Volume 29, Number 1—January 2023
Dispatch
Efficient Inactivation of Monkeypox Virus by World Health Organization‒Recommended Hand Rub Formulations and Alcohols
Figure 1
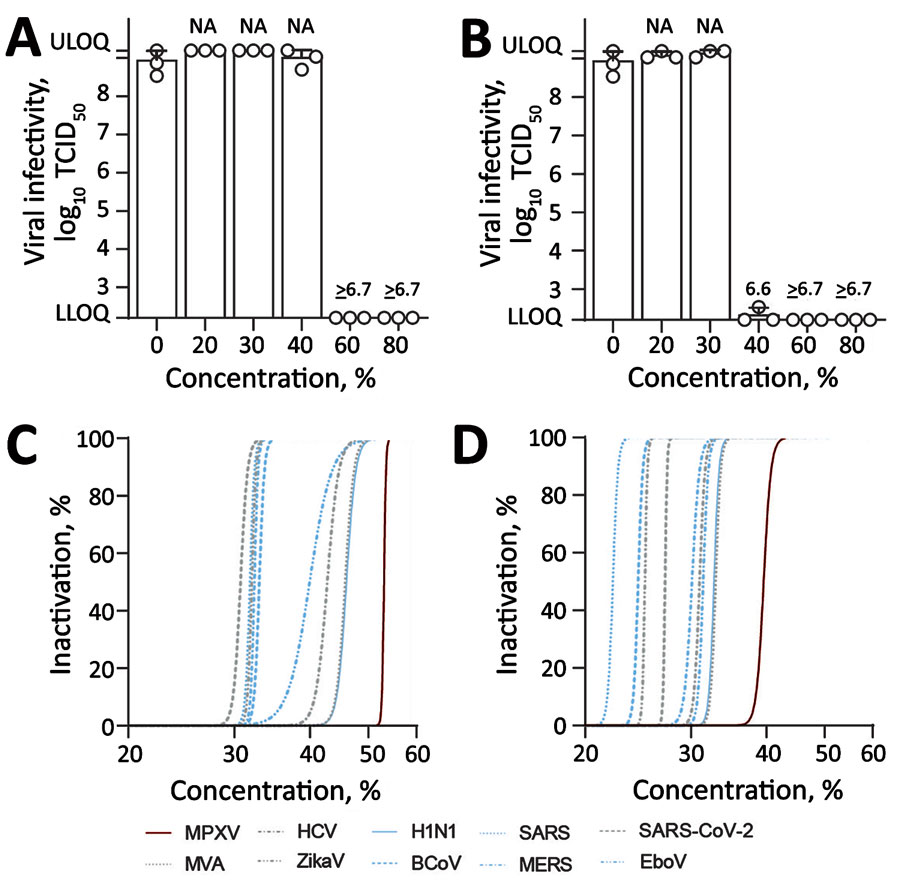
Figure 1. Virucidal activity of World Health Organization (WHO)–recommended hand rub formulations I and II for inactivating MPXV. A, B) Viral infectivity for WHO formulation I (A) and formulation II (B). Means of 3 independent experiments with SDs (error bars) and reduction factors (numbers above the bar) are shown. C, D) Regression analyses of inactivation of MPXV and (re)emerging enveloped or reference viruses, including ZIKV, EBOV, SARS-CoV, SARS-CoV-2, MERS-CoV, influenza A(H1N1) virus, BCoV, HCV, and MVA for WHO formulation I (C) and WHO formulation II (D). Dilutions of the WHO formulations ranging from 0% to 80% with an exposure time of 30 s. Viral titers are displayed as TCID50/mL. BCoV, bovine coronavirus; EBOV, Ebola virus; HCV, hepatitis C virus; MERS-CoV, Middle East respiratory syndrome coronavirus; LLOQ, lower limit of quantification (1.58 × 102 TCID50/mL); MPXV, monkeypox virus; MVA, modified vaccinia Ankara; NA, not applicable; SARS-CoV2, severe acute respiratory syndrome coronavirus 2; TCID50, 50% tissue culture infectious dose; ULOQ, upper limit of quantification (1.58 × 109 TCID50/mL); ZIKV, Zika virus.