Volume 19, Number 12—December 2013
Dispatch
Powassan Virus in Mammals, Alaska and New Mexico, USA, and Russia, 2004–2007
Cite This Article
Citation for Media
Abstract
Powassan virus is endemic to the United States, Canada, and the Russian Far East. We report serologic evidence of circulation of this virus in Alaska, New Mexico, and Siberia. These data support further studies of viral ecology in rapidly changing Arctic environments.
Powassan virus (POWV) is a tick-borne virus (family Flaviviridae, genus Flavivirus) with recent and increasing prevalence. The only member of the tick-borne encephalitis (TBE) serogroup of flaviviruses endemic to North America, POWV is an emerging cause of human illness and death (1,2). Transmitted primarily by Ixodes spp. ticks and maintained in enzootic cycles involving small- to medium-size mammals, POWV exists as 2 genetically divergent and spatially distinct lineages that are serologically indistinguishable: lineage I, prototype POWV and lineage II, deer tick virus (DTV) (3,4). The 2 lineages are maintained in different vector and host species.
First discovered in eastern Canada, POWV is now known to also circulate in the northeastern United States and the Russian Far East and has been documented in the western United States and Canada in wildlife and human infections (5–9). Clinical signs range from self-limiting febrile illness to severe neurologic disruption and death (2). Both lineages have been isolated from persons with fatal cases, and the incidence of human infection increased from an average of 0.7 cases/year (1958–1998) to 1.9 cases/year (1999–2007) (2,10). This apparent increase, coupled with the relatively recent discovery of lineage II and the well-documented diversity of TBE serogroup flaviviruses in the Old World, highlights the medical role of POWV and related viruses in North America.
Several TBE serogroup viruses, including POWV, also occur in the Russian Far East (6). Two hypotheses have emerged regarding the geographic distribution of POWV. The first hypothesis is that TBE serogroup flaviviruses in the Old and New Worlds persisted during the Pleistocene Epoch in Palearctic and Nearctic refugia (refuge areas), respectively, and then spread across continents (11). The second hypothesis is that POWV was introduced into Russia from North America in the 20th century (6,12). These hypotheses are not mutually exclusive, and POWV or closely related TBE serogroup viruses may be endemic to Beringia, the region surrounding the Bering Strait that connects Asia and North America. Because high latitude environments are experiencing rapid rates of change, and the distribution of POWV in North America is unclear, documenting potential sylvatic hosts of this pathogen is critical to evaluating its capacity to emerge into human populations.
The purpose of this study was to better understand the prevalence, distribution, and host specificity of POWV in western North America and Siberia. We also investigated the history and dynamics of POWV or related TBE serogroup viruses in Beringia.
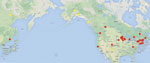
Animals were collected in live traps and snap traps from sites in Siberia (2006), Alaska (2004–2005) and throughout the southwestern United States (2005–2007 (Table 1, Figure 1) under University of New Mexico Institutional Animal Care and Use Committee protocol 12–100764-MCC. Blood was collected on site during specimen processing. We screened blood samples from > 600 wild small-to-medium sized mammals representing 31 host species for POWV-specific antibodies.
Serum samples and supernatants were tested by using a strip immunoblot assay (SIA) with recombinant DTV envelope glycoprotein. Because POWV and DTV are serologically indistinguishable, this antigen binds antibodies specific for DTV, POWV, or other closely related viruses.
In brief, antigen was adhered to a nitrocellulose membrane, and strips were produced with the following antigens and markers: Coomassie blue (orientation control), specific pathogen–free mouse serum (negative control), serum from mice inoculated with DTV envelope glycoprotein (DTV-positive control), mouse IgG (IgG-positive control), and purified DTV E-glycoprotein (test). Approximately 0.25 µg of DTV envelope glycoprotein was used per 2-mm test strip. Samples were tested at a 1:200 dilution, and antibody was detected by using an alkaline phosphatase–conjugated secondary antibody (goat antimouse IgG). Colorimetric intensity was assessed and DTV envelope glycoprotein–positive results were compared with 3+ and 1+ IgG control bands.
In Siberia and central Alaska, antibodies reacting with DTV antigen were detected exclusively in northern red-backed voles (Myodes rutilus) (6.2%) (Table 1). In southern Alaska, DTV-reactive antibodies were detected in the only species tested, the southern red-backed vole (M. gapperi) (6.7%). In the southwestern United States, DTV-reactive antibodies were found in New Mexico in 2 Peromyscus species mice: the piñon mouse (P. truei) and the deer mouse (P. maniculatus) (22.2% and 6.0%, respectively) that were collected sympatrically. The deer mouse is of particular interest because it is the primary host of Sin Nombre virus, the etiologic agent of hantavirus cardiopulmonary syndrome in North America (13).
To identify the virus responsible for serologic reactivity, we collected ticks (Ixodes angustus) from coastal southeastern Alaska (61.3210°N, 145.3030°W; 59.2459°N, 135.1753°W; and 55.8717°N, 132.3481°W) in 2009 from captured mammals (Table 2). Reverse transcription PCR was performed for ticks and tissues from seronegative animals collected proximally to seropositive animals and thus potentially in the acute stage of infection. No viral RNA was detected in ticks or in seronegative rodent tissue.
Although we used a DTV antigen because of its technical convenience, we do not believe that DTV per se is present in these rodent populations. POWV is present throughout western United States and western Canada. However, the virus responsible for the observed seropositivity in Alaska is unknown. The most likely candidate is POWV but without an isolate or sequence data, tick-borne encephalitis virus or other Eurasian flavivirus cannot be ruled out, and we cannot rule out the possibility that the virus is a flavivirus with no known vector. The utility of the SIA is partially based on known cross-reactivity of flaviviruses because it enables detection of divergent lineages. Determination of endpoint antibody titers and confirmation of POWV specificity by plaque-reduction neutralization tests were not possible because of freezer failure.
Because few wild rodent antibodies are commercially available, our methodology used anti-Mus secondary antibody, which may have varying sensitivity against the 31 species tested. Thus, low-level reactivity may have been missed. However, the prevalence of antibodies detected by SIA in our study is consistent with that reported from known POWV transmission foci (14).
These serologic results enable us to conclude that ≥1 flaviviruses antigenically similar to DTV circulate in Siberia, Alaska, and the southwestern United States (Table 1). Transmission appears to involve Myodes spp. voles in northern regions and Peromyscus mice in southern regions. Considerable overlap in the geographic ranges of these species may provide continuous populations of competent amplifying hosts from Mexico (P. maniculatus and P. truei) to Siberia (M. rutilus) (Figure 2). The seropositivity in Siberia may be from introduced POWV, native TBE virus, or other related virus. Viral RNA sequence is necessary to delineate the viral species that are circulating among M. rutilus in Siberia. Additional host species may be involved; considering the small sample for the current study, seropositivity rates and distributions, although consistent with expectations, may be considerably refined with increased sampling (Table 1). The incidence and host association of Ix. angustus ticks were similar to those of a previous report (15), and further vectorial studies are warranted.
Our findings augment knowledge of distribution of TBE serogroup flavivirus in the Nearctic and will guide further studies of New World TBE serogroup flavivirus ecology. Future work will focus on acquisition of viral isolates and nucleic acid sequences from Myodes spp. voles in Alaska and Siberia and from Peromyscus spp. mice in the southwestern United States.
Dr Deardorff is a postdoctoral researcher in the Biology Department of the University of New Mexico, Albuquerque, New Mexico. Her research interests include emerging zoonotic viral disease and host switching in changing environments.
Acknowledgments
We thank Kendra Pesko and Diane Goode-Hanfelt for providing laboratory assistance; the US Department of Agriculture Forest Service (Tongass Forest and Pacific Northwest Laboratory), the Alaska Department of Fish and Game, the US Fish and Wildlife Service, the National Park Service, and the US Geological Survey for providing assistance; and the National Science Foundation for providing logistics for our investigations in western North America and Siberia.
This study was supported by the National Science Foundation (grant 0415668).
References
- Ebel GD. Update on Powassan virus: emergence of a North American tick-borne flavivirus. Annu Rev Entomol. 2010;55:95–110 . DOIPubMedGoogle Scholar
- Hinten SR, Beckett GA, Gensheimer KF, Pritchard E, Courtney TM, Spears SD, Increased recognition of Powassan encephalitis in the United States, 1999–2005. Vector Borne Zoonotic Dis. 2008;8:733–40 . DOIPubMedGoogle Scholar
- Beasley DW, Suderman MT, Holbrook MR, Barrett AD. Nucleotide sequencing and serological evidence that the recently recognized deer tick virus is a genotype of Powassan virus. Virus Res. 2001;79:81–9 . DOIPubMedGoogle Scholar
- Kuno G, Artsob H, Karabatsos N, Tsuchiya KR, Change GJ. Genomic sequencing of deer tick virus and phylogeny of Powassan-related viruses of North America. Am J Trop Med Hyg. 2001;65:671–6 .PubMedGoogle Scholar
- Johnson HN. Isolation of Powassan virus from a spotted skunk in California. J Wildl Dis. 1987;23:152–3 . DOIPubMedGoogle Scholar
- Leonova GN, Kondratov IG, Ternovoi VA, Romanova EV, Protopopova EV, Chausov EV, Characterization of Powassan viruses from Far Eastern Russia. Arch Virol. 2009;154:811–20 . DOIPubMedGoogle Scholar
- Fitch WM, Artsob H. Powassan encephalitis in New Brunswick. Can Fam Physician. 1990;36:1289–90 .PubMedGoogle Scholar
- McLean DM, Crawford MA, Ladyman SR, Peers PR, Purvin-Good KW. California encephalitis and Powassan virus activity in British Columbia, 1969. Am J Epidemiol. 1970;92:266–72 .PubMedGoogle Scholar
- Kettyls GD, Verrall VM, Wilton LD, Clapp JB, Clarke DA, Rublee JD. Arbovirus infections in man in British Columbia. Can Med Assoc J. 1972;106:1175–9 .PubMedGoogle Scholar
- Tavakoli NP, Wang H, Dupuis M, Hull R, Ebel GD, Gilmore EJ, Fatal case of deer tick virus encephalitis. N Engl J Med. 2009;360:2099–107 . DOIPubMedGoogle Scholar
- Telford SR III, Armstrong PM, Katavolos P, Foppa I, Garcia AS, Wilson ML, A new tick-borne encephalitis-like virus infecting New England deer ticks, Ixodes dammini. Emerg Infect Dis. 1997;3:165–70 . DOIPubMedGoogle Scholar
- Pesko KN, Torres-Perez F, Hjelle BL, Ebel GD. Molecular epidemiology of Powassan virus in North America. J Gen Virol. 2010;91:2698–705. DOIPubMedGoogle Scholar
- Nichol ST, Spiropoulou CF, Morzunov S, Rollin PE, Ksiazek TG, Feldmann H, Genetic identification of a hantavirus associated with an outbreak of acute respiratory illness. Science. 1993;262:914–7. DOIPubMedGoogle Scholar
- Ebel GD, Campbell EN, Goethert HK, Spielman A, Teflored SR III. Enzootic transmission of deer tick virus in New England and Wisconsin sites. Am J Trop Med Hyg. 2000;63:36–42 .PubMedGoogle Scholar
- Murrell BP, Durden LA, Cook JA. Host associations of the tick, Ixodes angustus (Acari: Ixodidae), on Alaskan mammals. J Med Entomol. 2003;40:682–5 . DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 19, Number 12—December 2013
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Eleanor R. Deardorff, Biology Department and Museum of Southwestern Biology, University of New Mexico, MS C03-2020 Biology, 1 University of New Mexico, Albuquerque, NM 87131, USAEleanor R. Deardorff, Biology Department and Museum of Southwestern Biology, University of New Mexico, MS C03-2020 Biology, 1 University of New Mexico, Albuquerque, NM 87131, USA
Top