Volume 29, Number 4—April 2023
Research Letter
Helicobacter ailurogastricus in Patient with Multiple Refractory Gastric Ulcers, Japan
Cite This Article
Citation for Media
Abstract
We report the isolation of Helicobacter ailurogastricus, a Helicobacter species that infects cats and dogs, from a person with multiple refractory gastric ulcers. In addition to H. suis, which infects pigs, Helicobacter species that infect cats and dogs should be considered as potential gastric pathogens in humans.
A 61-year-old man in Japan had multiple ulcers diagnosed on esophagogastroduodenoscopy (EGD) performed during his annual health checkup and was referred to Tokai University Hospital (Kanagawa, Japan) because of an inadequate therapeutic response. Histologic examination of tissue from the ulcer site showed inflammatory cells and few findings suggestive of malignancy. Hematoxylin and eosin staining showed spiral bacteria resembling a Helicobacter species.
Test results for H. pylori serum antibodies and stool antigen were negative. The patient had onset of epigastric discomfort after his work became busy but attributed his symptoms to his work burden and did not seek medical care. Although he had not taken nonsteroidal antiinflammatory drugs or aspirin, he did not respond to therapy, even with the administration of the antisecretory agent vonoprazan (20 mg), and had multiple refractory gastric ulcers diagnosed.
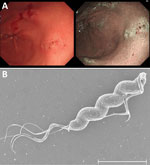
After obtaining informed consent, we enrolled the patient in a clinical trial investigating the effects of non–H. pylori Helicobacter (NHPH) infections on intractable ulcers and gastric mucosa–associated lymphoid tissue lymphoma. On August 24, 2021, we assessed the patient for NHPH by using culture and PCR of gastric biopsy samples collected during EGD (Appendix). EGD showed no atrophy in the background gastric mucosa, healing of the ulcers observed previously, multiple erosions, and residual ulcers in the antrum (Figure, panel A). The PCR test result for NHPH was positive, but the bacterial culture result was negative. On November 30, 2021, a repeat EGD to assess ulcer healing status showed further healing. Repeat culture and PCR tests for NHPH were both positive. We isolated Helicobacter spp. strain NHP21-4376 from the greater curvature of the gastric antrum and NHP21-4377 from the lesser curvature.
The microorganisms had a corkscrew-like spiral form (Figure, panel B) resembling that of Helicobacter suis, the most prevalent NHPH species in the human stomach. We performed whole-genome sequencing of the NHP21-4376 and NHP21-4377 strains by using the Illumina platform (Illumina, https://www.illumina.com) (Appendix). We assembled the Illumina reads de novo by using Shovill 1.1.0 (https://github.com/tseemann/shovill) with the default parameters. We determined the bacterial species by calculating the average nucleotide identity (ANI) using pyani 0.2.12 (https://github.com/widdowquinn/pyani). Strains NHP21-4376 and NHP21-4377 had >98% identity with H. ailurogastricus strains, including the type strain ASB7T, indicating that they were H. ailurogastricus (Appendix Figure 1).
Phylogenetic analysis based on 342 core genes among gastric Helicobacter species also confirmed that NHP21-4376 and NHP21-4377 are in the same clade as H. ailurogastricus strains ASB7T and ASB9 and are distinct from H. suis strains (Appendix Figure 2). We deposited draft genome sequences of H. ailurogastricus into GenBank (NHP21-4376 accession nos. BSCV01000001–64 and NHP21-4377 accession nos. BSCW01000001–66).
Antimicrobial susceptibility tests showed that H. ailurogastricus NHP21-4376 had a high MIC for levofloxacin (Table) and that the NHP21-4376 strain had a Ser to Arg mutation at position 78 in the quinolone resistance–determining region of DNA gyrase A (Appendix Figure 3). This position corresponds to Asn at position 87, where its mutation is responsible for fluoroquinolone resistance in H. pylori (1).
H. suis, which is the most prevalent NHPH species in humans, is believed to originate in pigs. Virulence-associated features were recently shown in H. suis isolates obtained from human stomachs (2); gastric ulcer recurrence was not observed in the patient infected with H. suis after H. suis eradication (2). Furthermore, H. ailurogastricus and H. heilmannii are 2 of the most prevalent NHPH strains infecting the human stomach, after H. suis (3,4). H. ailurogastricus was formerly classified as H. heilmannii. H. heilmannii and H. ailurogastricus are prevalent Helicobacter species that infect the stomachs of cats (5). Moreover, H. ailurogastricus is shown to be the prevalent gastric Helicobacter species infecting the stomach of cats and dogs in Japan (Appendix Table, Figure 4).
In this case, the patient was strongly suspected to have acquired the infection from his cats, although the stool of his pets could not be analyzed because the patient’s consent was not obtained. The patient has not had a recurrence of multiple ulcers but remains positive for H. ailurogastricus. The limitation of this case report is that, although we succeeded in culturing H. ailurogastricus in the stomach of this patient and the drug-susceptibility test has determined the regimen for eradication, we have not yet been able to perform eradication therapy. Therefore, the efficacy of eradication in H. ailurogastricus infections has not been confirmed. H. ailurogastricus eradication therapy will be administered at the next patient visit to prevent ulcer recurrence.
The clinical importance of NHPH infection in the human stomach has been increasing in the post–H. pylori era. Because NHPH species such as H. suis and H. ailurogastricus cannot be detected by most H. pylori diagnostic tests, such as the urea breath test and stool antigen test, NHPH infections should be considered when routine H. pylori tests are negative, despite the presence of inflammatory findings in the gastric mucosa.
Dr. Sano is an instructor at the Division of Gastroenterology and Hepatology, Tokai University School of Medicine. His primary research interests are endoscopic treatment of gastrointestinal diseases and elucidation of pathological mechanisms.
Acknowledgment
This work was supported by MEXT/JSPS KAKENHI (grant no. 20H03667, awarded to H.S.), and by AMED (grant no. JP20fk0108148, awarded to E.R. and H.S.).
References
- Nishizawa T, Suzuki H. Mechanisms of Helicobacter pylori antibiotic resistance and molecular testing. Front Mol Biosci. 2014;1:19. DOIPubMedGoogle Scholar
- Rimbara E, Suzuki M, Matsui H, Nakamura M, Morimoto M, Sasakawa C, et al. Isolation and characterization of Helicobacter suis from human stomach. Proc Natl Acad Sci U S A. 2021;118:
e2026337118 . DOIPubMedGoogle Scholar - Nakamura M, Øverby A, Michimae H, Matsui H, Takahashi S, Mabe K, et al. PCR analysis and specific immunohistochemistry revealing a high prevalence of non-Helicobacter pylori Helicobacters in Helicobacter pylori-negative gastric disease patients in Japan: High susceptibility to an Hp eradication regimen. Helicobacter. 2020;25:
e12700 . DOIPubMedGoogle Scholar - Shafaie S, Kaboosi H, Peyravii Ghadikolaii F. Prevalence of non Helicobacter pylori gastric Helicobacters in Iranian dyspeptic patients. BMC Gastroenterol. 2020;20:190. DOIPubMedGoogle Scholar
- Joosten M, Lindén S, Rossi M, Tay AC, Skoog E, Padra M, et al. Divergence between the highly virulent zoonotic pathogen Helicobacter heilmannii and its closest relative, the low-virulence “Helicobacter ailurogastricus” sp. nov. Infect Immun. 2015;84:293–306. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleOriginal Publication Date: March 18, 2023
Table of Contents – Volume 29, Number 4—April 2023
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Hidekazu Suzuki, Division of Gastroenterology and Hepatology, Department of Internal Medicine, Tokai University School of Medicine, 143 Shimokasuya, Isehara, Kanagawa, 259-1193, Japan
Top