Volume 21, Number 11—November 2015
Dispatch
Pneumococcal Infection among Children before Introduction of 13-Valent Pneumococcal Conjugate Vaccine, Cambodia
Cite This Article
Citation for Media
Abstract
Vaccination of children with pneumococcal conjugate vaccine (PCV13) was initiated in Cambodia in 2015. To determine baseline data, we collected samples from children in 2013 and 2014. PCV13 serotypes accounted for 62.7% of colonizing organisms in outpatients and 88.4% of invasive pneumococci overall; multidrug resistance was common. Thus, effectiveness of vaccination should be high.
Infection with Streptococcus pneumoniae remains a substantial cause of death among children (1). In high-income countries, introduction of pneumococcal conjugate vaccine (PCV) has substantially decreased incidence of invasive pneumococcal disease (IPD) (2). Data for PCV effect in low-income countries are less robust (2). We therefore studied the characteristics of pneumococci responsible for colonization and invasive disease among children in Cambodia before the early 2015 introduction of 13-valent PCV (PCV13).
The study was conducted at Angkor Hospital for Children, Siem Reap, Cambodia. Before enrollment of a child, written consent was obtained from the parent/guardian. Ethical approval was granted by the hospital institutional review board and the Oxford Tropical Research Ethics Committee. For the colonization study, which was conducted in January (cool/dry season) and August (hot/wet season) 2014, colonization surveys were conducted in the outpatient department. Nasopharyngeal swab samples were collected from children 1 month to 15 years of age who had minor illnesses, excluding nonsevere pneumonia, not requiring hospital admission. Children were eligible for enrollment 1 time per survey. For the invasive disease study, which was conducted during August 1, 2013–July 31, 2014, samples were collected from hospitalized children 1 month to 15 years of age who met World Health Organization (WHO) clinical case definitions for pneumonia, meningitis, or sepsis (3). Children readmitted within 14 days were excluded from reenrollment. Samples were processed according to the WHO pneumococcal colonization detection protocol (4). Pneumococci were confirmed by optochin susceptibility and/or bile solubility and were serotyped by latex agglutination (5). Antimicrobial drug susceptibilities were determined according to Clinical and Laboratory Standards Institute guidelines (6). Serotype and antimicrobial drug susceptibilities were also determined for all invasive pneumococcal isolates cultured from patients during January 1, 2013–December 1, 2014. Pneumococci were grouped into vaccine serotypes (PCV13: 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19F, 19A, 23F), nonvaccine serotypes (all others), and nontypeable isolates. Multidrug resistance was defined as resistance to >3 agents (Technical Appendix) (7).
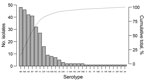
The outpatient colonization study included 974 children (Table 1; Technical Appendix Figure 1). None were known to be HIV infected. Pneumococcal colonization was detected in 601 (61.7%) of children (Technical Appendix Table 1). Colonization prevalence declined with age: 78.6% (206/262) in those 1–11 months, 61.9% (284/459) in those 12–59 months, and 43.9% (111/253) in those ≥5 years of age. The proportion colonized were 75.2% (342/455) in the cool/dry season and 49.9% (259/519) in the hot/wet season (p<0.001). The adjusted odds ratio for colonization in the hot/wet season was 0.38 (95% CI 0.28–0.51, p<0.001) after controlling for age, household size, cohabitation with other young children, current upper respiratory tract symptoms, and recent antimicrobial use. A total of 667 pneumococci were isolated (Figure 1). Among 601 colonized children, >1 serotype was identified in 11.0% (66/601). PCV13 serotypes accounted for 62.7% (418/667), nonvaccine serotypes for 29.5% (197/667), and nontypeable isolates for 7.8% (52/667) of isolates. The proportion of children colonized by PCV13 serotypes was greater among those <5 years of age (70.2% [344/490]) than among older children (48.6% [54/111]); p<0.001; whereas the opposite was true for colonization with nonvaccine serotypes (27.8% [136/490] vs. 48.6% [54/111]; p<0.001). Colonization with nontypeable isolates did not vary by age (data not shown). Overall, 68.8% (459/667) of pneumococci were multidrug resistant: 85.4% of PCV13 isolates, 50.0% of nontypeable isolates, and 38.6% of nonvaccine serotypes (p<0.001). Among colonized children, multidrug-resistant pneumococci were more commonly cultured from children <5 years of age (75.1% [368/490]) than from older children (53.2% [59/111]); p<0.001.
From August 1, 2013, through July 1, 2014, a total of 2,613 cases of medical admissions were screened; of these, 1,009 were included in the analysis (Technical Appendix Figure 1). Median patient age at admission was 1.2 years (interquartile range 0.5–2.4), 56.5% (570/1,009) of patients were male, and 1.4% (14/1,006) were HIV positive. Most cases met the WHO category of severe pneumonia (Technical Appendix Table 2). Pneumococcal colonization was identified in 29.1% (293/1,008) of children from whom a swab sample was obtained (Technical Appendix Table 3). Colonization was less frequent in those who had received >1 dose of an antimicrobial drug (most frequently ceftriaxone) in hospital before the swab sample collection (23.8% [187/785]) than among those who had not (48.5% [95/196]); p<0.001. Colonization was identified in 31.3% (175/559) of children during the dry seasons (hot: March–May; cool: November–February) and in 26.3% (118/449) during the wet season (June–October); p = 0.08. A total of 305 pneumococci were isolated, comprising 27 serotypes plus nontypeable isolates (Figure 2). PCV13 serotypes accounted for 71.1% (217/305) of isolates, nonvaccine serotypes for 15.4% (47/305), and nontypeable isolates for 13.4% (41/305). Multidrug resistance was found in 79.3% (242/305) of isolates.
During 2013–2014, a total of 43 cases of IPD were culture proven (online Technical Appendix). Median patient age was 2.5 years (interquartile range 1.4–8.6). Overall, PCV13 serotypes accounted for 38 (88.4%, 95% CI 74.9–96.1) infections (Table 2). Multidrug resistance was identified in 55.8% (24/43): 22/38 (57.9%) of PCV13 serotypes and 2 (40.0%) of 5 nonvaccine serotypes; p = 0.6. Full resistance profiles are provided in Technical Appendix Table 4.
This study highlights the high potential for reduction of IPD among children after introduction of PCV13 in Cambodia; 88.4% (95% CI 74.9–96.1) of invasive isolates from this 1 surveillance site were serotypes covered by the vaccine. Vaccination should result in decreased drug-resistant pneumococcal infections, although the substantial reservoir of resistance in nonvaccine type and nontypeable pneumococci will probably erode any reduction over time (8–10).
Colonization was high among outpatients and similar to that in other Southeast Asia locations (5,11). Multidrug resistance was common, probably the result of poor regulation of antimicrobial drug use in Cambodia (12); 72.1% of colonizing isolates and 55.8% of invasive isolates were multidrug resistant. For comparison, a recent study of children in Thailand found 31.6% of colonizing pneumococci to be multidrug resistant (13).
The range of serotypes detected in the colonization study was broad but slightly more restricted than that detected in other low-income country studies. In a longitudinal colonization study of refugee infants on the Thailand–Myanmar border, 67 serotypes were identified (5). This finding may reflect the high prevalence of antimicrobial drug use in the community, which would reduce the colonization prevalence of less resistant nonvaccine serotypes. However, the identification of several serotypes emerging as causes of IPD in South Africa, the United Kingdom, and the United States after introduction of PCV13 (e.g., serotypes 15A, 15B/C, 23B, 24F; which accounted for 7.8% of colonizing pneumococci in our study) is noteworthy, indicating the need for close monitoring for changes in colonization and IPD serotype distribution after PCV13 introduction (7,14,15).
The study has several limitations. The absolute number of IPD cases was small, and it was not possible to calculate disease incidence rates. The high prevalence of prehospitalization antimicrobial drug use hampered accurate IPD surveillance. Failure to detect more antimicrobial-drug susceptible nonvaccine type infections as a result of prehospitalization antimicrobial drug use may have falsely elevated the proportion of disease covered by PCV13. The low prevalence of colonization among hospitalized children highlights the need for swab sample collection before in-hospital antimicrobial drug administration for accurate evaluation of colonization in unwell children. Because the study was conducted at 1 site, caution is required when extrapolating the results to the general population of Cambodia. These data provide a baseline against which to monitor effectiveness of vaccinating children with PCV13 in Cambodia.
Dr. Turner in is a clinical microbiologist specializing in pediatric infections. His research interests focus on the epidemiology of vaccine-preventable infections, most notably those caused by Streptococcus pneumoniae, in Southeast Asia.
Acknowledgment
This study was funded by a grant from the Li Ka Shing University of Oxford Global Health Programme and by the Wellcome Trust as part of the Wellcome Trust–Mahidol University–Oxford Tropical Medicine Research Programme.
References
- World Health Organization. Estimated Hib and pneumococcal deaths for children under 5 years of age, 2008 [cited 2014 Mar 9]. http://www.who.int/immunization/monitoring_surveillance/burden/estimates/Pneumo_hib/en/
- Feikin DR, Kagucia EW, Loo JD, Link-Gelles R, Puhan MA, Cherian T, Serotype-specific changes in invasive pneumococcal disease after pneumococcal conjugate vaccine introduction: a pooled analysis of multiple surveillance sites. PLoS Med. 2013;10:e1001517 . DOIPubMedGoogle Scholar
- World Health Organization Coordinated Invasive Bacterial Vaccine Preventable Diseases (IB-VPD) Surveillance Network. Surveillance network case definitions [cited 2014 Dec 4]. http://www.who.int/immunization/monitoring_surveillance/resources/IB-VPD_Case_Defs.pdf?ua=1
- Satzke C, Turner P, Virolainen-Julkunen A, Adrian PV, Antonio M, Hare KM, Standard method for detecting upper respiratory carriage of Streptococcus pneumoniae: updated recommendations from the World Health Organization Pneumococcal Carriage Working Group. Vaccine. 2013;32:165–79 . DOIPubMedGoogle Scholar
- Turner P, Turner C, Jankhot A, Helen N, Lee SJ, Day NP, A longitudinal study of Streptococcus pneumoniae carriage in a cohort of infants and their mothers on the Thailand–Myanmar border. PLoS ONE. 2012;7:e38271 . DOIPubMedGoogle Scholar
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. 17th informational supplement. CLSI document M100–S23. Wayne (PA): The Institute; 2013.
- von Gottberg A, de Gouveia L, Tempia S, Quan V, Meiring S, von Mollendorf C, Effects of vaccination on invasive pneumococcal disease in South Africa. N Engl J Med. 2014;371:1889–99 . DOIPubMedGoogle Scholar
- Dagan R, Klugman KP. Impact of conjugate pneumococcal vaccines on antibiotic resistance. Lancet Infect Dis. 2008;8:785–95 . DOIPubMedGoogle Scholar
- Mitchell PK, Lipsitch M, Hanage WP. Carriage burden, multiple colonization and antibiotic pressure promote emergence of resistant vaccine escape pneumococci. Philos Trans R Soc Lond B Biol Sci. 2015;370:20140342 . DOIPubMedGoogle Scholar
- Chewapreecha C, Harris SR, Croucher NJ, Turner C, Marttinen P, Cheng L, Dense genomic sampling identifies highways of pneumococcal recombination. Nat Genet. 2014;46:305–9 . DOIPubMedGoogle Scholar
- Schultsz C. Vien le M, Campbell JI, Chau NV, Diep TS, Hoang NV, et al. Changes in the nasal carriage of drug-resistant Streptococcus pneumoniae in urban and rural Vietnamese schoolchildren. Trans R Soc Trop Med Hyg. 2007;101:484–92.PubMedGoogle Scholar
- Emary KR, Carter MJ, Pol S, Sona S, Kumar V, Day NP, Urinary antibiotic activity in paediatric patients attending an outpatient department in north-western Cambodia. Trop Med Int Health. 2015;20:24–8 . DOIPubMedGoogle Scholar
- Thummeepak R, Leerach N, Kunthalert D, Tangchaisuriya U, Thanwisai A, Sitthisak S. High prevalence of multi-drug resistant Streptococcus pneumoniae among healthy children in Thailand. J Infect Public Health. 2015;8:274–81 . DOIPubMedGoogle Scholar
- Moore MR, Link-Gelles R, Schaffner W, Lynfield R, Lexau C, Bennett NM, Effect of use of 13-valent pneumococcal conjugate vaccine in children on invasive pneumococcal disease in children and adults in the USA: analysis of multisite, population-based surveillance. Lancet Infect Dis. 2015;15:301–9 . DOIPubMedGoogle Scholar
- Waight PA, Andrews NJ, Ladhani SN, Sheppard CL, Slack MP, Miller E. Effect of the 13-valent pneumococcal conjugate vaccine on invasive pneumococcal disease in England and Wales 4 years after its introduction: an observational cohort study. Lancet Infect Dis. 2015;15:535–43 . DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 21, Number 11—November 2015
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Paul Turner; Cambodia Oxford Medical Research Unit, Microbiology Department, Angkor Hospital for Children, PO Box 50, Siem Reap, Cambodia
Top