Volume 18, Number 1—January 2012
Letter
Crimean-Congo Hemorrhagic Fever Virus in Ticks from Imported Livestock, Egypt
Cite This Article
Citation for Media
To the Editor: Crimean Congo hemorrhagic fever, a tick-borne illness caused by Crimean Congo hemorrhagic fever virus (CCHFV), is endemic to Africa, the Balkans, the Middle East, and parts of Asia. The hard ticks (Ixodidae), especially those of the genus Hyalomma, serve as reservoirs and vectors for CCHFV, and a variety of animals, such as cattle, sheep, and camels, are considered amplifying hosts for the virus. Although CCHFV may cause little or no disease in zoonotic hosts, the virus can cause severe disease in humans who may be exposed by tick bites or by contact with blood or tissues from infected patients or animals (1). Surveillance for CCHFV in animal and vector populations provides an opportunity to monitor a disease of potentially severe impact.
In North Africa and the Middle East, trade in live animals, meat, and meat products poses noticeable risk to human and animal health (2,3) and can serve as a mobile pool of diseases with potentially large economic and health effects. Animals often originate in distant areas of a country or its neighbors, where they may be exposed to zoonotic pathogens not endemic to their final location, and may collect vectors that carry additional pathogens.
As part of a broader study examining occupational risk of exposure to vector-borne and zoonotic pathogens in a high-risk abattoir worker population, we collected ectoparasites from freshly-slaughtered livestock and examined them for CCHFV. This study and was conducted in compliance with the Animal Welfare Act and in accordance with the principles set forth in the Guide for the Care and Use of Laboratory Animals (4).
Sample collection took place over a 2-week period in July 2009 at the Muneeb abattoir in the Giza Governorate of Egypt. Ectoparasites were removed from 43 freshly slaughtered animals by using blunt forceps and were placed in glass vials. In total, 342 ectoparasites were collected: 70 (20.5%) from 14 cattle, 52 (15.2%) from 17 buffalo, 6 (1.8%) from 2 sheep, and 214 (62.6%) from 10 camels. Cattle, buffalo, and sheep originated in Egypt; camels were imported from Sudan and Somalia.
Ectoparasites were transported to US Naval Medical Research Unit No. 3 in Cairo, Egypt, for taxonomic identification and pathogen detection. Ninety-seven percent (334) of the ectoparasites were ticks from the family Ixodidae. The genus Hyalomma accounted for 254 (76.0%) of these ticks, including nearly all of those collected from sheep (100%) and camels (99.5%) but only 60.4% and 9.1% of those collected from buffalo and cattle, respectively. The remaining ticks of the family Ixodidae belonged to genus Boophilus.
After identification, ticks were grouped into pools by species, sex, and animal source. RNA was then extracted by using the QIAamp Viral RNA Kit (QIAGEN, Valencia, CA, USA). CCHFV small fragment RNA was detected with SuperScript III Platinum SYBR Green One-Step qRT-PCR (Invitrogen, Carlsbad, CA, USA) and published primers as described (5).
Of 138 pools tested (258 ticks of the genera Hyalomma and Boophilus), 6 pools were positive for CCHFV. These 6 pools contained ticks collected from 1 camel imported from Somalia and 4 from Sudan. One positive pool comprised female H. excavatum (Koch, 1844), and 5 comprised female H. dromedarii (Koch, 1844), both proven vectors of CCHFV.
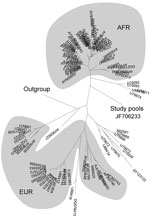
Sequence analysis showed that each CCHFV-positive pool contained an identical yet previously unrecorded 229-bp RNA fragment (GenBank accession no. JF706233; Figure). This fragment had 92% identity with many published CCHFV isolates (BLAST analysis [www.ncbi.nlm.nih.gov/BLAST], GenBank nonredundant database); however, most base changes were synonymous substitutions. Translated protein queries (tblastx) identified an amino acid (serine at codon 26) in this new variant that differed from all but 1 (AJ010648.1) of the 100 most similar published sequences. The functional consequence of a S26N substitution is unknown but may be minimal because of the similar physicochemical properties of asparagine and serine.
Despite the low number of camels sampled in this study, 5 of the 10 camels were found to harbor ticks carrying RNA from an undocumented variant of CCHF. Although none of the domestic animals harbored infected ticks, it is not possible to conclude if these data reflect importation of CCHFV or infection acquired within Egypt because details about conditions under which animals were kept before slaughter are unavailable. Previous serologic studies in Egypt have shown antibodies to CCHFV were prevalent among imported camels at a quarantine station in Aswan governorate (6) and among domestic cattle, buffalo, sheep, and goats in Sharkia Governorate (7). We plan to further investigate the presence of CCHFV within camel and ectoparasite populations in Egypt by expanding protocol activities to a camel market in the same area as the abattoir.
Acknowledgments
We acknowledge management and workers from the Muneeb abattoir for their cooperation and support during specimen collection. We also recognize members of the US Naval Medical Research Unit No. 3 Vector Biology Research Program who assisted with specimen collection: Noha Watany, Reham Mohamed, and Emad El-Din Yehia.
This project was funded by the US Department of Defense Global Emerging Infections System, work unit 847705.82000.25GB.E0018.
References
- Nalca A, Whitehouse CA. Crimean-Congo hemorrhagic fever virus infection among animals. In: Ergonul O, Whitehouse C, editors. Crimean-Congo hemorrhagic fever: a global perspective. Amsterdam; Springer; 2007. p. 155–65.
- Ibrahim B, Salama M, Gawish N, Haridy F. Serological and histopathological studies on Toxoplasma gondii among the workers and the slaughtered animals in Tanta Abattoir, Gharbia Governorate. J Egypt Soc Parasitol. 1997;27:273–8.PubMedGoogle Scholar
- Seimenis A. Zoonoses: A social and economic burden. East Mediterr Health J. 1998;4:220–2.
- Institute of Laboratory Animals Resources. Guide for the care and use of laboratory animals. Washington: The National Academies Press; 1996.
- Drosten C, Göttig S, Schilling S, Asper M, Panning M, Schmitz H, Rapid detection and quantification of RNA of Ebola and Marburg viruses, Lassa virus, Crimean-Congo hemorrhagic fever virus, Rift Valley fever virus, dengue virus, and yellow fever virus by real-time reverse transcription-PCR. J Clin Microbiol. 2002;40:2323–30. DOIPubMedGoogle Scholar
- Morrill JC, Soliman A, Imam I, Botros B, Moussa M, Watts D. Serological evidence of Crimean-Congo haemorrhagic fever viral infection among camels imported into Egypt. J Trop Med Hyg. 1990;93:201–4.PubMedGoogle Scholar
- Mohamed M, Said A-R, Murad A, Graham R. A serological study of Crimean-Congo haemorrhagic fever in animals in the Sharkia Governorate of Egypt. Vet Ital. 2008;44:513–7.PubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 18, Number 1—January 2012
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Jeffrey T. Villinski, Vector Biology Research Program, US Naval Medical Research Unit No. 3, PSC 452 Box 5000, FPO, AE 09835, USA
Top