Volume 3, Number 3—September 1997
Perspective
Emerging and Reemerging Helminthiases and the Public Health of China
Cite This Article
Citation for Media
Abstract
Despite great strides in their control throughout the People's Republic of China, helminth infections remain an important public health problem. The Institute of Parasitic Diseases of the Chinese Academy of Preventive Medicine, under the guidance of the Chinese Ministry of Health, completed a nationwide survey of more than 1 million people that showed the high prevalence and intensity of intestinal nematode infections; prevalence can sometimes exceed 50% in the Yangtze River valley provinces. Schistosoma japonicum is also a major cause of illness in this region. Attempts to control Chinese helminthic diseases with conventional anthelminthic drugs have been partially thwarted by high posttreatment rates of reinfection. Recently, several new human trematode pathogens have been identified. Novel approaches to chemoprophylaxis and vaccination may alleviate the public health problem caused by Chinese helminths. However, recombinant helminth vaccine development will depend on first cataloguing the extensive genetic diversity of Chinese helminths and candidate vaccine antigens. Evidence from biogeography, genetics, and systematics suggests that the genetic diversification of Chinese helminths and their vectors is an ongoing evolutionary process that began 12 million years ago near the convergence of major Asian river systems. Construction of the Three Gorges Super Dam on the Yangtze River may promote the emergence and reemergence of new helminths and their snail vectors.
Since the early 1900s, hundreds of millions of Chinese living near the Yangtze River have been at high risk for infection with helminths (1,2). Aggressive campaigns to control schistosomiasis among the peasants in Hunan Province and elsewhere in South China were a cornerstone of Chairman Mao's "patriotic health campaigns" (3). During the 1950s and 1960s, entire populations in schistosomiasis-endemic areas were mobilized against Oncomelania snails and recruited to drain rivers and ditches; millions of people were treated with anthelminthics (3). As a result, the number of schistosome-infected persons was reduced from 10 million in 1955 to 1.52 million in 1989 (4); today fewer than 1 million people harbor schistosomes of the Schistosoma japonicum complex. Similarly, lymphatic filariasis caused by Wuchereria bancrofti and Brugia malayi has been reduced by selective or reduced mass chemotherapy control programs, which have included the use of salt medicated with diethylcarbamazine (4). By the year 1994, all 864 filariasis-endemic counties and cities in the country had reached the criterion for effective control of filariasis (less than 1% microfilaria rate in every village) through checkup. Although great strides have been made over the last 40 years in controlling helminths in China, many helminthiases remain public health problems, particularly in the Yangtze River valley provinces (Anhui, Hubei, Hunan, Jiangxi, Jiangsu, Sichuan, Yunnan). The application of modern biotechnology to their study suggests that Chinese helminths are reemerging.
In a nationwide effort to assess the prevalence of parasitic diseases, the Chinese Academy of Preventive Medicine and the Ministry of Health initiated the largest human parasite survey ever undertaken. From 1988 to 1992, 1,477,742 persons were examined in 2,848 study sites in 726 sampled counties (5-8). China's 30 provinces were divided into sectors; the counties and townships in each sector were then grouped into high, intermediate, and low on the basis of socioeconomic parameters (including levels of sanitation, income, and literacy) (8). All long-term residents in each randomly selected site (5,8,9) were asked to submit a fecal specimen for examination (8); compliance was at least 90%. Fecal specimens were collected at village offices or township health centers and examined by personnel trained specifically to participate in the nationwide survey.
The identification and quantitation of helminth eggs was determined by the Kato-Katz thick-smear technique on all slides positive for hookworm and Trichuris, but in only one slide of every 10 Ascaris-positive slides (8). To examine eggs of Enterobius vermicularis, adhesive transparent tape was applied to the perianal area on children under 12 years of age (5,8). To ensure quality control for fecal examinations, random checks of selected slides were made throughout the study. Specimens of previously unidentified or questionable samples were referred to either a provincial institute of parasitic diseases or to the Institute of Parasitic Diseases in Shanghai. Data from the study were ultimately transferred to a VAX computer for processing and analysis (8).
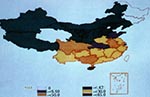
Survey results are summarized in Tables 1-3. An estimated 707 million Chinese (62.6%) were found to harbor one or more parasites (5-8). The overall infection rate for most parasites was higher in females than in males (7). A high number of Chinese were infected with the "unholy trinity" of intestinal nematodes, Ascaris lumbricoides, Trichuris trichiura, and hookworms Ancylostoma duodenale and Necator americanus -- an estimated 531 million, 212 million, and 194 million cases, respectively (8; Table 1). Of the 374,753 children under 12 years of age tested for E. vermicularis, 26.4% were infected (8). Intestinal nematode infections were particularly abundant in the agriculturally intensive Yangtze River provinces (Table 2, Figure 1). In addition, more than 60% of the South China Sea island population of Hainan Province were infected with one or more intestinal nematodes (10-12). A significant percentage of infected persons had moderate and heavy intestinal nematode infections, although most had light infections (Table 3). This pattern of aggregated distribution has been encountered in other disease-endemic areas (8). High hookworm prevalence with clinical disease was found throughout Hainan and the rural areas of the Yangtze River provinces, where adequate moisture and warm temperatures ensure survival of hookworm larvae in the soil. By comparison, Ascaris and Trichuris eggs withstand greater extremes of moisture and temperature, which may explain why ascariasis and trichuriasis are also endemic in urban settings and other harsh environments usually unfavorable for hookworm transmission (13,14).
The survey linked all three intestinal nematodes to foodborne infections, which in China are caused by the common agricultural practice of using uncomposted human manure for fertilizing crops such as corn, sweet potatoes, mulberry trees, and bananas (15-18); foodborne nematode infections are commonly transmitted by the ingestion of Ascaris eggs on sugar cane or A. duodenale hookworm infective larvae on raw vegetables (19). Vegetable garden farmers have the highest prevalence of hookworm infection (31.0%) (5,8). Vegetable growers also have high rates of Ascaris (36.7%) and Trichuris (47.9%) infections (8).
Intestinal nematodes affect maternal-child health. Schoolchildren have the highest prevalence and intensity of ascariasis (48.5%) (5,8), and all three major intestinal nematodes retard the intellectual and physical growth of children. Approximately 66 million women 15 to 49 years of age, many with underlying dietary iron-deficiency anemia, harbor hookworms (8). Cases of infantile hookworm caused by A. duodenale were frequent; children with infantile hookworm failed to thrive and had diarrhea, melena, and extreme pallor resulting from hemoglobin concentrations below 6 g/dl (20,21). Illness caused by heavy hookworm infection in infants was high (20,21). Among the hypothetical sources of infantile hookworm infection is the use of contaminated soil-filled diapers or the ingestion of contaminated breast milk (21).
Current efforts to control intestinal nematode infections with conventional anthelminthic chemotherapeutic agents (e.g., albendazole) or combination therapy with either pyrantel pamoate and oxantel or mebendazole and levamisole have been only moderately successful. All regimens reduce the number of worms (22,23). However, without altering ancient agricultural practices and improving sanitation, reinfection to pretreatment levels may occur within 6 months (24). Moreover, the benzimidazole anthelminthics have shown low efficacy against N. americanus infections in China and have stimulated the migration of adult Ascaris worms. The Institute of Parasitic Diseases and Yale University have embarked on a basic research program to evaluate genetically engineered polypeptides from hookworms as recombinant subunit vaccines as an alternative control approach (25). The goal is to develop a recombinant vaccine that reduces worm levels below the disease-causing threshold. One recombinant molecule from the hookworm Ancylostoma caninum has shown promise at reducing worm levels during preclinical testing in mice (25,26).
Some foodborne helminthic zoonoses are on the rise in China (17). Outbreaks of trichinellosis have increased in Yunnan Province over the last 10 years, in part because of the consumption of raw pork (called "oru" or "shengpi") by some residents (27). The seroprevalence of antibodies to Trichinella spiralis in this region averages 13.5%, and there is evidence that trichinellosis has spread from the southwestern province of Yunnan to other parts of China (5).
Among the foodborne trematode zoonoses, several previously undescribed human infections have been reported, including those caused by Echinostoma angustitestis, Centrocestus formosanus, Echinochasmus fujianensis, and Echinochasmus liliputanus (5,28,29). Clonorchiasis and paragonimiasis remain highly endemic in China. Many of the Clonorchis sinensis infections are in southern China, where nearly 3 million Cantonese (Guangdong Province) are infected because of their raw fish consumption (30). High prevalence rates are also found among Korean minority populations in the northeastern provinces of Heilongjiang, Jilin, and Liaoning (30). Among the reservoir hosts are mammals such as dogs, cats, pigs, and rats. Chronic C. sinensis infection in childhood has been recently linked to delayed physical growth and development, while chronic infection in adulthood is associated with cholangiocarcinoma (30). Paragonimiasis is estimated to occur in 1 million Chinese, and approximately 100 million Chinese are at risk for infection. The study of Chinese Paragonimus infections is in its infancy, having been eclipsed by a comparatively larger amount of investigative work in schistosomiasis. However, preliminary studies suggest that the taxonomy of these human parasites will be extensively revised.
The number and identity of species infecting humans in China is being investigated through molecular phylogenetic studies at the Institute of Parasitic Diseases, in collaboration with the Academy of Natural Sciences and James Cook University. Approximately 25 species, of which some are likely synonyms, have been recorded in China. Paragonimus westermani and Paragonimus skrjabini are most commonly associated with human infection. The former species produces the classical lung-fluke infection, while juveniles of the latter rarely mature in humans but instead undergo extrapulmonary migrations. Some Chinese investigators ascribe P. skrjabini to a distinct genus Pagumogonimus. To complicate matters, both P. westermani and P. skrjabini probably represent complexes of species that differ in clinical importance and biologic properties. Several subspecies of diploid sexually reproducing P. westermani have been proposed on the basis of their clinical picture (31). In addition, a triploid parthenogenetic variety is common in northeastern China, where it is a serious pathogen. Almost nothing is known about strains and species within the P. skrjabini complex. However, novel findings may soon be made. For instance, cercariae of P. skrjabini have been reported from no fewer than 23 snail families distributed among three tribes and two subfamilies of the Pomatiopsidae throughout southern China (32). Barring misidentifications, these records suggest that P. skrjabini must be unique among digeneans in its lack of host specificity. A more likely explanation is that a complex of different, as yet unrecognized, Paragonimus species occurs in these regions (32,33). Also, studies of snail hosts of these parasites are being initiated; most species transmitting P. skrjabini have not been described, and many others are known only from shells and radulae. Snail hosts of P. westermani in China have never been studied in terms of modern systematics.
Asian schistosomiasis is caused by blood flukes of the S. japonicum complex (S. japonicum, S. mekongi, and S. malayensis). Only S. japonicum has been described in China, although identification of unique Chinese strains based on molecular genetic data suggests emergence of new members of this complex (34). Also found in China, S. sinensium, which may occupy its own taxonomic complex, may ultimately be shown to infect humans. Although the national control program through aggressive efforts over the last 30 years has reduced the number of infected persons and cattle, schistosomiasis japonica remains a major public health threat in China (35). The number of infected persons and cattle is estimated at just under 1 million and 70,000, respectively (35). The major endemic foci are in the marsh and lake regions of southern China and the Yangtze River Basin (Hubei, Hunan, Jianxi, and Anhui Provinces), which account for 86% of the cases. Sixty to 100 million persons are at risk in these schistosomiasis-endemic areas. The Oncomelania snail (the intermediate host)-infested areas have been reduced to 3.64 billion m2 in the lakes region and mountainous territories in Yunnan and Sichuan Provinces. Aside from intestinal, liver, and central nervous system damage from heavy infections with S. japonicum, ample evidence has also documented the negative impact of moderate infections on childhood growth and development.
The present policy to control Chinese schistosomiasis relies primarily on two approaches: large-scale chemotherapy with praziquantel (produced in China since 1978) and snail control with molluscicides and environmental modification. Although praziquantel can reduce the incidence of severe forms of the disease, it does not prevent reinfection and may not reverse many of the hepatosplenic complications of heavy S. japonicum infections. Three novel schistosomiasis control programs (using qinghaosu, bromoacetamide, and vaccine) are under investigation at the Institute of Parasitic Diseases.
On the basis of studies suggesting that derivatives of the antimalarial drug qinghaosu (artemisinin) are also effective in animal schistosome infections (36), human clinical investigations have been initiated with oral artemether (37). In field studies conducted in a disease-endemic area in the southern Dongting Lake region of Hunan Province, residents who frequently contacted infested water were pretreated with praziquantel and then, in a randomized pattern, received either artemether or a placebo control (37). Approximately 1 month after the final dose, patients who received artemether had significantly reduced rates of newly acquired schistosomiasis. Further studies are under way to evaluate artemether for schistosomiasis chemoprophylaxis.
Bromoacetamide, a new type of molluscicide, was synthesized and developed in China during the early 1980s and is now being produced (35). The lethal dose of bromoacetamide is 1 ppm for Oncomelania snails -- approximately one tenth that required when pentachlorophenate, a traditional molluscicide, is used (35). At 1 ppm, bromoacetamide has been shown to be nontoxic to fish.
A third control strategy -- schistosome vaccination -- is also being developed at the Institute of Parasitic Diseases, in collaboration with the Queensland Institute of Medical Research. Injection of mice and pigs with a recombinant S. japonicum 26 kDa glutathione-S-transferase (Sjc26GST) vaccine induced a pronounced antifecundity effect after experimental infection with a Chinese strain of S. japonicum (38,39). This finding is important because the eggs released from female schistosomes cause both the pathogenic sequence leading to clinical schistosomiasis and transmission of the infection. Encouraged by these results, the Institute of Parasitic Diseases and the Queensland Institute of Medical Research vaccinated water buffaloes, a principal animal reservoir host for S. japonicum in southern China. Results suggest that immunization of animal reservoir hosts might reduce schistosomiasis transmission to humans. Field studies to test this hypothesis are in progress.
One concern about the development of recombinant antigen-based vaccines is the marked antigenic diversity of different geographic isolates of Chinese schistosomes (40). The genetic diversity of Chinese schistosomes probably parallels an often underappreciated diversity of Oncomelania snail vectors. By both classical and molecular phylogenetic techniques, the Academy of Natural Sciences has provided evidence for the coevolution of S. japonicum with their snail vectors (41). The coevolution of Schistosoma and snail hosts involves reciprocal selective pressures affecting the genetic diversification of both parasites and snails (41). These data may explain the high degree of snail host specificity compared with very loose mammalian host specificity for S. japonicum. Thus, through the careful genetic analysis of host snails, the emergence of S. mekongi within the S. japonicum complex was predicted years before it could be confirmed by using parasites from clinical specimens (42). A similar analysis led to the discovery in China of a separate triculine snail-transmitted S. sinensium complex (43), which may yet reveal new species of human schistosomes. The discovery of a new triculine-transmitted schistosome species may be easily overlooked since Oncomelania (transmitting S. japonicum) is frequently sympatric with triculine snails and a schistosome infection could erroneously be identified as schistosomiasis japonica. Indeed, members of the S. japonicum species complex have worm and egg morphologic characteristics that are virtually identical qualitatively. If snail evolution is a predominant driving force behind the evolution of the trematode parasite, then the marked genetic variation observed among populations of the genus Oncomelania, as it evolved down emerging river systems from Xizang (Tibet) and Yunnan Provinces, China, will drive the evolution of new and emerging schistosomes.
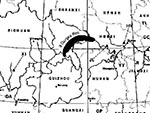
Superimposed on the preexisting coevolutionary forces driving schistosomes and snails is a human-made intervention of enormous scale. Begun in 1994 and scheduled to be completed by the year 2009, the Three Gorges Super Dam, which rises to a height of 180 m on the Yangtze, will affect Hubei and Sichuan Provinces by creating a reservoir area of 50,700 km2 and submerging more than 220 counties extending from Yichang County, Hubei Province, in the east to Jiangjin and Hechuan Counties, Sichuan Province, in the west (Figure 2). The reservoir will displace an estimated 1.4 million people. Several of the environmental changes created by the dam and the resultant reservoir will affect both Oncomelania populations and schistosomiasis transmission. 1) After the dam is completed, the perennial high water level in the reservoir region will elevate the groundwater level and contribute to the breeding and propagation of Oncomelania spp. One site of special concern is the Jianghan Plain, Hubei Province. 2) Alluvial land and beaches will appear near many of the Yangtze River tributaries and provide beds suitable for the development of snails; immigrants to the area exploiting these new environments for aquaculture and agriculture will be exposed to schistosomes. 3) The sand deposits and beach areas of Dongting and Boyang lakes will be altered in a way that may either increase or decrease snail breeding. 4) For the first time in history, lateral canals may permit easy distribution of Oncomelania snails around the gorges from Sichuan to Hubei and downstream.
Investigations by the Chinese Academy of Sciences and the Water Conservancy Committee of the Yangtze River have resulted in programs to evaluate the distribution of Oncomelania snails; the infection rates among children ages 7 to 14 years; changes in census and immigration; and alterations in soil composition, vegetation, rainfall, and topographic features, to identify high risk areas for schistosomiasis transmission. Observation sites will be established in selected schistosomiasis-endemic administrative villages (of approximately 10,000 residents) located midstream and downstream from the Yangtze River - two village sites will be in Jiangsu Province and four in Hunan, Hubei, Jiangxi, and Anhui Provinces. Data will be collected on census, socioeconomic status, daily activities, duration and frequency of water contact, and schistosomiasis infection rates among the residents and domestic animals. In addition, two or three beaches will be selected at each observation site to measure the change in monthly water level, the duration of beach submersion, change in beach vegetation, soil type and water content, snail density and range, and infected-snail density and range.
Chinese helminthiases are intimately connected to the nation's social and ecologic fabric. Traditional agricultural and foodhandling practices account for the high prevalence of ascariasis, hookworm infection, paragonimiasis, and schistosomiasis near the Yangtze River. Increasing evidence also suggests that the major helminthiases are intricately woven into the relationships between families and between mothers and their children (44-47). China is undergoing major social changes due to economic reforms which, in some rural areas, have not improved the access to healthcare (48). Major ecologic changes are anticipated as a consequence of the Three Gorges Super Dam project. The Institute of Parasitic Diseases of the Chinese Academy of Preventive Medicine is now poised to examine the impact of these huge changes on the genetic diversity of helminths and their snail vectors. This information will be used to design novel helminth control measures for specific geographic regions of China.
Dr. Hotez is associate professor of epidemiology and public health and pediatric infectious diseases, Yale University School of Medicine. He heads a laboratory group that develops chemically defined vaccines for helminth infections, including hookworm infection. Dr. Hotez is also assistant program director for the Tropical Medicine Research Center on Emerging Helminthiases in the People's Republic of China, sponsored by the National Institutes of Health.
References
- Faust EC, Meleney HE. Studies on schistosomiasis japonica, with a supplement on the molluscan hosts of the human blood fluke in China and Japan, and species liable to be confused with them. American Journal of Hygiene, Monogr Ser No 3 1924; Baltimore: Johns Hopkins Press.
- Cort WW, Grant JB, Stoll NR, Researches on hookworm in China. American Journal of Hygiene, Monogr Ser No 7 1926; Baltimore: Johns Hopkins University Press.
- Oksenberg MC. Chinese politics and the public health issue. In: Bowers JZ, Purcell EF, editors. Medicine and society in China. New York: Josiah Macy Foundation, 1974;128-61.
- Senhai Y. Control of parasitic diseases in China, current status and prospects. Chin Med J. 1996;109:259–65.PubMedGoogle Scholar
- Yu S-H, Xu L-Q, Jiang Z-X, Xu S-H, Han J-J, Zhu Y-G, Special report, nationwide survey of human parasites in China. Southeast Asian J Trop Med Public Health. 1994;25:4–10.PubMedGoogle Scholar
- Yu SH, Xu LQ, Jiang ZX, Xu SH, Han JJ, Zhu YG, Report on the first nationwide survey of the distribution of human parasites in China. 1. Regional distribution of parasite species. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:241–7.PubMedGoogle Scholar
- Xu LQ, Jiang ZX, Yu SH, Xu S, Huang D, Yang SX, Nationwide survey of the distribution of human parasites in China -- infection with parasite species in human population. Chinese Journal of Parasitology and Parasitic Diseases 1995;1-7.
- Xu L-Q, Yu S-H, Jiang Z-X, Yang J-L, Lai C-Q, Zhang X-J, Soil-transmitted helminthiases: nationwide survey in China. Bull World Health Organ. 1995;73:507–13.PubMedGoogle Scholar
- Cocharne WG. Sampling techniques, 3rd ed. New York: John Wiley and Sons 1977;66-91.
- Xu F, Wu R, Chen J, Guo R. Investigation on human parasite distribution in Hainan Province. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:27–30.PubMedGoogle Scholar
- Chen JZ, Xu FS, Wu RZ, Guo RN, Xing YZ, Huang DL, Investigation on hookworm infection in Hainan Province. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:148–51.
- Chen JZ, Xu FS, Wu RZ, Guo RN, Xing YZ, Huang DL, Investigation and analysis of intestinal parasite infections in rural children in Hainan Province. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:157–9.
- Ma X, Cai L, Fu Y, Huan D. Influence of some factors on prevalence of intestinal parasites in Shanghai. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:30–8.
- Ma X-B, Cai L, Fu Y-F, Huang D-S. Present status of intestinal parasite infections in Pudong new area of Shanghai. Chinese Journal of Parasitology and Parasitic Diseases 1991;9-11.2
- Yang JL, Yang HM, Wang YK, Zhang LL, Yu H, Zhang BX, A survey of epidemiological factors in hookworm, Ascaris and Trichuris infections. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:145–7.
- Xu XR, Cheng YZ, Lin AQ, Guo ZF, Hong L, Zhang XB, Investigation of hookworm infection in banana planting field in Zhangzhou City, Fujian Province. Chinese Journal of Parasitology and Parasitic Diseases. 1991;9:50–2.PubMedGoogle Scholar
- Xu LQ, Jiang ZX, Yu SH, Xu S, Chang J, Wu ZX, Characteristics and recent trends in endemicity of human parasitic diseases in China. Chinese Journal of Parasitology and Parasitic Diseases. 1995;13:214–7.PubMedGoogle Scholar
- Han JJ, Liu C, Liu CY, Wan ZY, Zheng DF, Epidemiological aspects of human parasitoses in Sichuan province. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:8–16.
- Lin J-X, Lin A-Q, Chen B-J, Xu X-R. An investigation of acute Ascaris infection resulted from eating sugarcane in children. Chinese Journal of Parasitology and Parasitic Diseases. 1991;9:48–50.
- Wu C-S, Ni F-Y, Cheng W-J, Wang C-H. Hookworm disease in infants: report of eight cases (with two cases of leukemoid reaction due to infant hookworm disease). Acta Parasitol Sinica. 1966;3:138–40.
- Yu S-H, Jiang Z-X, Xu L-Q. Infantile hookworm disease in China. A review. Acta Trop. 1995;:265–70.PubMedGoogle Scholar
- Xu LQ, Jiang ZX, Yu SH, Ding XM, Bin XH, Yang HF, Treatment of soil-transmitted helminth infections by anthelminthics in current use. Chinese Journal of Parasitology and Parasitic Diseases. 1992;10:95–9.PubMedGoogle Scholar
- Chen ZZ, Fang YY, Huo LC, Liu MZ, Gu SH, Chen XQ, Efficacy of albendazole medicated-salt on treatment of intestinal helminth infections. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:122–4.PubMedGoogle Scholar
- Albonico M, Smith PG, Ercole E, Rate of reinfection with intestinal nematodes after treatment of children with mebendazole or albendazole in a highly endemic area. Trans R Soc Trop Med Hyg. 1995;89:538–41. DOIPubMedGoogle Scholar
- Hotez PJ, Hawdon JM, Ghosh K, Cappello M, Jones BF, Volvovitz F, Molecular approaches to vaccinating against hookworm disease. Pediatr Res. 1996;40:515–21. DOIPubMedGoogle Scholar
- Ghosh K, Hawdon JM, Hotez PJ. Vaccination with alum-precipitated ASP-1 protects mice against challenge infections with infective hookworm (Ancylostoma caninum) larvae. J Infect Dis. 1996;174:1380–3.PubMedGoogle Scholar
- Yang H-M. Studies on epidemiology and control of trichinosis in Yunnan Province. Chinese Journal of Parasitology and Parasitic Diseases. 1991;9:79–82.
- Chen YZ, Xu XL, Chen BJ, Guo ZF, Zhen HY, Lin SS, First report on human infection of Centrocestus formosanus (in Fujian). Chinese Journal of Parasitology and Parasitic Diseases. 1991;9:273.
- Chen YZ, Lin JX, Fang YY, Lin AQ, Chen BJ, Chin J, Discovery on infections of Echinostoma angustitestis in humans. Chin J Zoonoses. 1992;8:7–8.
- Chen M, Lu Y, Hua X, Mott KE. Progress in assessment of morbidity due to Clonorchis sinensis infection: a review of recent literature. Trop Dis Bull. 1994;91:R7–65.
- Xu Z-B. Studies on clinical manifestations, diagnosis and control of paragonimiasis in China. Southeast Asian J Trop Med Public Health. 1991;22(supplement):345–8.PubMedGoogle Scholar
- Lei C, Wang P. Analysis on pathogencity of Paragonimus westermani with diploid form in Lanting of Shaoxing County, Zhejiang Province. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:209–11.
- Davis GM, Chen C-E, Kang Z-B, Liu Y-Y. Snail hosts of Paragonimus in Asia and the Americas. Biomed Environ Sci. 1994;7:369–82.PubMedGoogle Scholar
- He YX, Hu YQ, Yu QF, Tang ZJ. Characteristics of different isolates of Schistosoma japonicum from China in the final hosts. Southeast Asian J Trop Med Public Health. 1991;22:240–1.PubMedGoogle Scholar
- Chen M-G. Control and research on schistosomiasis in China. Chin Med J (Engl). 1995;108:643–6.PubMedGoogle Scholar
- Xiao SH, You JQ, Yang YQ, Wang CZ. Experimental studies on early treatment of schistosomal infection with artemether. Southeast Asian J Trop Public Health. 1995;26:306–18.
- Xiao SH, Shi ZG, Zhuo SJ, Wang CZ, Zhang ZG, Chu B, Field studies on the preventive effect of oral artemether against schistosomal infection. Chin Med J (Engl). 1996;109:272–5.PubMedGoogle Scholar
- Liu S, Song G, Xu Y, Wen Y, McManus DP. Immunization of mice with recombinant Sjc26GST induces a pronounced anti-fecundity effect after experimental infection with Chinese Schistosoma japonicum. Vaccine. 1995;13:603–7. DOIPubMedGoogle Scholar
- Liu SX, Song GC, Xu YX, Yang W, McManus DP. Anti-fecundity immunity induced in pigs vaccinated with recombinant Schistosoma japonicum 26 kDa glutathione-S-transferase. Parasite Immunol. 1995;17:335–40. DOIGoogle Scholar
- Xue H-C, Qiu L-S, He Y-X, Zhang Y-H, Zhu C-W. SDS-PAGE protein pattern and its antigenicity analysis of different isolates of Schistosoma japonicum in China. Chin Med J (Engl). 1994;107:25–9.PubMedGoogle Scholar
- Davis GM. Evolution of prosobranch snails transmitting Asian Schistosoma: coevolution with Schistosoma: a review. Prog Clin Parasitol. 1993;3:145–204.PubMedGoogle Scholar
- Davis GM, Kitikoon V, Temcharoen P. A monograph on Lithoglyphopsis aperta, the snail host of Mekong River schistosomiasis. Malacologia. 1976;15:241–78.PubMedGoogle Scholar
- Davis GM, Greer G. A new genus and two new species of triculinae and the transmission of a malaysian mammalian Schistosoma sp. Proc Acad Nat Sci Philadelphia. 1980;132:245–76.
- Xu LQ, Jiang ZX, Yu SH, Xu SH, Chen SL, Lin JX, Status and strategy of parasitosis control in preventive health care in China. Chinese Journal of Parasitology and Parasitic Diseases. 1995;13:264–8.PubMedGoogle Scholar
- Xu LQ, Jiang ZX, Yu SH, Xu SH, Meng N, Gan YC, Nationwide survey of the distribution of parasites in China -- the characteristics and rules of geographic distribution of human helminth infection. Chinese Journal of Parasitology and Parasitic Diseases. 1995;13:99–103.PubMedGoogle Scholar
- Han J-J, Liu C-H, Yin G-Y, Zheng D-F. Human intestinal parasite infections and their family aggregation in Sichuan. Chinese Journal of Parasitology and Parasitic Diseases. 1991;9:76–9.
- Li BJ, Li YB, Ge FT, Shang LJ, Hou FL, Liu HB, Analysis on family clustering of human intestinal parasitic infections in Hebei Province. [special issue]. Chinese Journal of Parasitology and Parasitic Diseases. 1994;12:215–6.
- Hsiao WCL, Liu Y. Economic reform and health -- lessons from China. N Engl J Med. 1996;335:430–2. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 3, Number 3—September 1997
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Feng Zheng, Institute of Parasitic Diseases, Chinese Academy of Preventive Medicine, 207 Rui Jin Er Lu, Shanghai 200025, P.R. China; fax: 86-21-6-4332670
Top