Volume 28, Number 8—August 2022
Dispatch
Novel Reassortant Avian Influenza A(H5N6) Virus, China, 2021
Cite This Article
Citation for Media
Abstract
Although reports of human infection with influenza A(H5N6) increased in 2021, reports of similar H5N6 virus infection in poultry are few. We detected 10 avian influenza A(H5N6) clade 2.3.4.4b viruses in poultry from 4 provinces in China. The viruses showed strong immune-escape capacity and complex genetic reassortment, suggesting further transmission risk.
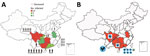
Severe human infection with influenza A(H5N6) virus was identified in China in 2014. During 2014–2020, a total of 26 cases of human infection were laboratory confirmed (1,2). Sporadic cases did not attract widespread attention until 2021 (3,4). During February–October 2021, China reported 24 laboratory-confirmed cases of human infection with H5N6 virus and 5 deaths (Figure 1, panel A); the number of human infections within only 8 months was close to the total for the previous 7 years.
The policy of compulsory poultry immunizations in China was adopted to prevent and control infection with highly pathogenic avian influenza (HPAI) subtype H5Nx (5). Although vaccination can reduce the likelihood of severe clinical disease and reduce shedding of virus in poultry, it cannot prevent sporadic infections with H5N6 virus in waterfowl. Because it is difficult to achieve a qualified 100% rate of H5N6 virus antibodies in waterfowl (6), these birds have become a weak link in prevention and control of the virus. In the context of selection pressure for vaccines and the absence of immunity in waterfowl, antigenic drift causes the H5N6 virus to continuously evolve (7), making currently available H5N6 vaccines ineffective.
On November 27, 2020, an outbreak of influenza A(H5N8) virus infections among wild swans was reported in China, resulting in the death of 2 swans (8,9). Since then, H5N8 clade 2.3.4.4b virus has spread throughout China, resulting in co-endemicity of H5N6 clade 2.3.4.4h/b and H5N8 clade 2.3.4.4b viruses. This 2020 outbreak was not the first outbreak of H5N8 virus in China; the earliest introduction of the virus into China was reported in Liaoning on September 12, 2014 (10,11). Because of China’s immunization policies for poultry, H5N8 virus was quickly eliminated, only to reemerge in 2020.
The reappearance and spread of H5N8 virus is a serious threat to the poultry industry. The ecologic environment of the virus has been altered, given the increasing number of influenza A(H5N6) cases in humans. The current prevalence and mode of virus reassortment is of great concern. We discovered a novel H5N6 virus that has spread throughout the poultry industry, and similar viruses caused a sharp rise in human infections.
In June 2021, we isolated an H5N6 virus from a sick duck on a live poultry farm in Chengdu, Sichuan, China. In July 2021, we isolated another H5N6 virus from a dead chicken in the backyard of a human patient with confirmed infection in Chongqing. In August 2021, we detected an H5N6 virus on a chicken farm in Maoming City and another on a goose farm in Huizhou City (both cities in Guangdong, China). In September 2021, we detected an H5N6 virus on a chicken farm in Qinzhou, Guangxi. Last, in October 2021, we detected 5 H5N6 viruses at live poultry markets in Dongguan City of Guangdong Province (3 from ducks, 1 from a goose, and 1 from the environment) (Figure 1, panel B).
To examine the genetic relationships of the viruses, we sequenced the genomes of the 10 H5N6 viruses and constructed maximum-likelihood phylogenetic trees divided into H5N6 epidemic clades (12), according to the protocol established by the World Health Organization (Figure 2, panel A). The H5N6 virus fell within 8 hemagglutinin (HA) clades (2.3.4.4a to 2.3.4.4h). The similarity between the HA genes of all 10 viruses was 99.1%–100%, and all belonged to clade 2.3.4.4b. The HA genes of all the strains had the typical HPAI virus amino acid sequence RRKR↓GLF at the cleavage site (Appendix Table 1). All viruses contain the S137A and T192I mutations in the HA gene, which enable it to bind to the human α-2, 6-linked sialic acid receptor, thereby increasing human susceptibility to the virus (8,13). Mammalian adaptive mutations, such as T33K, L89V, and G309D (14), were detected in the polymerase basic (PB) 2 gene of all strains, which increases the virulence of H5 viruses in mice (Table 1). Those variants are uncommon in previously circulating H5N6 (clade 2.3.4.4h) viruses.
Because there was an epidemiologic correlation between the avian virus strains from Chongqing and the human infections in Chongqing, we used Chongqing avian strains as a representative virus for the control analysis with human-origin virus sequences (Table 2). We deemed this approach to be the appropriate way to reveal links between the human-origin and avian-origin viruses. We found many similarities between the PB2, polymerase acidic, and nonstructural genes of the Chongqing avian-origin strain and those in influenza A(H3N2) virus, which suggests that the novel virus reassorted with H3N2 virus. The PB1 gene is derived from influenza A(H1N2) virus. The HA gene of the Chongqing H5N6 virus was 99.2% similar to that of the H5N8 virus and the matrix genes were 99.9% similar to those of the H5N8 virus, which leads us to believe that both genes originated from the H5N8 virus. The neuraminidase gene was derived from the H5N6 virus, however, and we speculate that the novel H5N6 virus is a reassortment of the H5N8 and H5N6 viruses. The HA and matrix genes of all viruses were derived from H5N8, which suggests that the novel H5N6 virus may use the gene backbone of the H5N8 virus. Other low-pathogenicity avian influenza viruses have been found to be involved in reassortment, which makes the internal genes of all strains appear complex but inconsistent (Appendix Tables 2–11). Notably, >1 pattern of reassortment seems to be present. Some human-derived strains have internal genes that are close to known H5N6 HPAI virus genes, and others are less closely related (Figure 2, panel B).
Bayesian analysis indicated that the viruses in the Pearl River Delta region (Guangdong), the upper reaches of the Yangtze River (Sichuan/Chongqin), and the middle and lower reaches of the Yangtze River (Hunan/Hangzhou) formed 3 subclades according to geographic characteristics (Appendix Figure 1). Compared with the study of Gu et al. (15), we found that the domestic novel H5N6 virus initially formed 3 subclades and an additional 7 types of genomes (Figure 2, panel B). Current vaccine strains lack protection (hemagglutination inhibition test) against novel H5N6 circulating virus strains (Appendix Table 12). This result differs from that reported by Gu et al. (15), which may result from isolation of the virus from different regions. Our study suggests that the virus poses a high risk for further transmission, which can be reduced or avoided by updating the vaccine strains. In January 2022, the Chinese government introduced new vaccine strains H5-Re14 and rHN5801 to prevent an epidemic of poultry infection with H5N6 virus.
Because reported cases in humans were concentrated in southern China within a short time and most of these cases happened during the noninfluenza season, we suspect that transmissibility or viral load of the novel H5N6 viruses has increased. Furthermore, the virus that was isolated from environmental swab specimens (sewage ditch swab in a live poultry market) in Dongguan indicates that the virus is already present in the surrounding environment, which could increase the likelihood that the virus will infect humans.
At the peak of human cases, we isolated a total of 10 novel reassortment H5N6 virus strains from local poultry and the environment that were highly similar to the H5N6 (human-origin) virus reported during the same period. The human and avian viruses belong to clade 2.3.4.4b. The initial epidemic strains clustered into 3 geographically characterized subclades, and each avian strain had the same mammalian susceptibility mutation. The apparent antigenic differences between the virus and vaccine antiserum suggest further transmission risk.
Dr. Chen is a postgraduate student at South China Agricultural University in Guangzhou, China. His research interests are the epidemiology and pathogenesis of emerging and re-emerging infectious diseases.
Acknowledgments
We acknowledge all contributors who submitted the sequence data on which this research is based to the GISAID EpiFlu Database. All submitters of data may be contacted directly through GISAID (http://www.gisaid.org). Human case reports and data were obtained from http://www.flu.org.cn/scn/news.
This work was supported by the Science and Technology Program of Guangdong Province (2021B1212030015), China Agriculture Research System of MOF and MARA (CARS-41), China National Animal Disease Surveillance and Epidemiological Survey Program(2021-2025) (No.202111), Yunnan Liaoming expert workstation, Basic research project of Yunnan Animal Science and Veterinary Institute (2019RW011).
References
- Bi F, Jiang L, Huang L, Wei J, Pan X, Ju Y, et al. Genetic characterization of two human cases infected with the avian influenza A (H5N6) viruses—Guangxi Zhuang Autonomous Region, China, 2021. China CDC Wkly. 2021;3:923–8. DOIPubMedGoogle Scholar
- Xiao C, Xu J, Lan Y, Huang Z, Zhou L, Guo Y, et al. Five independent cases of human infection with avian influenza H5N6—Sichuan Province, China, 2021. China CDC Wkly. 2021;3:751–6. DOIPubMedGoogle Scholar
- Zhu W, Li X, Dong J, Bo H, Liu J, Yang J, et al. Epidemiologic, clinical, and genetic characteristics of human infections with influenza A(H5N6) viruses, China. Emerg Infect Dis. 2022;28:1332–44. DOIPubMedGoogle Scholar
- Jiang W, Dong C, Liu S, Peng C, Yin X, Liang S, et al. Emerging novel reassortant influenza A(H5N6) viruses in poultry and humans, China, 2021. Emerg Infect Dis. 2022;28:1064–6. DOIPubMedGoogle Scholar
- Bai R, Sikkema RS, Li CR, Munnink BBO, Wu J, Zou L, et al. Antigenic variation of avian influenza A(H5N6) viruses, Guangdong Province, China, 2014–2018. Emerg Infect Dis. 2019;25:1932–45. DOIPubMedGoogle Scholar
- Qin J, Zhang Y, Shen X, Gong L, Xue C, Cao Y. Biological characteristics and immunological properties in Muscovy ducks of H5N6 virus-like particles composed of HA-TM/HA-TMH3 and M1. Avian Pathol. 2019;48:35–44. DOIPubMedGoogle Scholar
- Chen J, Li X, Xu L, Xie S, Jia W. Health threats from increased antigenicity changes in H5N6-dominant subtypes, 2020 China. J Infect. 2021;83:e9–11. DOIPubMedGoogle Scholar
- Li J, Zhang C, Cao J, Yang Y, Dong H, Cui Y, et al. Re-emergence of H5N8 highly pathogenic avian influenza virus in wild birds, China. Emerg Microbes Infect. 2021;10:1819–23. DOIPubMedGoogle Scholar
- He G, Ming L, Li X, Song Y, Tang L, Ma M, et al. Genetically divergent highly pathogenic avian influenza A(H5N8) viruses in wild birds, eastern China. Emerg Infect Dis. 2021;27:2940–3. DOIPubMedGoogle Scholar
- Zhou LC, Liu J, Pei EL, Xue WJ, Lyu JM, Cai YT, et al. Novel avian influenza A(H5N8) viruses in migratory birds, China, 2013-2014. Emerg Infect Dis. 2016;22:1121–3. DOIPubMedGoogle Scholar
- Poen MJ, Venkatesh D, Bestebroer TM, Vuong O, Scheuer RD, Oude Munnink BB, et al. Co-circulation of genetically distinct highly pathogenic avian influenza A clade 2.3.4.4 (H5N6) viruses in wild waterfowl and poultry in Europe and East Asia, 2017-18. Virus Evol. 2019;5:
vez004 . DOIPubMedGoogle Scholar - Zhang D, Gao F, Jakovlić I, Zou H, Zhang J, Li WX, et al. PhyloSuite: An integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies. Mol Ecol Resour. 2020;20:348–55. DOIPubMedGoogle Scholar
- Huang J, Wu S, Wu W, Liang Y, Zhuang H, Ye Z, et al. The biological characteristics of novel H5N6 highly pathogenic avian influenza virus and its pathogenesis in ducks. Front Microbiol. 2021;12:
628545 . DOIPubMedGoogle Scholar - Wu Y, Lin J, Yang S, Xie Y, Wang M, Chen X, et al. The molecular characteristics of avian influenza viruses (H9N2) derived from air samples in live poultry markets. Infect Genet Evol. 2018;60:191–6. DOIPubMedGoogle Scholar
- Gu W, Shi J, Cui P, Yan C, Zhang Y, Wang C, et al. Novel H5N6 reassortants bearing the clade 2.3.4.4b HA gene of H5N8 virus have been detected in poultry and caused multiple human infections in China. Emerg Microbes Infect. 2022;11:1174–85. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleOriginal Publication Date: July 12, 2022
1These authors contributed equally to this article.
Table of Contents – Volume 28, Number 8—August 2022
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Address for correspondence Weixin Jia and Ming Liao, College of Veterinary Medicine, South China Agricultural University, 483 Wushan Rd, Tianhe, Guangzhou, Guangdong 510642, China
Top