Volume 28, Supplement—December 2022
SUPPLEMENT ISSUE
Clinical
Effects of Decreased Immunization Coverage for Hepatitis B Virus Caused by COVID-19 in World Health Organization Western Pacific and African Regions, 2020
Figure 1
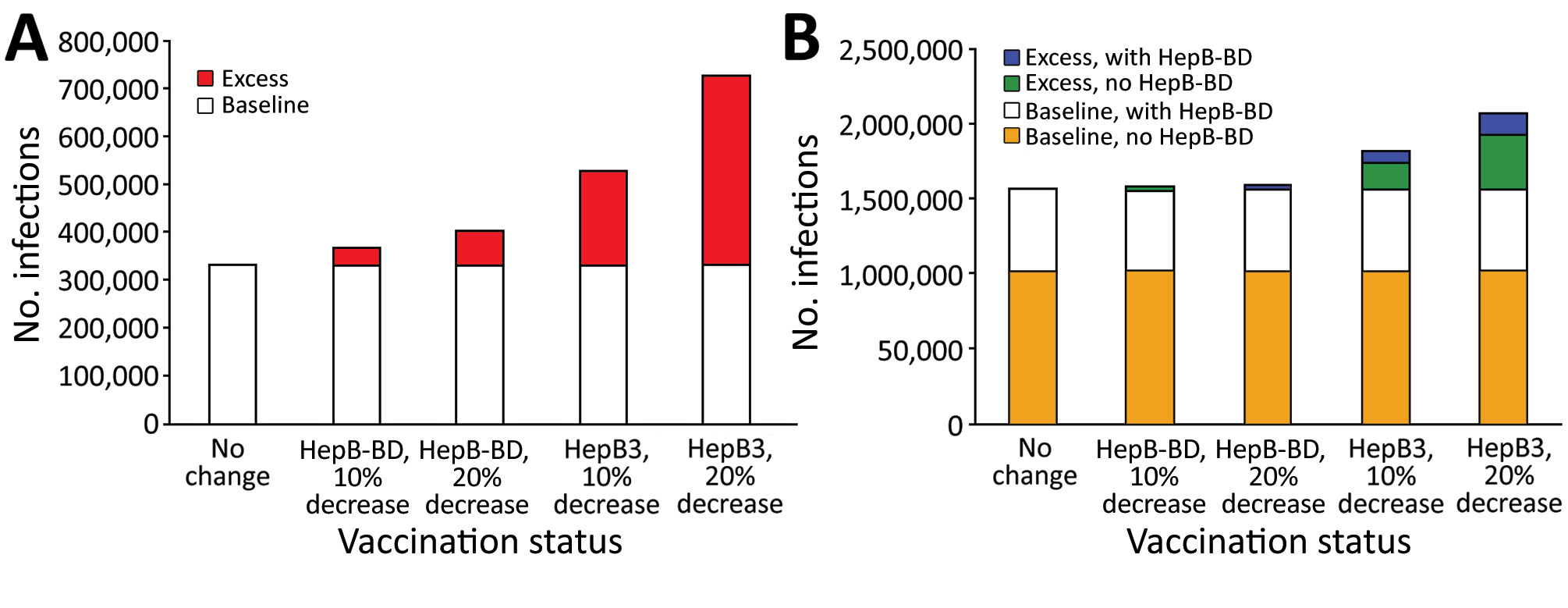
Figure 1. Numbers of additional chronic hepatitis B cases after decreased coverage for hepatitis B vaccine caused by COVID-19 in World Health Organization (WHO) Western Pacific Region (WPR) and African Region (AFR), 2020. We used a mathematical model to estimate the effect of decreased hepatitis B vaccination coverage on hepatitis B virus (HBV) infections among children born in 2020 compared with 2019. A) Total number of chronic HBV infections determined from 2019 data (baseline) and estimates of excess chronic HBV infections from the model after 10% or 20% decrease in HepB-BD or HepB3 vaccination coverage in the World Health Organization WHO Western Pacific Region. All countries and areas in the WPR have introduced HepB-BD, including 2 countries that provide HepB-BD only to infants born to hepatitis B surface antigen–positive mothers. B) Total number of chronic HBV infections (baseline) and estimates of excess chronic HBV infections after 10% or 20% decrease in HepB-BD or HepB3 vaccination coverage in the WHO AFR. Comparisons were made between countries with and without HepB-BD. Fourteen countries in the AFR have introduced HepB-BD, including 1 country that provides HepB BD-only to infants born to hepatitis B surface antigen–positive mothers. HepB-BD coverage data were only available for countries that provided universal birth doses. HepB-BD, birth dose; HepB3, third-dose hepatitis B.
1These authors contributed equally to this article.