Volume 20, Number 5—May 2014
Dispatch
Rickettsia spp. in Seabird Ticks from Western Indian Ocean Islands, 2011–2012
Cite This Article
Citation for Media
Abstract
We found a diversity of Rickettsia spp. in seabird ticks from 6 tropical islands. The bacteria showed strong host specificity and sequence similarity with strains in other regions. Seabird ticks may be key reservoirs for pathogenic Rickettsia spp., and bird hosts may have a role in dispersing ticks and tick-associated infectious agents over large distances.
Rickettsia infections have been explored in many tick species parasitizing a wide variety of animal hosts (1). Rickettsia africae, the agent of African tick-bite fever, has been detected in the seabird tick Amblyomma loculosum (2), suggesting a role for seabirds in the maintenance, transmission, and large-scale dispersal of emerging Rickettsia spp.
In tropical regions, 2 common and abundant tick species are associated with seabird colonies: the hard tick, A. loculosum, and the soft tick, Carios capensis (3). Rickettsia spp. have been documented in these tick species from different areas, although only 1 report is available for tropical areas (2). R. hoogstraalii, R. felis, and an undescribed species (Rickettsia sp. scc49) were detected in C. capensis ticks in South Carolina, USA (4,5). R. hoogstraalii was also reported in C. capensis ticks from Japan (6), raising questions regarding the host and geographic range of Rickettsia spp. in seabird ticks.
To better understand the role of seabirds and seabird ticks as reservoirs and dispersers of pathogens, we investigated Rickettsia spp. infections in A. loculosum and C. capensis ticks. These 2 tick species are commonly found on remote tropical islands of the western Indian Ocean. Using molecular detection techniques, we examined variations in detection rates and estimated the genetic diversity of Rickettsia spp. between islands and between tick species.
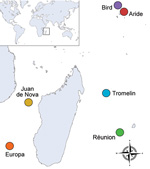
During 2011 and 2012, we collected ticks from seabird colonies on 6 islands of the western Indian Ocean: Aride, Bird, Europa, Juan de Nova, Tromelin, and Réunion Islands (Figure 1; Table). Sample collection on Réunion, Juan de Nova, Tromelin, and Europa Islands was conducted under the approval of the Direction de l'Environnement, de l'Aménagement et du Logement, the Conservatoire du Littoral–Antenne Océan Indien, and the Terres Australes and Antarctiques Françaises. Sample collection on Aride and Bird Islands and their export to Réunion Island were performed with the approval of the Seychelles Bureau of Standards and the Ministry of Environment.
All ticks were morphologically identified as A. loculosum or C. capensis by using standard taxonomic keys (Figure 2). Ticks were individually washed in distilled water and homogenized in Dulbecco modified essential medium or Buffer AVL (QIAGEN, Valencia, CA, USA). Total nucleic acids were extracted by using the EZ1 Viral mini kit v2 and the BioRobot EZ1 System or the QIAmp Viral RNA Mini Kit (all from QIAGEN). We used a commercial cDNA kit (Promega, Madison, WI, USA) to generate cDNAs by reverse transcription. cDNA and PCR, with primers selecting for a 1,064-bp fragment of the citrate synthase–encoding gene (gltA), were used to detect Rickettsia spp. (7). Sequencing of this fragment enabled putative identification of the detected rickettsiae, although complete genotyping is required to fully characterize them (8).
Overall, 143 of the 318 ticks tested were positive for Rickettsia spp., corresponding to a global detection rate (± 95% CI) of 45% (± 5%) (Table); detection from A. loculosum (47% ± 8%) and C. capensis (43% ± 8%) ticks was similar (χ2 = 0.79, df = 1, p = 0.37). However, detection rates differed significantly among the islands (χ2 = 37.96, df = 5, p<0.001). For example, the detection rate was lower in C. capensis ticks on Bird (8% ± 15%) and Juan de Nova (16% ± 11%) Islands compared with other islands. The detection rate in A. loculosum ticks on Réunion Island was particularly high (93% ± 11%) compared with that on other islands.
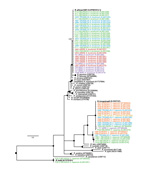
Partial sequencing was performed on the Rickettsia spp. gltA gene from 60 ticks, including ticks from each island and both species. Phylogenetic analyses revealed 7 haplotypes clustering in 4 well-supported clades; some of the haplotypes corresponded to previously described species (Figure 3). No association was found between Rickettsia spp. and collection location (island), but strong host (tick) specificity was observed (Figure 3). All A. loculosum ticks were infected with a Rickettsia strain showing 100% nt identity with the R. africae strain infecting A. loculosum ticks in New Caledonia and with the ESF-5 strain isolated from the cattle tick, A. variegatum, in Ethiopia (GenBank accession no. CP001612.1). Rickettsia spp. in C. capensis ticks had 3 well-supported genetic clusters and, thus, were more diverse. The most frequent Rickettsia sp. in these ticks (64% of the sequences) was a probable strain of R. hoogstraalii. Other haplotypes corresponded to an unknown Rickettsia sp. that was closely related to R. hoogstraalii and to a probable strain of R. bellii (28% and 8% of the sequences, respectively). The haplotype of R. bellii was detected only on Réunion Island in 2 C. capensis ticks collected within the same nest of the wedge-tailed shearwater (Puffinus pacificus).
We provide evidence of rickettsia infection in seabird ticks from 6 islands in the western Indian Ocean. Although the tick infection rate varied among the islands, Rickettsia spp. were detected in all studied seabird populations and overall Rickettsia infection rates were comparable to those reported for ticks parasitizing terrestrial hosts (9).
Phylogenetic analyses revealed a diversity of Rickettsia spp. showing strong host specificity. The most frequently detected Rickettsia sp. was R. africae, the causative agent of African tick-bite fever. R. africae was found only in the hard tick, A. loculosum. These findings support the hypothesis that R. africae is well adapted to this tick host, which may act as a major reservoir for the pathogen (8). Moreover, the similarity of R. africae sequences from our study with those of strains found in other parts of the world indicates that host shifts may be frequent for this bacterium, facilitating its spread in different tick populations. A better understanding of exchanges between wild and domestic fauna and the possible emergence of R. africae at regional levels will require further investigations of the link between R. africae in seabird ticks and in cattle ticks (10).
We detected 2, possibly 3, Rickettsia spp. in C. capensis ticks; this genetic diversity was higher than that detected in A. loculosum ticks. In accordance with previous descriptions (4,6), R. hoogstraalii was the most common Rickettsia species infecting C. capensis ticks, confirming that this tick species likely represents a major host reservoir for this bacterium. An unclassified rickettsial lineage, genetically related to R. hoogstraalii, was also identified in C. capensis from Réunion, Europa, and Tromelin Islands, showing that 2 distinct Rickettsia spp. cocirculate in these tick populations. Additional sampling and further genetic characterization (8) are required to better describe the distribution of this lineage and its relationships to R. hoogstraalii.
R. bellii has mainly been documented in the Americas, but recent studies have detected the presence of closely related R. bellii strains in Australia (11) and Europe (12). Although we detected R. bellii in only 2 ticks on 1 island, our study findings show the potential for broad distribution of this bacterium because of its large arthropod host range (13). The R. bellii genome has been completely sequenced (13), but current knowledge on its epidemiology and pathogenicity in humans remains to be evaluated. In addition, our study was restricted to analysis of the gltA gene, so additional genotyping studies are needed to fully characterize the Rickettsia spp. that we identified (8).
The presence of Rickettsia spp. on remote islands indicates a possible role of bird hosts in the dispersal of ticks and their associated infectious agents over large distances (14,15). An understanding of the historic colonization and current population structure of A. loculosum and C. capensis ticks, together with knowledge of bird migratory patterns, is required to properly assess the risk for emergence of Rickettsia spp. on these islands.
Dr Dietrich is a postdoctoral research associate in the Center for Research on Emerging Infectious Diseases in the Indian Ocean on Réunion Island. She is interested in parasite biodiversity and the evolutionary ecology of host–parasite interactions.
Acknowledgments
We thank Sébastien Lefort, Aurélien Prudor, Chris Feare, Christine Larose, Gérard Rocamora, and Sarah Temmam for assistance with tick sampling and molecular work.
This work was supported by the Fond Européen de Développement Régional Pathogenes associés à la Faune Sauvage Océan Indien (Programme Opérationnel de Coopération Territoriale 2007–2013; no. 31189) and by the Centre National de la Recherche Scientifique–Institut National de l’Ecologie et de l'Environnement/Terres Australes et Antarctiques Françaises (Iles Eparses PathOrnithoTiques project). Postdoctoral fellowships (to M.D., C.L., and A.J.) were funded by RUN-Emerge European project funded by European Commission under FP7 program. C.LeR. and M.B. were supported by a training program fellowship from the Federation Environnement–Biodiversité–Santé.
References
- Parola P, Paddok CD, Raoult D. Tick-borne rickettsioses around the world: emerging diseases challenging old concepts. Clin Microbiol Rev. 2005;18:719–56 . DOIPubMedGoogle Scholar
- Eldin C, Mediannikov O, Davoust B, Cabre O, Barré N, Raoult D, Emergence of Rickettsia africae, Oceania. Emerg Infect Dis. 2011;17:100–2. DOIPubMedGoogle Scholar
- Dietrich M, Gómez-Díaz E, McCoy KD. Worldwide distribution and diversity of seabird ticks: implications for the ecology and epidemiology of tick-borne pathogens. Vector Borne Zoonotic Dis. 2011;11:453–70. DOIPubMedGoogle Scholar
- Reeves WK, Loftis AD, Sanders F, Spinks MD, Wills W, Denison AM, Borrelia, Coxiella, and Rickettsia in Carios capensis (Acari: Argasidae) from a brown pelican (Pelecanus occidentalis) rookery in South Carolina, USA. Exp Appl Acarol. 2006;39:321–9. DOIPubMedGoogle Scholar
- Mattila JT, Burkhardt N, Hutcheson J, Munderloh UG, Kurtti TJ. Isolation of cell lines and a rickettsial endosymbiont from the soft tick Carios capensis (Acari: Argasidae: Ornithodorinae). J Med Entomol. 2007;44:1091–101. DOIPubMedGoogle Scholar
- Kawabata H, Ando S, Kishimoto T, Kurane I, Takano A, Nogami S, First detection of Rickettsia in soft-bodied ticks associated with seabirds, Japan. Microbiol Immunol. 2006;50:403–6 . DOIPubMedGoogle Scholar
- Davis MJ, Ying Z, Brunner BR, Pantoja A, Ferwerda FH. Rickettsial relative associated with papaya bunchy top disease. Curr Microbiol. 1998;36:80–4. DOIPubMedGoogle Scholar
- Fournier PE, Dumler JS, Greub G, Zhang J, Wu Y, Raoult D. Gene sequence-based criteria for identification of new Rickettsia isolates and description of Rickettsia heilongjiangensis sp. nov. J Clin Microbiol. 2003;41:5456–65. DOIPubMedGoogle Scholar
- Mediannikov O, Trape JF, Diatta G, Parola P, Fournier PE, Raoult D. Rickettsia africae, western Africa. Emerg Infect Dis. 2010;16:571–3. DOIPubMedGoogle Scholar
- Parola P, Barre N. Rickettsia africae, agent de la fièvre à tique africaine: un pathogène émergent dans les Antilles et l’île de la Réunion. Bull Soc Pathol Exot. 2004;97:193–8 .PubMedGoogle Scholar
- Vilcins IME, Old JM, Deane E. Molecular detection of Rickettsia, Coxiella and Rickettsiella DNA in three native Australian tick species. Exp Appl Acarol. 2009;49:229–42 . DOIPubMedGoogle Scholar
- Sprong H, Wielinga PR, Fonville M, Reusken C, Brandenburg AH, Borgsteede F, Ixodes ricinus ticks are reservoir hosts for Rickettsia helvetica and potentially carry flea-borne Rickettsia species. Parasit Vectors. 2009;2:41–7.
- Ogata H, La Scola B, Audic S, Renesto P, Blanc G, Robert C, Genome sequence of Rickettsia bellii illuminates the role of amoebae in gene exchanges between intracellular pathogens. PLoS Genet. 2006;2:e76 . DOIPubMedGoogle Scholar
- Palomar AM, Santibáñez P, Mazuelas D, Roncero L, Santibáñez S, Portillo A, Role of birds in dispersal of etiologic agents of tick-borne zoonoses, Spain, 2009. Emerg Infect Dis. 2012;18:1188–91. DOIPubMedGoogle Scholar
- Palomar AM, Portillo A, Santibáñez P, Mazuelas D, Arizaga J, Crespo A, Crimean–Congo hemorrhagic fever virus in ticks from migratory birds, Morocco. Emerg Infect Dis. 2013;19:260–3 . DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 20, Number 5—May 2014
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Muriel Dietrich, Centre de Recherche et de Veille sur les maladies émergentes dans l'Océan Indien (CRVOI), Plateforme de Recherche CYROI, 2 rue Maxime Rivière, BP 80005, 97491 Sainte Clotilde, Réunion Island
Top