Volume 26, Number 9—September 2020
Dispatch
Human Borrelia miyamotoi Infection, Austria
Cite This Article
Citation for Media
Abstract
We report a human case of Borrelia miyamotoi infection diagnosed in Austria. Spirochetes were detected in Giemsa-stained blood smears. The presence of B. miyamotoi in the patient’s blood was confirmed by PCR, and phylogenetic analysis identified an infection with a strain from Europe.
Borrelia miyamotoi is a relapsing fever spirochete transmitted by the same genus of ticks that transmits B. burgdorferi sensu lato (s.l.), Anaplasma phagocytophilum, Babesia species, and tickborne flaviviruses (1–3). B. miyamotoi has been documented in ticks from the United States and in numerous countries in Europe (including Russia), as well as in Japan (1,4–6). B. miyamotoi also has been found in Ixodes scapularis ticks in the northeastern and north-central United States and adjoining areas of Canada, in I. pacificus ticks in the far western United States and British Columbia, in I. ricinus ticks in Europe, and in I. persulcatus ticks in Europe and Asia (1,7,8). I. pavlovskyi and I. ovatus ticks in northern Asia are 2 other species that have been shown to carry B. miyamotoi (9). Endemic areas of B. miyamotoi in Ixodes ticks overlap with those of B. burgdorferi s.l. but with 10-fold lower prevalence (4). Co-infection of Ixodes ticks with both spirochetes also has been identified (9).
Unlike Lyme borreliosis, patients with B. miyamotoi disease typically do not have skin lesions but instead have a nonspecific febrile illness, potentially associated with leukopenia, thrombocytopenia, and elevated liver function parameters (10). Highly immunocompromised patients might have chronic meningitis (2). Untreated patients with B. miyamotoi disease might experience a limited number of recurrent episodes of fever, similar to other relapsing fevers caused by Borrelia infections (6). The same antibiotic regimens used to treat Lyme borreliosis (e.g., 10−14-day courses of oral doxycycline or amoxicillin) are effective for B. miyamotoi disease. Parenteral therapy with ceftriaxone would be preferred for patients with meningitis (3).
Diagnosis of B. miyamotoi disease should be considered in any patient who has fever attacks and resides in or has spent time during tick season in a region where Lyme borreliosis is endemic. Diagnosis requires confirmation using PCR. If the density of spirochetes in the blood is >104/mL, spirochetes might be identified by examining several high-power fields of the blood smear or centifuged sample of cerebrospinal fluid stained with Giemsa or Wright stain (10). Several PCR assays can detect B. miyamotoi in whole blood, plasma, and cerebrospinal fluid by using primers specific for 16S ribosomal RNA and for the flaB and glpQ genes (2,3,8). Serologic testing based on glycerophosphodiester phosphodiesterase antigen of B. miyamotoi that is not found in B. burgdorferi s.l. is highly sensitive but only on convalescent-phase serum specimens (11).
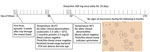
A 51-year-old woman who had a long medical history of seropositive rheumatoid arthritis treated with rituximab sought care at our outpatient clinic for relapsing fever that started 3 months before. Fever episodes occurred every 5 days, and duration ranged from 2 to 3 days. Four weeks before the onset of symptoms, the patient had returned from a 3-week trip through the United States, where, as a tourist, she visited the East and West Coasts and stayed in several national parks. She reported several insect bites and 1 tick bite without erythema migrans that occurred while she was in the United States but did not notice any tick bites before or after her travel. After her return, the patient did not travel abroad again but spent her time in her home in lower Austria.
No abnormal findings were observed on physical examination; in particular, no apparent rash on the skin was noted. Routine laboratory tests performed were normal (including kidney and liver function tests), except for the evidence of leukopenia with 3.3 × 109 cells/L (reference range 4–10 × 109 cells/L) and slightly elevated C-reactive protein of 3.5 mg/dL (reference range <0.5 mg/dL). Multiple blood and urine cultures were negative (Figure 1). An initial peripheral Giemsa-stained blood smear was performed during an afebrile period without any result, but when the test was repeated during the next fever episode, spirochetes were detected between blood cells (Figure 1). Detection of spirochetes in blood smear, which is not typical in cases of B. miyamotoi infection, could be attributable to prolonged spirochetemia likely associated with rituximab therapy. Borrelia spp. was identified in a routinely broad-range bacterial 16S gene-based PCR test (SepsiTest-UMD; Molzym GmbH & Co. KG, https://www.molzym.com). DNA was extracted from EDTA blood, and a real-time PCR assay specific for B. miyamotoi targeting the glpQ gene (5) was positive. To confirm the genospecies present in the patient’s blood, further PCRs targeting the 16S–23S internal transcribed spacer region (12,13), 16S rRNA, and glpQ gene (14,15) were performed, and amplicons were sent to a laboratory for bidirectional sequencing (Microsynth, Vienna, Austria). All PCRs confirmed the presence of B. miyamotoi in the patient’s blood, and all yielded 100% identity to various B. miyamotoi strains in the GenBank database. Using MEGA7 (https://www.megasoftware.net), we constructed a phylogenetic tree for our isolate Bm4667 on the basis of the obtained 16S rRNA gene sequence (Figure 2). All 3 sequences obtained during this investigation were submitted to GenBank (accession no. MN515386 for the 16S rRNA gene, MT396940 for the glpQ gene, and MT396941 for the 16S–23S internal transcribed spacer region).
The patient was treated with 200 mg doxycycline once daily for 2 weeks. On the first day of antibiotic administration, the patient was admitted to our hospital for observation in case of potential Jarisch–Herxheimer reaction. However, during the therapy, no reaction or adverse effects were detected. The patient recovered successfully, and no signs of recurrence were observed in the following 6 months.
We describe a human case of B. miyamotoi infection diagnosed in Austria. Although the patient’s report of a tick bite in the United States suggested that this infection was an imported case, the phylogenetic analysis of the B. miyamotoi strain clearly indicates an infection with a strain from Europe (and possibly Austria). Clinicians should be aware of the possibility of these infections.
Dr. Tobudic is an internal medicine specialist at the Department of Infection Diseases and Tropical Medicine, Medical University of Vienna. Her research interest includes yeast infections, biofilm research, pathomechanism of antimicrobial resistance, and research of tickborne illness.
Acknowledgment
We thank the patient for providing consent to publish information about her case.
References
- Fukunaga M, Takahashi Y, Tsuruta Y, Matsushita O, Ralph D, McClelland M, et al. Genetic and phenotypic analysis of Borrelia miyamotoi sp. nov., isolated from the ixodid tick Ixodes persulcatus, the vector for Lyme disease in Japan. Int J Syst Bacteriol. 1995;45:804–10. DOIPubMedGoogle Scholar
- Gugliotta JL, Goethert HK, Berardi VP, Telford SR III. Meningoencephalitis from Borrelia miyamotoi in an immunocompromised patient. N Engl J Med. 2013;368:240–5. DOIPubMedGoogle Scholar
- Hovius JW, de Wever B, Sohne M, Brouwer MC, Coumou J, Wagemakers A, et al. A case of meningoencephalitis by the relapsing fever spirochaete Borrelia miyamotoi in Europe. Lancet. 2013;382:658. DOIPubMedGoogle Scholar
- Crowder CD, Carolan HE, Rounds MA, Honig V, Mothes B, Haag H, et al. Prevalence of Borrelia miyamotoi in Ixodes ticks in Europe and the United States. Emerg Infect Dis. 2014;20:1678–82. DOIPubMedGoogle Scholar
- Reiter M, Schötta AM, Müller A, Stockinger H, Stanek G. A newly established real-time PCR for detection of Borrelia miyamotoi in Ixodes ricinus ticks. Ticks Tick Borne Dis. 2015;6:303–8. DOIPubMedGoogle Scholar
- Platonov AE, Karan LS, Kolyasnikova NM, Makhneva NA, Toporkova MG, Maleev VV, et al. Humans infected with relapsing fever spirochete Borrelia miyamotoi, Russia. Emerg Infect Dis. 2011;17:1816–23. DOIPubMedGoogle Scholar
- Scoles GA, Papero M, Beati L, Fish D. A relapsing fever group spirochete transmitted by Ixodes scapularis ticks. Vector Borne Zoonotic Dis. 2001;1:21–34. DOIPubMedGoogle Scholar
- Richter D, Schlee DB, Matuschka FR. Relapsing fever-like spirochetes infecting European vector tick of Lyme disease agent. Emerg Infect Dis. 2003;9:697–701. DOIPubMedGoogle Scholar
- Takano A, Toyomane K, Konnai S, Ohashi K, Nakao M, Ito T, et al. Tick surveillance for relapsing fever spirochete Borrelia miyamotoi in Hokkaido, Japan. PLoS One. 2014;9:
e104532 . DOIPubMedGoogle Scholar - Krause PJ, Fish D, Narasimhan S, Barbour AG. Borrelia miyamotoi infection in nature and in humans. Clin Microbiol Infect. 2015;21:631–9. DOIPubMedGoogle Scholar
- Schwan TG, Schrumpf ME, Hinnebusch BJ, Anderson DE Jr, Konkel ME. GlpQ: an antigen for serological discrimination between relapsing fever and Lyme borreliosis. J Clin Microbiol. 1996;34:2483–92. DOIPubMedGoogle Scholar
- Bunikis J, Garpmo U, Tsao J, Berglund J, Fish D, Barbour AG. Sequence typing reveals extensive strain diversity of the Lyme borreliosis agents Borrelia burgdorferi in North America and Borrelia afzelii in Europe. Microbiology. 2004;150:1741–55. DOIPubMedGoogle Scholar
- Wilhelmsson P, Fryland L, Börjesson S, Nordgren J, Bergström S, Ernerudh J, et al. Prevalence and diversity of Borrelia species in ticks that have bitten humans in Sweden. J Clin Microbiol. 2010;48:4169–76. DOIPubMedGoogle Scholar
- Fomenko NV, Livanova NN, Borgoiakov VI, Kozlova IV, Shulaĭkina IV, Pukhovskaia NM, et al. [Detection of Borrelia miyamotoi in ticks Ixodes persulcatus from Russia] [in Russian]. Parazitologiia. 2010;44:201–11.PubMedGoogle Scholar
- Fukunaga M, Hamase A, Okada K, Inoue H, Tsuruta Y, Miyamoto K, et al. Characterization of spirochetes isolated from ticks (Ixodes tanuki, Ixodes turdus, and Ixodes columnae) and comparison of the sequences with those of Borrelia burgdorferi sensu lato strains. Appl Environ Microbiol. 1996;62:2338–44. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: July 29, 2020
Table of Contents – Volume 26, Number 9—September 2020
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Selma Tobudic, Department of Internal Medicine I, Division of Infectious Diseases and Tropical Medicine, Währinger Gürtel 18-20, A-1090 Vienna, Austria
Top