Volume 28, Number 2—February 2022
Research
Effectiveness of mRNA BNT162b2 Vaccine 6 Months after Vaccination among Patients in Large Health Maintenance Organization, Israel
Figure 2
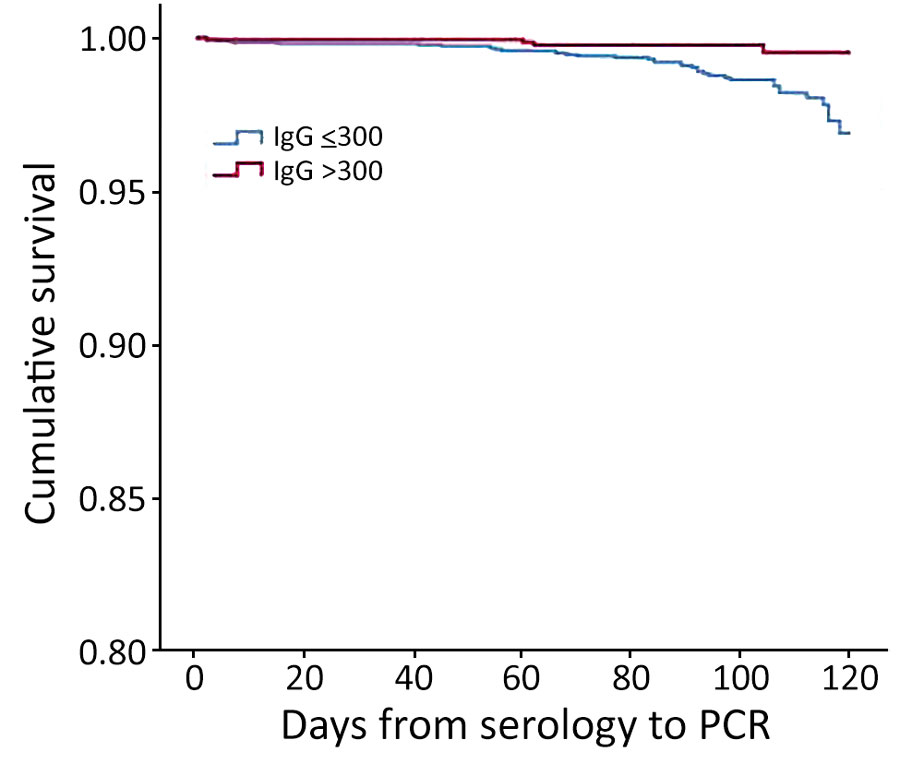
Figure 2. Kaplan-Meier cumulative survival for PCR-positive outcome for population vaccinated with mRNA BNT162b2 vaccine (Pfizer-BioNTech, https://www.pfizer.com) against coronavirus, by antibody (IgG) level, Maccabi Healthcare Services, Israel, June‒July 2021.
Page created: November 16, 2021
Page updated: January 22, 2022
Page reviewed: January 22, 2022
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.