Volume 20, Number 12—December 2014
Dispatch
Equine Influenza A(H3N8) Virus Isolated from Bactrian Camel, Mongolia
Figure 2
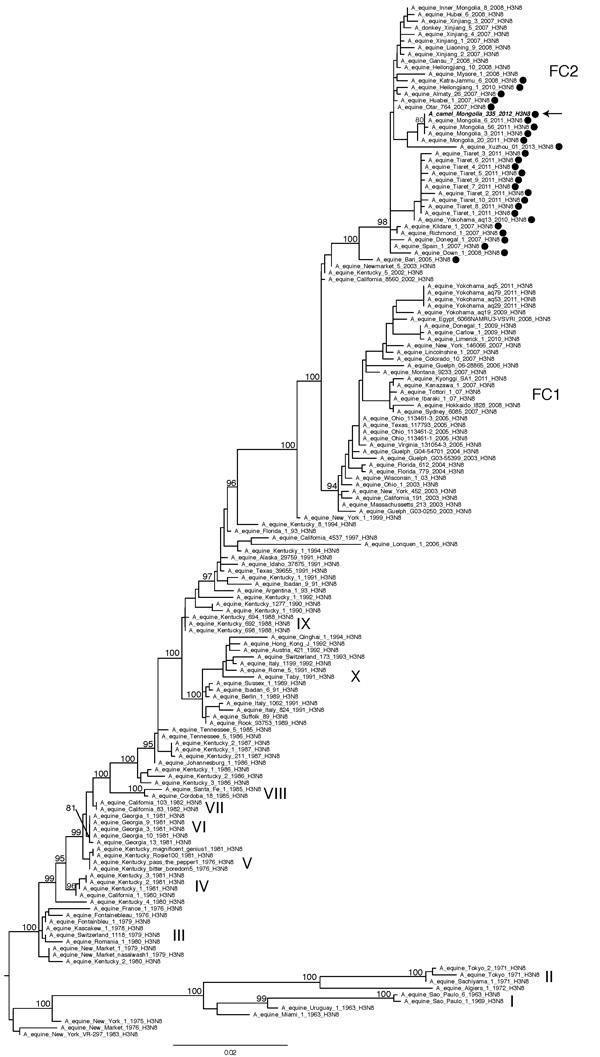
Figure 2. Evolutionary relationships of 155 full-length hemagglutinin sequences from equine A(H3N8)viruses collected globally and A/camel/Mongolia/335/2012 (arrow). The 2 clades associated with most recent equine influenza A(H3N8) viruses, Florida clade 1 and Florida clade 2, are denoted as FC1 and FC2, respectively, and with nomenclature adopted previously (13). The maximum-likelihood tree is midpoint rooted for clarity, and all branch lengths are drawn to scale. High (>70) bootstrap values are provided for key nodes. Hemagglutinin sequences containing a 2aa insertion are identified with a solid black circle. Scale bar indicates nucleotide substitutions per site.
References
- Waddell GH, Teigland MB, Sigel MM. A new influenza virus associated with equine respiratory disease. J Am Vet Med Assoc. 1963;143:587–90 .PubMedGoogle Scholar
- Crawford PC, Dubovi EJ, Castleman WL, Stephenson I, Gibbs EP, Chen L, Transmission of equine influenza virus to dogs. Science. 2005;310:482–5. DOIPubMedGoogle Scholar
- Hayward JJ, Dubovi EJ, Scarlett JM, Janeczko S, Holmes EC, Parrish CR. Microevolution of canine influenza virus in shelters and its molecular epidemiology in the United States. J Virol. 2010;84:12636–45. DOIPubMedGoogle Scholar
- Yondon M, Heil GL, Burks JP, Zayat B, Waltzek TB, Jamiyan BO, Isolation and characterization of H3N8 equine influenza A virus associated with the 2011 epizootic in Mongolia. Influenza Other Respir Viruses. 2013;7:659–65.
- Anchlan D, Ludwig S, Nymadawa P, Mendsaikhan J, Scholtissek C. Previous H1N1 influenza A viruses circulating in the Mongolian population. Arch Virol. 1996;141:1553–69. DOIPubMedGoogle Scholar
- Yamnikova SS, Mandler J, Bekh-Ochir ZH, Dachtzeren P, Ludwig S, Lvov DK, A reassortant H1N1 influenza A virus caused fatal epizootics among camels in Mongolia. Virology. 1993;197:558–63. DOIPubMedGoogle Scholar
- Caffar Elamin MA, Kheir SA. Detection of influenza antibody in animal sera from Kassala region, Sudan, by agar gel diffusion test. Rev Elev Med Vet Pays Trop. 1985;38:127–9 .PubMedGoogle Scholar
- Olaleye OD, Baba SS, Omolabu SA. Preliminary survey for antibodies against respiratory viruses among slaughter camels (Camelus dromedarius) in north-eastern Nigeria [in French]. Revue Scientifique et Technique de l'OIE. 1989;8:779–83.
- Haagmans BL, Al Dhahiry SH, Reusken CB, Raj VS, Galiano M, Myers R, Middle East respiratory syndrome coronavirus in dromedary camels: an outbreak investigation. Lancet Infect Dis. 2014;14:140–5. DOIPubMedGoogle Scholar
- World Health Organization. WHO information for molecular diagnosis of influenza virus in humans—update. 2011 [cited 2013 Jun 10]. http://www.who.int/influenza/resources/documents/molecular_diagnosis_influenza_virus_humans_update_201108.pdf
- Zhou B, Donnelly ME, Scholes DT, St George K, Hatta M, Kawaoka Y, Single-reaction genomic amplification accelerates sequencing and vaccine production for classical and swine origin human influenza A viruses. J Virol. 2009;83:10309–13. DOIPubMedGoogle Scholar
- Stamatakis A. RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics. 2006;22:2688–90. DOIPubMedGoogle Scholar
- Murcia PR, Wood JL, Holmes EC. Genome-scale evolution and phylodynamics of equine H3N8 influenza A virus. J Virol. 2011;85:5312–22. DOIPubMedGoogle Scholar
- Song D, Moon HJ, An DJ, Jeoung HY, Kim H, Yeom MJ, A novel reassortant canine H3N1 influenza virus between pandemic H1N1 and canine H3N2 influenza viruses in Korea. J Gen Virol. 2012;93:551–4. DOIPubMedGoogle Scholar
- Anthony SJ, St Leger JA, Pugliares K, Ip HS, Chan JM, Carpenter ZW, Emergence of fatal avian influenza in New England harbor seals. MBio. 2012;3:e00166–12. DOIPubMedGoogle Scholar
Page created: November 19, 2014
Page updated: November 19, 2014
Page reviewed: November 19, 2014
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.