Volume 17, Number 8—August 2011
Dispatch
Pandemic (H1N1) 2009–associated Deaths Detected by Unexplained Death and Medical Examiner Surveillance
Cite This Article
Citation for Media
Abstract
During the pandemic (H1N1) 2009 outbreak, Minnesota, New Mexico, and Oregon used several surveillance methods to detect associated deaths. Surveillance using unexplained death and medical examiner data allowed for detection of 34 (18%) pandemic (H1N1) 2009–associated deaths that were not detected by hospital-based surveillance.
The emergence of pandemic (H1N1) 2009 influenza illustrated the need for improved surveillance to identify deaths resulting from emerging pathogens. Common methods for identifying infectious cause–related deaths include reports by health care providers and review of death certificates. These methods have limitations for identifying deaths caused by emerging pathogens because the disease may not be fully defined or death certificates may not indicate an infectious cause. During an emerging pathogen epidemic, it is important to investigate deaths occurring outside of traditional settings to determine if sudden deaths occurring in the community are a result of the novel pathogen.
In 1995, the Centers for Disease Control and Prevention (CDC) Emerging Infections Program (EIP) Unexplained Deaths Program (UNEX) began in 4 states (1). Under UNEX, deaths likely resulting from an infection, but for which routine testing did not identify a pathogen, are investigated. State and CDC Infectious Diseases Pathology Branch researchers partner with medical examiners and hospital pathologists to review cases and autopsy reports. Expanded resources for specimen testing are provided, which increases the likelihood of a pathogen-specific diagnosis.
The Medical Examiner Infectious Disease Death Surveillance Program (Med-X) was developed in 1999 by the New Mexico Office of the Medical Investigator, New Mexico Department of Health, and Infectious Diseases Pathology Branch to review deaths for infectious causes on the basis of preestablished sets of symptoms and pathologic syndromes (2,3). If there is evidence of an infectious process, specimens are tested to achieve an organism-specific diagnosis. Both UNEX and Med-X have been shown to be useful for bioterrorism and infectious death surveillance (4–6).
The EIP has also established population-based active surveillance for all laboratory-confirmed influenza-related hospitalizations and deaths. Minnesota, New Mexico, and Oregon participate in the UNEX, Med-X, and EIP Influenza Surveillance programs to identify all potential influenza-associated deaths.
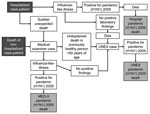
During the spread of pandemic (H1N1) 2009, UNEX cases were reported to the Minnesota and Oregon health departments by physicians, infection preventionists, and hospital pathologists (Figure 1). Both states also conducted statewide surveillance by using Med-X. New Mexico detected cases through the New Mexico Office of the Medical Investigator and its Med-X system. Medical examiners investigated all decedents for influenza-like illness (ILI) based on pre- or postmortem findings as well as sudden deaths in previously healthy persons <50 years of age. Each state expanded its EIP Influenza Surveillance statewide during the pandemic (H1N1) 2009 pandemic. In addition, hospitalized persons with ILI, including decedents, were reported to the state health department by physicians, infection preventionists, and hospital pathologists.
Pre- and/or postmortem specimens, including nasopharyngeal, nasal, or throat swabs; nasal or endotracheal aspirates; bronchial alveolar lavage specimens; sputum; frozen and fixed respiratory tissue; and serum specimens, were tested at state laboratories or at CDC for pandemic (H1N1) 2009 virus. Tests included PCR, virologic culture, immunohistochemistry, serology, and influenza antigen detection. In a few instances, it was not possible to characterize the virus beyond influenza type A because of limited specimen availability; these cases were assumed to be pandemic (H1N1) 2009. Because UNEX and Med-X are not mutually exclusive, all pandemic (H1N1) 2009–associated deaths were determined to be UNEX/Med-X cases if they were captured through either of those programs (Figure 1).
Data were collected on underlying medical conditions, symptoms, and clinical outcomes from medical records, case investigations, and autopsy reports. In Minnesota and New Mexico, all decedents with positive laboratory findings were reviewed to determine if their deaths were due entirely or in part to pandemic (H1N1) 2009. If influenza was determined not to be related to the death, it was not included as a pandemic (H1N1) 2009–associated death; 7 decedents in Minnesota and 2 in New Mexico were thus excluded. Oregon included all hospital surveillance deaths with positive influenza (H1N1) test results as subtype H1N1 associated without further review, but UNEX/Med-X cases were reviewed for a causal relationship to pandemic (H1N1) 2009. Deaths occurring during April–December 2009 were included in this analysis.
Characteristics of UNEX/Med-X cases versus hospital surveillance cases were compared by using the χ2 or Fisher exact test. The Wilcoxon Mann-Whitney test was used to compare medians. SAS version 9.1 software (SAS Institute Inc., Cary, NC, USA) was used for all analyses.
A total of 194 pandemic (H1N1) 2009–associated deaths were detected in this analysis, 160 (82%) through hospital surveillance and 34 (18%) through UNEX/Med-X. The additional surveillance resulted in the detection of 21% more total cases than hospital surveillance alone. Minnesota had the highest proportion of UNEX/Med-X–detected cases with 24% (16/66); Oregon had the lowest with 11% (8/76) (Table 1). Decedents detected by using UNEX/Med-X were more frequently of a nonwhite race (47% vs. 23%); an increased percentage of deaths of American Indians/Alaska Natives was detected through UNEX/Med-X versus hospital surveillance (21% vs. 4%).
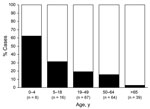
UNEX/Med-X decedents were more likely to have had an autopsy performed (85% vs. 17%) and were more likely to have died in their residences (53% vs. 8%) than decedents detected by hospital surveillance. The median age of UNEX/Med-X decedents was 37.5 years, compared with 51.0 years for hospital surveillance decedents (p<0.001) (Table 1). The percentage of UNEX/Med-X decedents among age groups decreased with increasing age (62.5% among those 0–4 years of age compared with 2.6% among those >65 years of age; Figure 2).
More hospital surveillance than UNEX/Med-X decedents (89% vs. 68%) were determined to have >1 underlying condition. Specific underlying conditions were more frequently identified among hospital surveillance than UNEX/Med-X decedents, except for obesity (Table 2). Pneumonia, including viral pneumonia, was frequently reported among decedents. Acute respiratory distress syndrome was documented for 37% of hospital and 15% of UNEX/Med-X decedents. Two previously healthy children with nasopharyngeal swabs positive for influenza had evidence at autopsy of viral myocarditis.
UNEX/Med-X surveillance captured 11%–24% of pandemic (H1N1) 2009–associated deaths in the 3 states. Other estimates of deaths resulting from pandemic (H1N1) 2009 may be increased with better data on nonhospitalized and sudden unexplained deaths (7,8). Estimates from surveillance in New York, New York, which included medical examiner and unexplained respiratory cause–related death surveillance, indicate 17% of decedents died at home and 6% had not sought any prior medical care (9–11).
UNEX/Med-X decedents were younger and more often previously healthy than hospital surveillance decedents, a finding that would change the estimated impact of pandemic (H1N1) 2009 among those populations in particular. Consistent with other studies (12), larger racial/ethnic disparities, particularly among Native American/Alaska Native populations, may be detected by UNEX/Med-X than have been detected through other surveillance methods. Although we were unable to determine the cause of these disparities, the findings warrant further study and attention to these populations regarding public health resources.
Even with an emphasis on deaths among those <50 years of age, UNEX and Med-X programs are critical for detecting severe illnesses that rapidly progress to death and could otherwise go undetected. Partnering with medical examiners and pathologists to identify infectious cause–related deaths among persons who were previously healthy is important to give a clear picture of the entire mortality spectrum.
Although it is important to accurately measure the impact of a disease, it is perhaps more important to quickly identify new serious disease threats. Approximately one tenth to one quarter of the influenza deaths detected in this study, and particularly those in younger, healthier persons, were not detected by hospital surveillance when influenza awareness was at its peak. This finding argues for surveillance systems like UNEX and Med-X as a means of quickly detecting emerging, severe infectious disease threats. Because pathogens are likely to emerge over broad geographic areas, we recommend a standardized approach to death investigations to fully understand the epidemiologic and clinical features of illness caused by a particular pathogen.
Ms Lees is a nurse epidemiologist with the EIP at the Minnesota Department of Health. Her work includes implementing the Med-X program in Minnesota and currently coordinating the UNEX program. Her research interests include fatal infectious outcomes in the community and how they inform emerging public health threats.
Acknowledgments
We thank Aaron DeVries, Billie Juni, Catherine Lexau, Craig Morin, Dave Boxrud, Joanne Bartkus, Jackie Mahon, Jaime Christensen, Jennifer Palm, Kate Klammer, Larry Carroll, David Determan, Lindsey Thomas, Susan Roe, Emily Duncanson, Andrew Baker, Mitch Morey, Owen Middleton, Lorren Jackson, Michael McGee, Kelly Mills,Victor Froloff, Butch Houston, Anne Bracey, Dennis Uncini, Mark Robia, Walter Connor, Ross Zumwalt, Ian Paul, Michelle Barry Aurelius, R. Ross Reichard, Clarissa Krinsky, Clare Bryce, Meredith Vandermeer, Ann Thomas, Melissa Powell, Madonna Grenz, Christianne Biggs, Karen Gunson, Chris Young, Sherif Zaki, Clifton Drew, Dianna Blau, Lindy Liu, Mitesh Patel, Wun-Ju Shieh, Brigid Batten, Amy Dension, and Marlene DeLeon-Carnes.
Financial support for this study was obtained from the Department of Health and Human Services, CDC EIP and Public Health Emergency Preparedness (PHEP): Minnesota EIP 3U01CI000313, PHEP 5U90TP516981, Oregon EIP 5U01CI000306, PHEP 5U90TP017007-10, and New Mexico PHEP 3U90TP616999.
References
- Hajjeh RA, Relman D, Cieslak PR, Sofair AN, Passaro D, Flood J, Surveillance for unexplained deaths and critical illnesses due to possibly infectious causes, United States, 1995–1998. Emerg Infect Dis. 2002;8:145–53. DOIPubMedGoogle Scholar
- Nolte KB, Lathrop SL, Nashelsky MB, Nine JS, Gallaher MM, Umland ET, "Med-X": a medical examiner surveillance model for bioterrorism and infectious disease mortality. Hum Pathol. 2007;38:718–25. DOIPubMedGoogle Scholar
- Nolte KB, Fischer M, Reagan S, Lynfield R; National Association of Medical Examiners Ad Hoc Committee for Bioterrorism and Infectious Diseases. Guidelines to implement medical examiner/coroner-based surveillance for fatal infectious diseases and bioterrorism (“Med-X”). Am J Forensic Med Pathol. 2010. Epub 2010 Jul 29. DOIPubMedGoogle Scholar
- Hanzlick R, Combs D. Medical examiner and coroner systems: History and trends. JAMA. 1998;279:870–4. DOIPubMedGoogle Scholar
- Palumbo JP, Meek JI, Fazio DM, Turner SB, Hadler JL, Sofair AN. Unexplained deaths in Connecticut, 2002–2003: failure to consider category A bioterrorism agents in differential diagnoses. Disaster Med Public Health Prep. 2008;2:87–94. DOIPubMedGoogle Scholar
- Wang T-H, Wei K-C, Jiang DD-S, Chiu C-H, Chang S-C, Wang J-D. Unexplained deaths and critical illnesses of suspected infectious cause, Taiwan, 2000–2005. Emerg Infect Dis. 2008;14:1653–5. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Updated CDC estimates of 2009 H1N1 influenza cases, hospitalizations and deaths in the United States, April 2009–April 10, 2010 [cited 2010 May 14]. http://www.cdc.gov/h1n1flu/estimates_2009_h1n1.htm
- Centers for Disease Control and Prevention. Surveillance for pediatric deaths associated with 2009 pandemic influenza A (H1N1) virus infection—United States, April–August 2009. MMWR Morb Mortal Wkly Rep. 2009;58:941–7.PubMedGoogle Scholar
- Hadler JL, Konty K, McVeigh KH, Fine A, Eisenhower D, Kerker B, Case fatality rates based on population estimates of influenza-like illness due to novel H1N1 influenza: New York City, May–June 2009. PLoS ONE. 2010;5:e11677. DOIPubMedGoogle Scholar
- Lee EH, Wu C, Lee Elsie U, Stoute A, Hanson H, Cook Heather A, Emerging infections: fatalities associated with the 2009 H1N1 influenza A virus in New York City. Clin Infect Dis. 2010;50:1498–504. DOIPubMedGoogle Scholar
- Presanis AM, De Angelis D, Hagy A, Reed C, Riley S, Cooper BS, The severity of pandemic H1N1 influenza in the United States, from April to July 2009: a Bayesian analysis. PLoS Med. 2009;6:e1000207. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Deaths related to 2009 pandemic influenza A (H1N1) among American Indian/Alaska Natives—12 states, 2009. MMWR Morb Mortal Wkly Rep. 2009;58:1341–4.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 17, Number 8—August 2011
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Christine H. Lees, Minnesota Department of Health, 625 Robert St N., St. Paul, MN 55164, USA
Top