Volume 27, Number 1—January 2021
Research
Post–13-Valent Pneumococcal Conjugate Vaccine Dynamics in Young Children of Serotypes Included in Candidate Extended-Spectrum Conjugate Vaccines
Figure 3
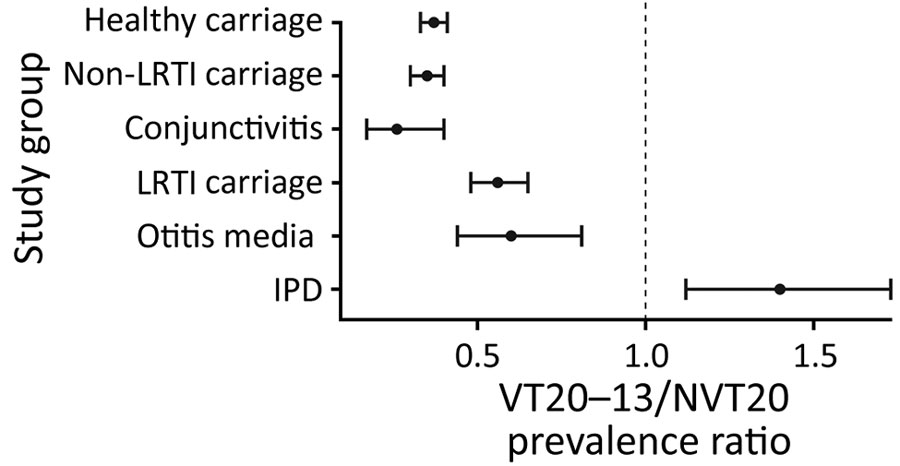
Figure 3. Ratio of prevalence of pneumococcal VT20–13/NVT20 ratio in children <24 months of age, Israel, during the late PCV13 period (2015–2017). Error bars represent 95% CI. IPD, invasive pneumococcal disease; LRTI, lower respiratory tract infection; NVT, nonvaccine serotype; PCV, pneumococcal conjugate vaccine; PCV13, 13-valent PCV; VT, vaccine serotype.
Page created: November 19, 2020
Page updated: December 21, 2020
Page reviewed: December 21, 2020
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.