Volume 22, Number 5—May 2016
Letter
Hemorrhagic Diathesis in Borrelia recurrentis Infection Imported to Germany
Cite This Article
Citation for Media
To the Editor: Relapsing fevers are paroxysmal bloodstream infections caused by spirochetes of the genus Borrelia. Louseborne relapsing fever (LBRF; i.e., epidemic relapsing fever) is caused by B. recurrentis and transmitted by the human body louse (Pediculus humanus). Soft ticks of the Argasidae family (e.g., Ornithodorus moubata) are vectors for tickborne relapsing fever (TBRF) borreliae, which encompass several human-pathogenic species. In Europe, LBRF was epidemic in the early 20th century but is now rarely seen. We report an infection with B. recurrentis imported to Germany by a Somalian refugee who had high fever and hemoptysis and describe the process of molecular diagnosis.
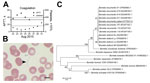
In August 2015, an 18-year-old man sought asylum in Germany after travel through Somalia, Ethiopia, Sudan, Libya, and Italy. He reported general weakness and fever while in Libya, ≈16 days before seeking care, and started coughing up blood after arriving in Italy. At hospital admission in Germany, he had a temperature up to 40.4°C, cough, and hemoptysis; his suspected diagnosis was tuberculosis. No ectoparasites were reported or found on physical examination. Abnormal laboratory findings included relative neutrophilia (91% [reference 39%–77%]), thrombocytopenia (platelets 112 × 10³/μL [reference 160–385 × 10³/μL]), and prolonged activated partial thromboplastin time (APTT) (Figure, panel A). Because of highly elevated levels of C-reactive protein (250 mg/L [reference <5 mg/L]) and procalcitonin (16.4 µg/L [reference <0.5 µg/L]), the patient was treated with ceftriaxone (2g/d intravenously), metronidazole (500 mg/d intravenously), and paracetamol (acetaminophen). Repeated examinations of Giemsa-stained thick and thin blood slides were negative for malaria parasites. Blood cultures, tests for tuberculosis, and PCRs for Rift Valley fever, yellow fever, dengue, and chikungunya viruses also were negative. With antimicrobial therapy, the patient’s fever declined within 12 hours, but platelet counts further decreased and APTT continued to increase (Figure, panel A).
The patient’s symptoms and travel history raised suspicion of a spirochete infection. A plasma sample from his second day in the hospital tested positive for Borrelia spp. 16S DNA by real-time PCR (1). Retrospective microscopy revealed a low number of extracellular spirochetes in thin blood smears (Figure, panel B). The antimicrobial regimen was changed to doxycycline (100 mg 2×/d) on day 7 after admission and, because species identification had not been completed, continued for 10 days. No signs of a Jarisch-Herxheimer reaction were seen. During days 4–9 after admission, APTT, platelet counts (Figure, panel A), and C-reactive protein values returned to normal, and the patient was discharged.
For species identification, we amplified the entire coding sequence of glpQ (glycerophosphodiester phosphodiesterase) with newly designed primers (Technical Appendix). The amplicon was 100% (1,002/1,002 bp) identical to B. recurrentis A1 (GenBank accession no. CP000993.1) and 99% identical (999/1,002 bp) to B. duttonii Ly (GenBank accession no. CP000976.1). A phylogenetic analysis that included 7 published glpQ sequences from B. recurrentis and 4 from B. duttonii suggested that the detected pathogen clustered with B. recurrentis and not B. duttonii (Figure, panel C).
Borreliae have been recognized as a frequent cause of febrile infections in West and East Africa (2). Data on the incidence in immigrants are not available, but the recent increase in asylum seekers from East Africa arriving in Central Europe has increased attention of Borrelia as a pathogen to be included in differential diagnoses of febrile infections (3,4). Because symptom onset in the patient we report occurred in Libya, he most likely acquired infection on the African continent, although local transmission in Europe can occur (4).
Blood slide examination, which would show spirochetes, is routinely requested to detect Plasmodium parasites, but its sensitivity in detecting borreliae is strikingly inferior to molecular tools (15%–56%, depending on laboratory conditions) (1). Pan-Borrelia real-time PCRs enable sensitive detection of DNA in blood samples, followed by sequencing (1) or confirmatory PCRs for relapsing fever Borrelia–specific genes (e.g., glpQ) (5,6). B. recurrentis is genetically highly similar to B. duttonii, suggesting it might be a degraded subset of its tickborne counterpart rather than a distinct species (7). Yet, phylogeny of whole glpQ sequences enables separation of B. recurrentis from B. duttonii on the basis of distinct single-nucleotide variations. Alternatively, differentiation can be achieved by phylogenetic analysis of concatenated partial 16s, glpQ and flaB (flagellin) sequences (5). Differentiation between TBRF and LBRF is crucial for the correct clinical decision on therapy duration, independent of the antimicrobial substance chosen: at least 7 days of treatment is recommended for TBRF to prevent relapses after early invasion of spirochetes into the central nervous system (8), whereas a single-dose regimen is sufficient for LBRF (9), although longer treatment courses tend to be used.
In summary, our report emphasizes that LBRF can be complicated by pulmonary hemorrhages associated with impaired platelet and plasmatic coagulation (10), which can be mistaken for signs of tuberculosis. Considering the poor hygienic conditions among refugees, LBRF has become an important differential diagnosis in Europe in times of increasing migration.
Acknowledgment
We thank the technician teams of the Institutes of Virology and Laboratory Medicine at the University Hospital Marburg for excellent technical assistance.
References
- Parola P, Diatta G, Socolovschi C, Mediannikov O, Tall A, Bassene H, Tick-borne relapsing fever borreliosis, rural Senegal. Emerg Infect Dis. 2011;17:883–5 . DOIPubMedGoogle Scholar
- Elbir H, Raoult D, Drancourt M. Relapsing fever borreliae in Africa. Am J Trop Med Hyg. 2013;89:288–92. DOIPubMedGoogle Scholar
- Wilting KR, Stienstra Y, Sinha B, Braks M, Cornish D, Grundmann H. Louse-borne relapsing fever (Borrelia recurrentis) in asylum seekers from Eritrea, the Netherlands, July 2015. Euro Surveill. 2015;20:21196. DOIPubMedGoogle Scholar
- Lucchini A, Lipani F, Costa C, Scarvaglieri M, Balbiano R, Carosella S, Louseborne relapsing fever among East African refugees, Italy, 2015. Emerg Infect Dis. 2016;22:298–301 and. DOIPubMedGoogle Scholar
- Naddaf SR, Ghazinezhad B, Bahramali G, Cutler SJ. Phylogenetic analysis of the spirochete Borrelia microti, a potential agent of relapsing fever in Iran. J Clin Microbiol. 2012;50:2873–6. DOIPubMedGoogle Scholar
- Schwan TG, Battisti JM, Porcella SF, Raffel SJ, Schrumpf ME, Fischer ER, Glycerol-3-phosphate acquisition in spirochetes: distribution and biological activity of glycerophosphodiester phosphodiesterase (GlpQ) among Borrelia species. J Bacteriol. 2003;185:1346–56. DOIPubMedGoogle Scholar
- Lescot M, Audic S, Robert C, Nguyen TT, Blanc G, Cutler SJ, The genome of Borrelia recurrentis, the agent of deadly louse-borne relapsing fever, is a degraded subset of tick-borne Borrelia duttonii. PLoS Genet. 2008;4:e1000185. DOIPubMedGoogle Scholar
- Cadavid D, Barbour AG. Neuroborreliosis during relapsing fever: review of the clinical manifestations, pathology, and treatment of infections in humans and experimental animals. Clin Infect Dis. 1998;26:151–64. DOIPubMedGoogle Scholar
- Guerrier G, Doherty T. Comparison of antibiotic regimens for treating louse-borne relapsing fever: a meta-analysis. Trans R Soc Trop Med Hyg. 2011;105:483–90. DOIPubMedGoogle Scholar
- Bryceson AD, Parry EH, Perine PL, Warrell DA, Vukotich D, Leithead CS. Louse-borne relapsing fever. Q J Med. 1970;39:129–70 .PubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 22, Number 5—May 2016
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Christian Keller, Institute of Virology, University Hospital Marburg, Hans-Meerwein-Str 2, 35043 Marburg, Germany
Top