Volume 24, Number 10—October 2018
Dispatch
Effectiveness of Whole, Inactivated, Low Pathogenicity Influenza A(H7N9) Vaccine against Antigenically Distinct, Highly Pathogenic H7N9 Virus
Figure 1
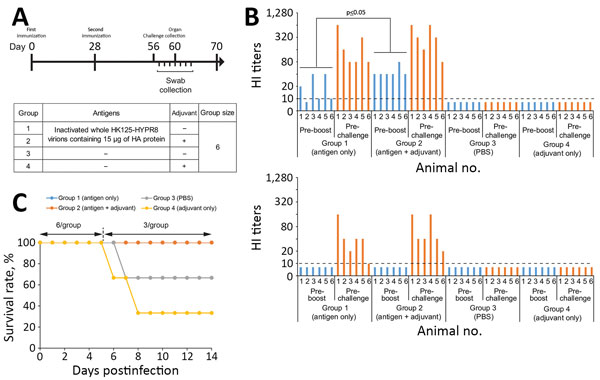
Figure 1. Study design, HI titers after vaccination, and survival rates of vaccinated and nonvaccinated ferrets challenged with highly pathogenic influenza A(H7N9) virus. A) Study design. Six ferrets per group were immunized with inactivated whole HK125–HYPR8 virions containing 15 μg of HA protein without (group 1) or with adjuvant (group 2); control animals were vaccinated with PBS (group 3) or adjuvant (group 4). Animals were vaccinated intramuscularly twice 28 days apart. Twenty-eight days after the second immunization, ferrets were challenged with highly pathogenic H7N9 rGD/3-NA294R virus. Throat and nasal swab specimens were collected on days 1–7 postchallenge; 3 animals per group were euthanized on day 4 postchallenge to assess virus titers in organs. B) HI titers after vaccination. HI assays were performed against HK125–HYPR8 (upper panel) and rGD/3-NA294R (lower panel) with ferret sera collected before the second immunization (preboost) and before challenge (prechallenge). Statistical significance was determined as described in the Technical Appendix. C) Survival rates. Survival was monitored for 14 days after challenge. Because 3 ferrets were euthanized on day 4 postchallenge for organ sampling, the survival rate was calculated on the basis of a group size of n = 3 thereafter. HA, hemagglutinin; HI, hemagglutination inhibition; PBS, phosphate-buffered saline.