Volume 29, Number 5—May 2023
Research Letter
Cystic Echinococcosis in Northern New Hampshire, USA
Cite This Article
Citation for Media
Abstract
In April 2022 and December 2022, the New Hampshire Department of Health and Human Services confirmed 2 cases of locally acquired human pulmonary cystic echinococcosis caused by Echinococcus granulosus tapeworms. Both patients reported dressing locally hunted moose and exposure to dogs.
Echinococcus granulosus is a zoonotic cestode that lives in the intestine of its definitive host, canids (e.g., dogs, coyotes, foxes, wolves). Embryonated eggs released in canid feces are immediately infectious when consumed by an intermediate host, usually an ungulate (e.g., moose, deer, sheep) or sometimes a human (aberrant intermediate host). An ingested egg hatches in the small intestine, becoming an oncosphere that travels to organs such as the lung, liver, spleen, bone, or brain where it forms a thick-walled cyst. Canids become infected when they ingest flesh and viscera of an infected intermediate host (1). We report 2 human cases of E. granulosus tapeworm infection in northern New Hampshire, USA, during 2022.
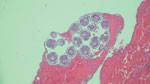
In April 2022, an otherwise healthy middle-aged man (patient 1) from rural northern New Hampshire sought care from his primary-care physician for a routine physical examination. On chest auscultation, the physician heard localized wheezes, prompting chest radiograph and subsequent computed tomography scan; the images showed a noncalcified 4-cm mass in the lower lobe of the right lung. After initial endobronchial biopsy of the mass was nondiagnostic, the patient underwent thoracoscopic lung wedge resection. A gray cystic lesion ruptured during the procedure, releasing clear fluid. The ruptured cyst was excised, and histologic examination showed the presence of a laminated outer cyst wall and numerous daughter cysts, each containing multiple protoscolices with internal hooklets and calcareous corpuscles (Figure), features diagnostic of E. granulosus tapeworm infection. The patient was prescribed a treatment course of albendazole and planned close follow-up, given the high risk for recurrence.
In December 2022, patient 2, an otherwise healthy middle-aged woman, sought medical attention for anaphylaxis that was determined to be caused by a ruptured cyst. Radiography showed 2 lung lesions and 1 small (<5 cm) liver lesion. Results of serum Echinococcus antibody testing was positive. A course of albendazole was prescribed (400 mg 2×/d) and, 4 weeks into therapy, the patient had the first larger pulmonary lesion successfully excised with plans for resection of the second excision.
Both patients reported similar epidemiologic risk factors of dressing locally hunted moose and being exposed to dogs in northern New Hampshire. Neither patient reported relevant travel history outside of New Hampshire. Patient 1 reported observing that the proportion of moose carcasses with extensive cystic lung lesions has increased in recent years. The stool of his 4-year-old domestic dog tested negative for E. granulosus through a commercial veterinary laboratory. We postulate that both patients acquired E. granulosus infection either by consuming produce contaminated with infected canid feces or through consumption of eggs shed in their dogs’ stool. The patients gave verbal permission to publish this report.
In the United States, locally acquired human E. granulosus infections remain rare. However, cases in Arctic and sub-Arctic areas of Canada and Alaska are well described, mainly among Native American populations (2–4). In the contiguous United States, a few human cases are reported annually in Arizona, New Mexico, California, and Utah (4). E. granulosus infections have been described in wildlife of northern New England (5,6); in 2012, surveillance of the hunter-harvested moose population (Alces alces) in Maine documented the presence of the G8 genotype E. granulosus (Canadensis) in 21/54 (39%) of lung sets examined. Domestic dogs and eastern coyotes (Canis latrans) were suggested as definitive hosts, given the absence of wolves in the area (5). Other areas in the contiguous United States where sylvatic cycles of transmission have been confirmed include Idaho, Minnesota, California, and Montana; stable cycles were described mainly in the Arctic and sub-Arctic areas of Canada and Alaska (2). Information about E. granulosus infections in local wild canids in northern New England is lacking.
New Hampshire Department of Health and Human Services has notified healthcare providers caring for patients involved in moose hunting about preventive measures. Such measures include wearing waterproof gloves when dressing moose carcasses, preventing domestic dogs and wild canids from consuming raw viscera, disposing of all viscera in accordance with recommendations from New Hampshire Fish and Game Department, thoroughly cooking all game meat that is fed to dogs, and administering to dogs a deworming agent effective against tapeworms at least twice per year.
Dr. AlSalman completed a fellowship in infectious diseases as well as residency in general preventive medicine and public health at Dartmouth Health in Lebanon, New Hampshire. He is now an infectious diseases consultant at UnityPoint Health in Des Moines, Iowa.
References
- US Centers for Disease Control and Prevention. Echinococcosis—biology. 2019 July 16 [cited 2022 May 12]. https://www.cdc.gov/parasites/echinococcosis/biology.html
- Cerda JR, Buttke DE, Ballweber LR. Echinococcus spp. tapeworms in North America. Emerg Infect Dis. 2018;24:230–5. DOIPubMedGoogle Scholar
- Himsworth CG, Jenkins E, Hill JE, Nsungu M, Ndao M, Andrew Thompson RC, et al. Emergence of sylvatic Echinococcus granulosus as a parasitic zoonosis of public health concern in an indigenous community in Canada. Am J Trop Med Hyg. 2010;82:643–5. DOIPubMedGoogle Scholar
- Deplazes P, Rinaldi L, Alvarez Rojas CA, Torgerson PR, Harandi MF, Romig T, et al. Global distribution of alveolar and cystic echinococcosis. Adv Parasitol. 2017;95:315–493. DOIPubMedGoogle Scholar
- Lichtenwalner A, Adhikari N, Kantar L, Jenkins E, Schurer J. Echinococcus granulosus genotype G8 in Maine moose (Alces alces). Alces. 2014;50:27–33.
- Schurer JM, Bouchard E, Bryant A, Revell S, Chavis G, Lichtenwalner A, et al. Echinococcus in wild canids in Québec (Canada) and Maine (USA). PLoS Negl Trop Dis. 2018;12:
e0006712 . DOIPubMedGoogle Scholar
Figure
Cite This ArticleOriginal Publication Date: March 27, 2023
Table of Contents – Volume 29, Number 5—May 2023
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Ahmad AlSalman, Iowa Methodist Medical Center, Infectious Disease, 1221 Pleasant St, Ste 300, Des Moines IA 50309-1406, USA
Top