Volume 20, Number 12—December 2014
Dispatch
Reduction of Baylisascaris procyonis Eggs in Raccoon Latrines, Suburban Chicago, Illinois, USA
Cite This Article
Citation for Media
Abstract
Baylisascaris procyonis, a common roundworm of raccoons, causes severe or fatal human infections, often in suburban areas. To evaluate the effectiveness of a baiting strategy requiring minimal labor, we distributed medicated baits near raccoon latrines in suburban Chicago, Illinois, USA. This strategy lowered B. procyonis prevalence in raccoons, possibly reducing risk to humans.
Modification of landscapes resulting in human-dominated ecosystems is one of the most important drivers of emerging diseases (1). In human-dominated landscapes, the transmission dynamics of diseases often change as host and pathogen population dynamics respond to loss or creation of habitat (1). As a result, contact rates can increase, and humans can be infected (2). Public health officials, veterinarians, and wildlife ecologists increasingly are focusing on mitigation strategies to decrease the potential for human disease in such landscapes (2).
Baylisascaris procyonis roundworms are ubiquitous ascarid parasites of raccoons; prevalence of infection can reach 82% (3). Infected raccoons can shed thousands of eggs daily; transmission generally occurs at raccoon latrines where fecal material accumulates (4). B. procyonis eggs are highly resistant and can remain viable in the environment for years (3). Thirty severe or fatal human cases of baylisascariasis have been reported (5–7; K.R. Kazacos, pers. comm), many of which occurred in human-dominated landscapes (K.R. Kazacos, pers. comm). Therefore, mitigation strategies appropriate for urban and suburban landscapes need to be developed.
Anthelmintic bait distribution has been evaluated for managing B. procyonis (5,6); however, to our knowledge, no B. procyonis mitigation study has been conducted in urban or suburban landscapes, where the risk for transmission to humans is highest. Additionally, previous studies implemented a regimen of latrine removal and substrate sterilization that might not be feasible for resource-restricted wildlife management officials. Furthermore, raccoon core habitat within suburban environments is fragmented with raccoon populations concentrated in preserves interspersed among residential areas (8). As a result, raccoons frequently move throughout adjacent residential areas, creating latrines in close proximity to homes and placing humans at risk to B. procyonis exposure (9). The ability of raccoons to exploit both natural and human-dominated landscapes can present challenges for B. procyonis mitigation, given that bait distribution is generally restricted to natural habitats (10). Our objective was to evaluate the effectiveness of a baiting strategy requiring minimal labor investment in a highly developed suburban region of Chicago, Illinois, USA. By modifying an existing mitigation strategy (5) and implementing it in a suburban landscape, we hope to provide wildlife managers and public health officials with a feasible strategy to decrease B. procyonis infection risks for children living in suburban landscapes.
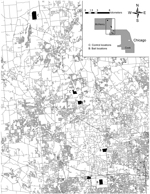
We conducted this study in Cook and McHenry Counties, Illinois, within the Chicago metropolitan area, a landscape characterized as 45% developed, 12% forested and open, 31% agricultural, 10% vacant or wetland, and 2% water (11) (Figure). Our experiment was conducted throughout 6 study sites associated with 6 forest preserves, 3 each in Cook and McHenry Counties. In September and October 2012, we sampled sixty-three 200-m2 quadrats across the 6 study sites. We sampled 8–16 quadrats per study site; the number of quadrats per site were related to the amount of forested habitat within each site (12). All fecal deposits were collected individually for analysis from each latrine within sampling quadrats (13). In contrast to previous studies (5,6), latrine substrates were not sterilized with a propane torch, nor were all latrines located and removed from the treatment sites. Similar to previous studies (5,6), we distributed anthelmintic baits monthly at 1.5 baits/hectare at each of the 3 randomly selected treatment sites for the duration (12 months) of the study. Baits were similar to oral rabies vaccine baits and consisted of a hollow fishmeal polymer food attractant (15 g, 33 mm × 32 mm × 21 mm) in which we placed 180 mg (base) of pyrantel pamoate mixed with marshmallow creme, which was sealed within the hollow chamber with paraffin wax. We distributed baits by hand along transects through forested portions of each site to achieve a relatively even distribution of 1.5/hectare. After 1 year of monthly bait distribution, we repeated latrine sampling in September and October 2013, using an equal sampling effort (sixty-three 200-m2 quadrats) during surveys before and after treatment. All fecal samples were stored at –20°C until they were examined for B. procyonis eggs. We used centrifugal fecal flotation in Sheather sugar solution and microscopic examination to evaluate all samples for eggs (3). Each fecal sample was determined to be positive or negative, and prevalence (proportion positive) was determined for each year, site, and treatment type. We evaluated differences in prevalence by year, site, and treatment type using χ2 analysis with a Fisher exact test. The Wheaton College Institutional Animal Care and Use committee approved this study.
We sampled 63 latrines (2.5 × 10−5/m2) in 2012, and 59 latrines (2.0 × 10−5/m2) in 2013. Latrine density did not differ between sampling years (F = 0.124, df = 1, p = 0.725). Pretreatment (2012) sampling of latrines resulted in 209 fecal samples, and a prevalence of 13% ± 4.56% across sites. The prevalence of B. procyonis roundworms did not differ between treatment (14% ± 6.91%) and control (12% ± 6.02%) sites before placement of anthelmintic baits (χ2 = 0.368, d.f. = 1, p = 0.544) (Table). Posttreatment sampling of latrines resulted in 124 fecal samples, and a prevalence of 11% ± 5.28% across sites. Prevalence across sites did not differ between years (χ2 = 0.44, d.f. = 1, p = 0.602) (Table); however, prevalence differed significantly between treatment (3% ± 3.94%) and control (21% ±1 1.07%) sites after treatment (χ2 = 11.28, d.f. = 1, p<0.001) (Table).
Previous strategies to decrease prevalence of B. procyonis roundworms required removing all latrines and heat sterilizing latrine substrates (5,6). Wildlife managers in urban or suburban settings often do not have the resources to implement such a labor-intensive strategy. We found that a modified strategy that eliminates latrine removal and sterilization but retains the monthly distribution of baits effectively reduced environmental contamination with B. procyonis eggs.
Implementation of this anthelmintic baiting strategy in suburban green spaces might significantly reduce risk for exposure by humans who use green spaces for recreation or live in close proximity to forested areas (9). However, both material and distribution costs can be substantial when such management action is implemented at landscapes scales. This study shows that the monthly distribution of baits and subsequent consumption by raccoons would keep reinfections from reaching patency (32–38 days) (3). For initial disease control, it is recommended that intervals between consecutive bait distributions not exceed the prepatency period (14). Once prevalence is reduced, interbaiting intervals can be extended while reduced prevalence is maintained (14). Although we implemented labor-intensive hand baiting, implementation of bait stations (10) could maintain management efficacy while reducing labor required for bait distribution. Bait stations have been used successfully for oral rabies vaccination in raccoons (10) and may enable more targeted bait distribution in areas of high raccoon density. Our study demonstrated that anthelmintic baiting successfully reduced environmental contamination with B. procyonis eggs; prevalence among treatment sites decreased nearly 80% with 1 year of treatment; however, further study is needed to identify optimal long-term bait distribution frequencies and bait distribution strategies to make anthelmintic baiting a viable and sustainable management solution for B. procyonis control.
Dr Page is a professor of biology at Wheaton College, Wheaton, Illinois. Her research interests include the transmission ecology of B procyonis roundworms and the link between human land use and disease transmission.
Acknowledgments
We thank Chris Anchor, the Cook County Forest Preserve District, and Tom Simpson for facilitating access to the study sites. We also thank Drew Dickelman for providing valuable field assistance.
The Wheaton College Faculty Achievement Award program provided financial support for this study.
References
- Patz JA, Graczyk TK, Geller N, Vittor AY. Effects of environmental change on emerging parasitic diseases. Int J Parasitol. 2000;30:1395–405 . DOIPubMedGoogle Scholar
- Ryser-Degiorgis MP. Wildlife health investigations: needs, challenges and recommendations. BMC Vet Res. 2013;9:223. DOIPubMedGoogle Scholar
- Kazacos KR. Baylisascaris procyonis and related species. In: Samuel WM, Pybus MJ, Kocan AA, editors. Parasitic diseases of wild mammals. Ames (IA): Iowa State University Press; 2001. p. 301–41.
- Page LK, Swihart RK, Kazacos KR. Implications of raccoon latrines in the epizootiology of baylisascariasis. J Wildl Dis. 1999;35:474–80. DOIPubMedGoogle Scholar
- Page K, Beasley JC, Olson ZH, Smyser TJ, Downey M, Kellner KF, Reducing Baylisascaris procyonis roundworm larvae in raccoon latrines. Emerg Infect Dis. 2011;17:90–3. DOIPubMedGoogle Scholar
- Smyser TJ, Page LK, Johnson SA, Hudson CM, Kellner KF, Swihart RK, Management of raccoon roundworm in free-ranging raccoon populations via anthelmintic baiting. J Wildl Manage. 2013;77:1372–9. DOIGoogle Scholar
- Hernandez SM, Glabreath B, Riddle DF, Moore AP, Palamar MB, Levy MG, Baylisascaris procyonis in raccoons (Procyon lotor) from North Carolina and current status of the parasite in the USA. Parasitol Res. 2013;112:693–8. DOIPubMedGoogle Scholar
- Prange S, Gehrt SD, Wiggers EP. Demographic factors contributing to high raccoon densities in urban landscapes. J Wildl Manage. 2003;67:324–33. DOIGoogle Scholar
- Page LK, Anchor C, Luy E, Kron S, Larson G, Madsen L, Backyard raccoon latrines and risk for Baylisascaris procyonis transmission to humans. Emerg Infect Dis. 2009;15:1530–1. DOIPubMedGoogle Scholar
- Boulanger JR, Bigler LL, Curtis PD, Lein DH, Lembo AJ Jr. Comparison of suburban vaccine distribution strategies to control raccoon rabies. J Wildl Dis. 2008;44:1014–23. DOIPubMedGoogle Scholar
- Chicago Metropolitan Agency for Planning. Data bulletin: 2001 land-use inventory for northeastern Illinois. Chicago: Northeastern Illinois Planning Commission; 2006.
- Page LK, Swihart RK, Kazacos KR. Raccoon latrine structure and its potential role in transmission of Baylisascaris procyonis to vertebrates. Am Midl Nat. 1998;140:180–5. DOIGoogle Scholar
- Smyser TJ, Page LK, Rhodes OE Jr. Optimization of raccoon latrine surveys for quantifying exposure to Baylisacaris procyonis. J Wildl Dis. 2010;46:929–33. DOIPubMedGoogle Scholar
- Hegglin D, Deplazes P. Control of Echinococcus multilocularis: strategies, feasibility and cost-benefit analyses. Int J Parasitol. 2013;43:327–37. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleTable of Contents – Volume 20, Number 12—December 2014
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Address for correspondence Kristen Page, 501 College Ave, Biology Department, Wheaton College, Wheaton, IL 60187, USA
Top