Volume 19, Number 8—August 2013
Dispatch
Norovirus Surveillance among Callers to Foodborne Illness Complaint Hotline, Minnesota, USA, 2011–2013
Cite This Article
Citation for Media
Abstract
Norovirus is the leading cause of foodborne disease in the United States. During October 2011–January 2013, we conducted surveillance for norovirus infection in Minnesota among callers to a complaint-based foodborne illness hotline who reported diarrhea or vomiting. Of 241 complainants tested, 127 (52.7%) were positive for norovirus.
Norovirus is the leading cause of foodborne disease and sporadic and outbreak-associated acute gastroenteritis in the United States (1,2), accounting for 21 million illnesses, 70,000 hospitalizations, and 800 deaths each year (3). Norovirus is not routinely tested for in clinical settings because detection requires molecular methods typically available only in public health and research laboratories. Therefore, characterization of norovirus epidemiology has been primarily through analysis of outbreak data.
Consistent with national trends (4), most foodborne disease outbreaks identified in Minnesota are caused by norovirus. In addition, most foodborne outbreaks in Minnesota, including virtually all norovirus outbreaks, are identified through a centralized foodborne illness complaint hotline system, operated by the Minnesota Department of Health (MDH) (5,6). However, most calls to the hotline represent sporadic (i.e., non–outbreak-associated) illness; only ≈7% of complaints are associated with known outbreaks (5). Systematic testing of hotline callers to determine illness etiology has not previously been conducted.
In this study, we conducted surveillance for norovirus among hotline callers. Our objectives were to characterize the role of norovirus as a cause of gastroenteritis in hotline callers and to describe trends in norovirus infection in this population as an indicator for norovirus activity in Minnesota.
The MDH foodborne illness complaint system has been described in detail (5,6). From October 1, 2011, through January 31, 2013, eligible hotline callers (complainants) were asked to submit a self-collected fecal sample to the MDH Public Health Laboratory (PHL). Complainants were eligible to submit a stool sample on the basis of reported symptoms (>3 loose stools in 24 hours or vomiting [symptom eligibility]) and other criteria, including timeliness of complaint (Technical Appendix). If the original complainant was not eligible for or refused testing, another ill person reported in the complaint (co-complainant) was asked to submit a stool sample, if eligible. Only 1 stool sample per complaint was used in analyses. This surveillance effort was exempted from review by the MDH Institutional Review Board.
Specimen vials were refrigerated on receipt at the MDH PHL and batch tested weekly. Detection and characterization of norovirus strains were performed by using the Centers for Disease Control and Prevention CaliciNet methods (7). Briefly, detection of norovirus genogroups I and II was performed by duplex real-time reverse transcription PCR. Genotypes were determined by sequence analysis of the viral capsid gene and phylogenetic comparison with CaliciNet reference strains.
On the basis of the known winter seasonality of norovirus outbreaks (8), norovirus season was defined as October–March and the off-season as April–September. Data analysis was performed by using SAS version 9.2 software (SAS Institute Inc., Cary, NC, USA).
During October 2011–January 2013, the Minnesota foodborne illness hotline received 1,060 calls (median 60 calls/mo) (Table 1). The mean number of monthly calls to the hotline was greater during the norovirus season than during the off-season (73.6 vs. 54.0; p = 0.025). A total of 633 (59.7%) complainants or co-complainants met the eligibility requirements for stool sample submission; of these, 241 (38.1%) submitted a sample that was included in analyses.
Of the 241 stool samples, 127 (52.7%) were positive for norovirus: 22 (17.3%) for genogroup I, 104 (81.9%) for genogroup II, and 1 for genogroups I and II (Table 1; Figure 1). The monthly percentage of norovirus-positive samples varied from 23.1% in May 2012 to 81.3% in December 2012 (Table 1; Figure 1). Complainants who called during the norovirus season were more likely to test positive for norovirus than were those who called during the off-season (62.8% vs. 27.5%; p<0.001) (Table 2). Norovirus-positive complainants were more likely than norovirus-negative complainants to report vomiting (87.3% vs. 64.9%; p<0.001) and fever (52.9% vs. 36.2%; p = 0.049) and to have longer illness duration (median 36 vs. 18 hours; p<0.001) (Table 2).
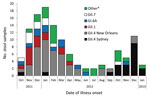
The most common genotypes among the 122 norovirus-positive specimens that could be sequenced were GII.4 New Orleans (44, 36.1%), GII.4 Sydney (20, 16.4%), GII.1 (14, 11.5%), GI.6 (12, 9.8%), and GII.7 (10, 8.2%) (Figure 2). GII.4 New Orleans was predominant during the 2011–2012 norovirus season, and GII.4 Sydney was most common during the first 4 months of the 2012–2013 norovirus season (Figure 2).
This study highlights the predominant role of norovirus infections among callers to a foodborne illness complaint hotline in Minnesota. Call volume may be partially driven by norovirus activity: more calls were taken during the norovirus season, when a higher proportion of callers were norovirus positive. GII.4 norovirus strains were more prominent during peak norovirus season, and GI and less common GII genotypes were more prominent in the off-season. A review of published norovirus outbreaks found that GII outbreaks were significantly associated with winter seasonality compared with GI outbreaks (9). Additionally, GII.4 outbreaks have been associated with severe outcomes, such as hospitalization and death (10), underscoring the importance of monitoring their emergence and effects.
The greater proportion of vomiting and fever and longer illness duration among norovirus-positive complainants suggests that a bacterial intoxication, especially with diarrheal toxin agents such as Clostridium perfringens, may have caused a substantial proportion of illness among norovirus-negative complainants. However, complainant samples were not routinely tested for bacterial intoxication agents in this study because of the lag time from onset to complaint. Differences in rates of fever and health care visits between eligible complainants and those tested (Table 2) limit the accuracy of extrapolated estimates if these variables affect the likelihood that a caller is norovirus positive. However, if all symptom-eligible complainants are assumed to have the same risk for norovirus infection as the subpopulation of those tested, an estimated 1 in 5 callers during the peak off-season and 3 in 4 callers during the peak season would be infected with norovirus.
These results have limited potential for extrapolation to norovirus incidence estimates for Minnesota. The proportion of the population who would call the hotline when ill is unknown; in addition, hotline callers are not necessarily representative of the general population. However, trends observed among hotline callers, including norovirus prevalence, genotype diversity, and call volume, can serve as indicators of general norovirus activity. For example, our study demonstrates the transition in predominant circulating norovirus strain from GII.4 New Orleans to the emergent GII.4 Sydney strain, as has been observed among US outbreaks (11). The emergence of a new GII.4 strain has sometimes been associated with an increase in norovirus outbreak activity (12). However, an increase in proportion of callers positive for norovirus during the beginning of the 2012–2013 season was not observed in our study after the emergence of GII.4 Sydney. During this same period, the number of norovirus outbreaks identified by MDH was likewise not higher than in recent years (12; MDH, unpub. data), suggesting that GII.4 Sydney did not cause increased norovirus activity in Minnesota. Of note, a complainant with a sporadic case from October 2011 tested through this project was initially identified as being infected with GII.4 New Orleans, but GII.4 Sydney infection was retrospectively identified after CaliciNet updated its reference strains in November 2012 to include GII.4 Sydney.
In conclusion, norovirus accounted for most cases of acute gastroenteritis among hotline callers in Minnesota, particularly during the fall and winter norovirus season. Trends in positive specimens, genotype distribution, and symptom histories observed during complaint-based surveillance can be used to better understand the epidemiology of norovirus gastroenteritis.
Ms Saupe is an epidemiologist in the foodborne diseases unit at the Minnesota Department of Health. Her research interests include norovirus genotype differences and viral shedding.
Acknowledgments
We thank Ginette Dobbins, Mary Horn, Victoria Lappi, Dave Boxrud, and other staff at MDH PHL for their vital work on this project. Thanks also to the foodborne diseases staff at MDH and to the public health student workers on Team Diarrhea for their assistance with kit preparation, courier services, and other parts of the study.
This work was supported in part through a cooperative agreement with the Centers for Disease Control and Prevention Emerging Infections Program, Foodborne Diseases Active Surveillance Network (FoodNet) (5U50CK000204), and Agriculture and Food Research Initiative Competitive Grant no. 2011-68003-30395 from the US Department of Agriculture, National Institute of Food and Agriculture.
References
- Hall AJ, Rosenthal M, Gregoricus N, Greene SA, Ferguson J, Henao OL, Incidence of acute gastroenteritis and role of norovirus, Georgia, USA, 2004–2005. Emerg Infect Dis. 2011;17:1381–8 .PubMedGoogle Scholar
- Hall AJ, Wikswo ME, Manikonda KL, Roberts VA, Yoder JS, Gould LH. Comprehensive surveillance for acute gastroenteritis outbreaks, United States, 2009–2010. Emerg Infect Dis. 2013;19:1305–9.
- Hall AJ, Lopman BA, Payne DC, Patel MM, Vinje J, Parashar UD. Norovirus disease, United States. Emerg Infect Dis. 2013;19: 1198–1205.
- Hall AJ, Eisenbart VG, Etingue AL, Gould LH, Lopman BA, Parashar UD. Epidemiology of foodborne norovirus outbreaks, United States, 2001–2008. Emerg Infect Dis. 2012;18:1566–73 and. DOIPubMedGoogle Scholar
- Li J, Smith K, Kaehler D, Everstine K, Rounds J, Hedberg C. Evaluation of a statewide foodborne illness complaint surveillance system in Minnesota, 2000 through 2006. J Food Prot. 2010;73:2059–64 .PubMedGoogle Scholar
- Li J, Maclehose R, Smith K, Kaehler D, Hedberg C. Development of a Salmonella screening tool for consumer complaint–based foodborne illness surveillance systems. J Food Prot. 2011;74:106–10 . DOIPubMedGoogle Scholar
- Vega E, Barclay L, Gregoricus N, Williams K, Lee D, Vinjé J. Novel surveillance network for norovirus gastroenteritis outbreaks, United States. Emerg Infect Dis. 2011;17:1389–95 .PubMedGoogle Scholar
- Yen C, Wikswo ME, Lopman BA, Vinje J, Parashar UD, Hall AJ. Impact of an emergent norovirus variant in 2009 on norovirus activity in the United States. Clin Infect Dis. 2011;53:568–71 . DOIPubMedGoogle Scholar
- Matthews JE, Dickey BW, Miller RD, Felzer JR, Dawson BP, Lee AS, Systematic review: the epidemiology of published norovirus outbreaks: a review of risk factors associated with attack rate and genogroup. Epidemiol Infect. 2012;140:1161–72 . DOIPubMedGoogle Scholar
- Desai R, Hembree CD, Handel A, Matthews JE, Dickey BW, McDonald S, Severe outcomes are associated with genogroup 2 genotype 4 norovirus outbreaks: a systematic literature review. Clin Infect Dis. 2012;55:189–93. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Notes from the field: emergence of new norovirus strain GII.4 Sydney—United States, 2012. MMWR Morb Mortal Wkly Rep. 2013;62:55 .PubMedGoogle Scholar
- Leshem E, Wikswo M, Barclay L, Salehi E, Storm W, Brandt E, Effects and clinical significance of GII.4 Sydney norovirus, United States, 2012–2013. Emerg Infect Dis. 2013;19:1231–8.
Figures
Tables
Cite This ArticleTable of Contents – Volume 19, Number 8—August 2013
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Amy Saupe, Minnesota Department of Health, 625 Robert St N, PO Box 64975, St. Paul, MN 55164, USA
Top