Volume 19, Number 12—December 2013
Letter
Vaccinia Virus in Household Environment during Bovine Vaccinia Outbreak, Brazil
Cite This Article
Citation for Media
To the Editor: Several exanthematic vaccinia virus (VACV) outbreaks have affected dairy cattle and rural workers in Brazil and Asia, and have caused economic losses and affected health services (1–3). VACV, the prototype of the genus Orthopoxvirus (OPV), exhibits serologic cross-reactivity with other OPV species and was used during the smallpox eradication campaign (1). Several VACV strains have been isolated during bovine vaccinia outbreaks in Brazil and have been characterized by molecular and biologic methods (3,4). Bovine vaccinia infections in humans are frequently related to occupational contact with sick animals during milking but have never been shown to be associated with fomites or indoor environments (1,3).
In August 2011, a bovine vaccinia outbreak was reported in Carangola County, Minas Gerais State, Brazil. During this outbreak, several farms were affected, and the outbreak involved humans and dairy cattle. A 41-year-old man (patient 1) who worked on a farm (20°36′30.7′′S, 42°17′53.9′′W) was hospitalized. He had painful lesions on the hands, high fever, lymphadenopathy, malaise, and fainting episodes. This patient reported recent contact with sick animals on the farm during milking.
At the same time, a 57-year-old man (patient 2), the owner of the farm, had a lesion on the right hand. This infection was also related to occupational exposure. Some days after the appearance of the hand lesion, this patient presumably inoculated himself at the site of an abrasion he had recently received on his nose. This resulted in development of a large and painful lesion. This patient reported milking cows daily. He had been vaccinated against smallpox before 1977.
A total of 5 humans and 15 cows were involved in this outbreak on 5 farms. Clinical samples were obtained from the 2 patients and from 3 sick cows. Dried swab specimens from lesions were soaked in 200 μL of phosphate-buffered saline containing penicillin (200 U/mL), amphotericin B (4 μg/mL), and gentamicin (100 μg/mL); homogenized, and centrifuged at 2,000 × g for 3 min. The supernatants were used for molecular diagnosis and virus isolation (3,5,6). Supernatants were tested by using OPV-specific PCRs that targeted the C11R gene, which encodes viral growth factor (vgf), and the A26L gene, which encodes A-type inclusion (ATI) protein. Samples from the 2 patients were positive for vgf and ATI (7). At least 1 sample (blood or scabs) from each sick animal was also positive by PCR.
To assess the risk for virus spread in indoor environments, we collected swab specimens from several objects, including doorknobs, bathroom surfaces, and the pillow of patient 2. The pillow was positive for vgf and ATI by PCR.
To isolate the virus, we infected monolayers of BSC-40 cells cultured in a 6-well plate with sample supernatants and incubated the cells at 37°C for 72 h or until a cytopathic effect was detected (3,5,6). We isolated virus from a sample from patient 1 and from an environmental sample (the pillow of patient 2), which showed positive results in the molecular diagnostic assays.
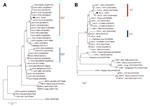
To confirm that the isolated VACV was the OPV involved in this outbreak, we sequenced partial fragments of the A56R and A26L genes from the isolated virus. Fragments obtained were directly sequenced in both orientations in triplicate (MegaBACE 1000 Sequencer; GE Healthcare, Little Chalfont, UK). Sequences were aligned with published OPV sequences in GenBank by using the ClustalW (www.clustal.org/) method and manually aligned by using MEGA version 4.0 (Arizona State University, Phoenix, AZ, USA). VACV molecular signatures of 18-nt and 12-nt deletions were observed in the A56R and A26L genes, respectively. Phylogenetic trees (Figure), which were constructed by using the neighbor-joining method, the Tamura-Nei model of nucleotide substitutions, and 1,000 bootstrap replicates in MEGA 4.0, demonstrated that this isolate clustered with other group 1 VACV isolates from Brazil. We named this isolate Carangola virus.
We isolated VACV from an indoor environment during a bovine vaccinia outbreak. VACV infections have been frequently associated with occupational activities, primarily direct contact with sick animals (1,3). However, in some cases, the source of the infection is unknown, especially in patients who did not participate in milking activities.
Human-to-human transmission has been suggested to have occurred in some bovine vaccinia outbreaks in Brazil, and nosocomial infection has been reported Asia (2,8). Household transmission of VACV has also been described in the United States after contact with lesions of a smallpox vaccinee in the military (9). VACV from Brazil shows long-lasting stability under environmental conditions, especially when associated with organic matter (10). Although the wife of patient 2 did not exhibit any typical clinical symptoms of VAVC infection, we believe that relatives sharing household environments with patients with lesions may be at risk for VACV infection. Isolation of VACV from a household environment raises new questions about nonoccupational risk factors related to bovine vaccinia transmission.
Acknowledgments
We thank our colleagues at the Laboratório de Vírus, Universidade Federal de Minas Gerais, for excellent technical support.
This study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Fundação de Amparo à Pesquisa do Estado de Minas Gerais, Pro-Reitoria de Pesquisa da Universidade Federal de Minas Gerais, and Ministério da Agricultura, Pecuária e Abastecimento. F.L.A., I.A.B., P.C.F., G.S. T., and E.G.K. were supported by fellowships from CNPq.
References
- Damaso CR, Joseph JE, Richard CC, Nissin M. An emergent poxvirus from humans and cattle in Rio de Janeiro State: Cantagalo virus may derive from Brazilian smallpox vaccine. Virology. 2000;277:439–49 . DOIPubMedGoogle Scholar
- Zafar A, Swanepoel R, Hewson R, Nizam M, Ahmed A, Husain A, Nosocomial buffalopoxvirus infection, Karachi, Pakistan. Emerg Infect Dis. 2007;13:902–4 . DOIPubMedGoogle Scholar
- Trindade GS, Lobato ZI, Drumond BP, Leite JA, Trigueiro RC, Guedes MI, Short report: isolation of two vaccinia virus strains from a single bovine vaccinia outbreak in rural área from Brazil: implications on the emergence of zoonotic orthopoxviruses. Am J Trop Med Hyg. 2006;75:486–90 .PubMedGoogle Scholar
- Ferreira JM, Drumond BP, Guedes MI, Xavier MA, Leite CM, Arantes RM, Virulence in murine model shows the existence of two distinct populations of Brazilian vaccinia virus strains. PLoS ONE. 2008;3:e3043. DOIPubMedGoogle Scholar
- Abrahão JS, Guedes MI, Trindade GS, Fonseca FG, Campos RK, Mota BF, One more piece in the VACV ecological puzzle: could peridomestic rodents be the link between wildlife and bovine vaccinia outbreaks in Brazil? PLoS ONE. 2009;4:e7428 . DOIPubMedGoogle Scholar
- Campos RK, Brum MC, Nogueira CE, Drumond BP, Alves PA, Siqueira-Lima L, Assessing the variability of Brazilian vaccinia virus isolates from a horse exanthematic lesion: coinfection with distinct viruses. Arch Virol. 2011;156:275–83. DOIPubMedGoogle Scholar
- Abrahão JS, Larissa SL, Assis FL, Alves PA, Andre TS, Cota MM, Nested-multiplex PCR detection of orthopoxvirus and parapoxvirus directly from exanthematic clinical samples. Virol J. 2009;6:140. DOIPubMedGoogle Scholar
- Batista VH, Scremin J, Aguiar LM, Schatzmayr HG. Vulvar infection and possible human-to-human transmission of bovine poxvirus disease. Virus Rev Res. 2009;14:1–10.
- Centers for Disease Control and Prevention (CDC). Household transmission of vaccinia virus from contact with a military smallpox vaccinee—Illinois and Indiana, 2007. MMWR Morb Mortal Wkly Rep. 2007;56:478–81 .PubMedGoogle Scholar
- Abrahão JS, Trindade Gde S, Ferreira JM, Campos RK, Bonjardim CA, Ferreira PC, Long-lasting stability of vaccinia virus strains in murine feces: implications for virus circulation and environmental maintenance. Arch Virol. 2009;154:1551–3. DOIPubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 19, Number 12—December 2013
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Jonatas S. Abrahão, Laboratório de Vírus, Departamento de Microbiologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais, Belo Horizonte, Minais Gerais 31270-901, BrazilJonatas S. Abrahão, Laboratório de Vírus, Departamento de Microbiologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais, Belo Horizonte, Minais Gerais 31270-901, Brazil
Top