Volume 20, Number 7—July 2014
Dispatch
Highly Pathogenic Fowlpox Virus in Cutaneously Infected Chickens, China
Cite This Article
Citation for Media
Abstract
We investigated an acute outbreak of the cutaneous form of fowlpox among chickens in China in November 2009. Using pathologic and virologic methods, we identified a novel type of fowlpox virus that carried an integrated genomic sequence of reticuloendotheliosis virus. This highly pathogenic virus could lead to severe ecologic effects and economic losses.
Fowlpox has been reported worldwide as a mild to severe poultry disease (1). Caused by fowlpox virus (FWPV), the disease is primarily found in 2 forms, cutaneous and diphtheritic (2). The cutaneous form is usually mild and characterized by multifocal cutaneous lesions on unfeathered areas of the skin. The more severe diphtheritic form is characterized by fibrous necrotic proliferative lesions on the mucous membranes of the respiratory and gastrointestinal tracts and causes more deaths than the cutaneous form, usually resulting from asphyxiation.
In recent years, fowlpox outbreaks in poultry flocks have been gradually increasing because of an emerging novel type of FWPV (3–5). The pathogenic traits of this virus type are likely enhanced by integrated reticuloendotheliosis virus (REV) sequences of various lengths in the FWPV genome (6–8). Although this variant FWPV has been found widely (7,9–14), the reported illness and death rates from the cutaneous form of fowlpox in chickens have not reached 100%. We investigated a severe outbreak of cutaneous fowlpox in a poultry flock in northeastern China in which all infected chickens died. The flock had not been vaccinated with an FWPV vaccine.
In November 2009, a natural outbreak of the cutaneous form of fowlpox occurred in a poultry flock in Jilin Province in northeastern China (125°35′ E, 43°88′ N). A total of 10,000 brown breeding, 46-day-old chickens (Jilin Zhengda Co., Ltd, Changchun, China) used for egg production were affected. The flock had not received vaccination against FWPV.
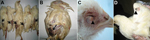
Clinical signs, including listlessness, anorexia, and typical skin pock lesions, were observed in all infected chickens. Lesions types varied in size and type: ulcerated, multifocal, or coalescing proliferative cutaneous exanthema variolosum. The lesions appeared on the skin in unfeathered areas of the backs, the eyelids, and the wings (Figure 1). All of the birds died within 10 days after clinical signs first appeared.
Postmortem examinations were performed for pathol ogic evaluation. Samples submitted for histopathologic examination included skin from the varioliform exanthema areas, trachea, thymus gland, bursa of fabricius, and internal organs. Microscopic examination of skin lesions showed swelling, vacuolation, and characteristic eosinophilic cytoplasmic inclusion bodies in the stratified squamous epithelial cells of the folliculus pili (Technical Appendix Figure 1). No obvious lesions were observed in other organs.
Electron microscopy of the clarified supernatant of the scab specimens collected from the skin of infected chickens showed characteristic FWPV virions, which have an ovoid shape (Technical Appendix Figure 1). We attempted to isolate the virus by injecting the chorioallantoic membranes (CAM) and allantoic cavities of 9-day-old specific pathogen free (SPF) chicken embryos with the scab specimens that were positive for FWPV. White, raised varioles were observed on the CAMs of the embryos 4 days after injection (Technical Appendix Figure 1). Electron microscopy also showed FWPV-shaped virions in the supernatant of the CAMs. After 5 blind passages at 4-day intervals, no other viruses were isolated from the allantoic cavities of the SPF chicken embryos.
We used indirect immunofluorescence and a DF-1 chicken embryo fibroblast cell line to test the ability of the FWPV isolate from the CAMs to invade cells in vitro. Chicken anti-FWPV polyclonal antibody was used as the primary antibody; the secondary antibody was fluorescein isothiocyanate–conjugated goat anti-chicken IgG. Cellular nuclei were stained by using 4′,6-diamidino-2-phenylindole. In some cells, typical bright, DNA-containing poxvirus factories were evident, often coincident with virus antigen–specific green fluorescence, at 3 days postinfection (dpi) (Technical Appendix Figure 1).
Viral genomic DNA was extracted from scab specimens, and PCR amplification was performed immediately by using the specific primers for FWPV P4b gene (P4b Fw1: 5′-GATAGAGGATCGTACATCCA-3′; and P4b Rv1: 5′-CATCTACTCATGACTGGCAA-3′). The size of the product was 1,381 bp (Technical Appendix Figure 2). The amplicons were sequenced, and the obtained P4b gene sequence was submitted to GenBank (accession no. KF875986). We then used the neighbor-joining method in MEGA4 (15) to construct a phylogenetic tree on the basis of the nucleotide sequences of P4b gene with corresponding reference sequences (Technical Appendix Figure 3). The resulting tree showed that the FWPV isolate clustered in the same branch with other FWPVs and that the P4b gene shared a close relationship with other FWPVs (99.9%–100%). This result indicates that the P4b genes were highly conserved among FWPV isolates. No nucleic acid sequences of other potentially pathogenic viruses (i.e., avian influenza virus, Newcastle disease virus, Marek’s disease virus, chicken anemia virus, avian leukosis virus J subgroup, infectious bursal disease virus) were detected by using PCR or reverse transcription PCR. These findings indicate that FWPV may have been the causative pathogen in the infected chickens.
To investigate the possibility of an integration of an REV gene sequence into the FWPV genome, we designed another 2 sets of primers for the amplification of a partial REV env gene and the REV env–FWPV open reading frame 203, which contains the entire REV 3′ long terminal repeat. The primer sequences were as follows: REV-env Fw1, 5′-ACCACTCTCGACTCAAGAAA-3′; REV-env Rv1, 5′-CCACACACAAATACATGACCC-3′; REV env-FWPV 203 Fw1, 5′-GAAATCTTACGAGGCTATGTC-3′; and REV env-FWPV 203 Rv1, 5′-TTCAACCACCAGGCTACATAAAGG-3′. Specific products of the expected sizes, 1,089 bp and 1,437 bp, were amplified from the skin lesions (Technical Appendix Figure 2). The results indicated that the FWPV isolate had integrated partial REV sequences.
We further determined the pathogenesis of the FWPV isolate by experimentally infecting 18-day-old, 53-day-old, and 145-day-old SPF chickens. The experimental groups (10 chickens per group) were inoculated by scarification of the wing and skin scarification into the feather folliculus pili by using purified virus containing 200 50% egg infectious doses of the virus. A control group (10 chickens) was injected with 0.2 mL of phosphate-buffered saline. All inoculated chickens had characteristic skin pock lesions develop at 7–14 dpi (Figure 2) and died at 18–25 dpi; illness and death rates were 100%. Scab specimens were collected at 7, 9, 14, and 20 dpi for histologic examination. The chickens in the control group did not show any clinical signs.
The paraffin sections of scab samples from the SPF chickens inoculated with FWPV were positive not only for FWPV, tested by using a chicken anti-FWPV polyclonal antibody, but also for REV, tested by using a monoclonal antibody that specifically recognized the envelope protein of REV in the cytoplasm of stratified squamous epithelial cells of the folliculus pili by immunohistochemical assay (Technical Appendix Figure 1).
Our investigation of an acute outbreak of the cutaneous form of fowlpox determined that the outbreak was caused by a novel type of FWPV that carried integrated REV genomic sequences. Illness and death rates of up to 100% occurred in this commercial poultry flock in northeast China. Our results show that the novel FWPV we isolated was much more pathogenic than common FWPV strains obtained from other chickens infected with the cutaneous form of fowlpox. This highly pathogenic FWPV variant is a potential threat to chickens and could lead to severe ecologic effects and economic losses. The virulence of this FWPV is probably dependent on the presence of the REV sequences in the FWPV genome, although this conclusion needs experimental confirmation. Because these sequences are also found in less virulent isolates, other determinants may account for this unusual phenotype. Identifying the genomic changes responsible for the increased pathogenicity of this FWPV variant will require considerable effort in sequencing and molecular virology.
Dr Zhao is a lecturer at the College of Veterinary Medicine, Jilin University, Changchun, China. Her research interests are identification and characterization of novel viruses, orf virus infections, and host–virus interactions.
Acknowledgment
This study was supported by the National Natural Science Foundation of China (no. 31072106), the Program for Changjiang Scholars and Innovative Research Team in University (PCSIRT, no. IRT1248), the Science & Technology Development Project Foundation of Jilin Province (no. 20120227), and the China Postdoctoral Science Foundation (no. 2012M510887).
References
- Bolte AL, Meurer J, Kaleta EF. Avian host spectrum of avipoxviruses. Avian Pathol. 1999;28:415–32. DOIGoogle Scholar
- Tripathy DN, Reed WM. Pox. In: Calnek BW, Barnes HJ, Beard CW, et al. Diseases of poultry, 10th ed. Ames (IA): Iowa State University Press; 1997. p. 643–59.
- Hess C, Maegdefrau-Pollan B, Bilic I, Liebhart D, Richter S, Mitsch P, Outbreak of cutaneous form of poxvirus on a commercial turkey farm caused by the species fowlpox. Avian Dis. 2011;55:714–8. DOIPubMedGoogle Scholar
- Tadese T, Reed WM. Detection of specific reticuloendotheliosis virus sequence and protein from REV-integrated fowlpox virus strains. J Virol Methods. 2003;110:99–104. DOIPubMedGoogle Scholar
- Davidson I, Shkoda I, Perk S. Integration of the reticuloendotheliosis virus envelope gene into the poultry fowlpox virus genome is not universal. J Gen Virol. 2008;89:2456–60. DOIPubMedGoogle Scholar
- García M, Narrang N, Reed WM, Fadly AM. Molecular characterization of reticuloendotheliosis virus insertions in the genome of field and vaccine strains of fowl poxvirus. Avian Dis. 2003;47:343–54. DOIPubMedGoogle Scholar
- Biswas SK, Jana C, Chand K, Rehman W, Mondal B. Detection of fowl poxvirus integrated with reticuloendotheliosis virus sequences from an outbreak in backyard chickens in India. Vet Ital. 2011;47:147–53 .PubMedGoogle Scholar
- Kim TJ, Tripathy DN. Reticuloendotheliosis virus integration in the fowl poxvirus genome: not a recent event. Avian Dis. 2001;45:663–9. DOIPubMedGoogle Scholar
- Fadly AM, Witter RL. Comparative evaluation of in vitro and in vivo assays for the detection of reticuloendotheliosis virus as a contaminant in a live virus vaccine of poultry. Avian Dis. 1997;41:695–701. DOIPubMedGoogle Scholar
- Moore KM, Davis JR, Sato T, Yasuda A. Reticuloendotheliosis virus (REV) long terminal repeats incorporated in the genomes of commercial fowl poxvirus vaccines and pigeon poxviruses without indication of the presence of infectious REV. Avian Dis. 2000;44:827–41. DOIPubMedGoogle Scholar
- Singh P, Kim TJ, Tripathy DN. Re-emerging fowlpox: Evaluation of isolates from vaccinated flocks. Avian Pathol. 2000;29:449–55. DOIPubMedGoogle Scholar
- Jia-bo D, Zhi-zhong C, Li-juan Y, Shu-hong S, Shi-jin J. Detection of reticuloendotheliosis virus genomic sequence in the genome of field and vaccine strains of fowl poxvirus. Acta Microbiol Sin. 2004;44:588–92.
- Arathy DS, Tripathy DN, Sabarinath GP, Bhaiyat MI, Chikweto A, Matthew V, Preliminary molecular characterization of a fowl poxvirus isolate in Grenada. Avian Dis. 2010;54:1081–5. DOIPubMedGoogle Scholar
- Jarmin SA, Manvell R, Gough RE, Laidlaw SM, Skinner MA. Retention of 1.2 kbp of ‘novel’ genomic sequence in two European field isolates and some vaccine strains of Fowlpox virus extends open reading frame FWPV 241. J Gen Virol. 2006;87:3545–9. DOIPubMedGoogle Scholar
- Tamura K, Dudley J, Nei M, Kumar S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol. 2007;24:1596–9. DOIPubMedGoogle Scholar
Figures
Cite This Article1These authors contributed equally to this article.
Table of Contents – Volume 20, Number 7—July 2014
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Feng Gao, Key Laboratory of Zoonosis, Ministry of Education, College of Veterinary Medicine, Jilin University, 5333 Xi’an Road, Changchun 130062, China; or
Top