Development and the US Department of Human Health Services.
References
- Guindon S, Gascuel O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol. 2003;52:696–704. DOIPubMedGoogle Scholar
- Huelsenbeck JP, Ronquist FR. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics. 2001;17:754–5. DOIPubMedGoogle Scholar
- World Health Organization/World Organisation for Animal Health/Food and Agriculture Organization H5N1 Evolution Working Group. Toward a unified nomenclature system for highly pathogenic avian influenza virus (H5N1) [conference summary]. Emerg Infect Dis [serial on the Internet]. 2008 Jul [cited 2009 Sep 14]. Available from http://www.cdc.gov/EID/content/14/7/e1.htm
- Buchy P, Mardy S, Vong S, Toyoda T, Aubin JT, Miller M, Influenza A/H5N1 infections in human in Cambodia. J Clin Virol. 2007;39:164–8. DOIPubMedGoogle Scholar
- Savill NJ, St Rose SG, Keeling MJ, Woolhouse ME. Silent spread of H5N1 in vaccinated poultry. Nature. 2006;442:757. DOIPubMedGoogle Scholar
- Gilbert M, Xiao X, Pfeiffer DU, Epprecht M, Boles S, Czarnecki C, Mapping H5N1 highly pathogenic avian influenza risk in Southeast Asia. Proc Natl Acad Sci U S A. 2008;105:4769–74. DOIPubMedGoogle Scholar
- Pfeiffer DU, Minh P, Martin V, Epprecht M, Otte J. An analysis of the spatial and temporal patterns of highly pathogenic avian influenza occurrence in Vietnam using national surveillance data. Vet J. 2007;174:302–9. DOIPubMedGoogle Scholar
- Gilbert M, Chaitaweesub P, Parakamawongsa T, Premashthira S, Tiensin T, Kalpravidh W, Free-grazing ducks and highly pathogenic avian influenza, Thailand. Emerg Infect Dis. 2006;12:227–34.PubMedGoogle Scholar
- Vétérinaires Sans Frontières. Review of the poultry production and assessment of the socio-economic impact of the highly pathogenic avian influenza epidemic in Cambodia. FAO. 2004 [cited 2009 Sep 14]. Available from http://www.livestockworkinggroup.org/Documents/HPAI_Poultry/AVSF-FAO%20AI%20Review%20Cambodia%202004%20(en).pdf
- Department of Agriculture. Forestry, and Fishery Statistics, Agricultural atlas of Viet Nam. A depiction of the 2001 rural agriculture and fisheries census. 2001 [cited 2009 Sep 14]. Available from http://www.fao.org/ag/AGAInfo/programmes/en/pplpi/maps/vnm_435_poultry.pdf
Figures
Cite This ArticleTable of Contents – Volume 15, Number 10—October 2009
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Philippe Buchy, Institut Pasteur Cambodia, Virology Unit, National Influenza Centre (WHO) 5, Monivong Blvd, PO Box 983, Phnom Penh, Cambodia
Top